Advanced One-Pot Synthesis of 6-Methoxy-1-Tetralone for Scalable Steroid Manufacturing
The strategic landscape of steroid drug manufacturing is undergoing a significant transformation, driven by the urgent need for more sustainable and cost-effective synthetic pathways. As detailed in the recent patent CN111333494B, a breakthrough methodology has been established for the synthesis of 6-methoxy-1-tetralone, a pivotal building block in the total synthesis of critical steroid hormones such as mifepristone, norethindrone, and levonorgestrel. Historically, the industry relied heavily on diosgenin extracted from natural sources, but rising costs and supply chain volatility have necessitated a shift toward robust total synthesis routes. This new technology addresses these challenges by introducing a highly efficient, one-pot Friedel-Crafts reaction sequence that bypasses the environmental and safety hazards associated with legacy processes. For R&D directors and procurement leaders, understanding this shift is crucial for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials at a competitive cost structure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
For decades, the industrial standard for producing 6-methoxy-1-tetralone involved a multi-step sequence starting from naphthalene derivatives, a route fraught with significant operational and environmental drawbacks. As illustrated in the traditional pathway below, the process typically requires methylation, followed by high-pressure hydrogenation using nickel catalysts, and finally, a harsh oxidation step utilizing chromium trioxide.
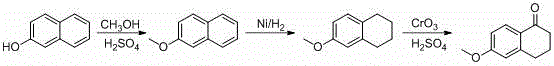
This conventional approach suffers from a notoriously low total yield, often hovering around merely 35 percent, which drastically inflates the cost of goods sold. Furthermore, the reliance on chromium trioxide introduces severe environmental compliance issues, generating toxic heavy metal waste that requires expensive treatment protocols before disposal. The involvement of high-pressure hydrogenation also demands specialized, high-cost reactor equipment and rigorous safety monitoring, creating bottlenecks in production capacity. Additionally, the oxidation step frequently generates difficult-to-remove 4-keto impurities, complicating downstream purification and compromising the final purity profile required for sensitive pharmaceutical applications.
The Novel Approach
In stark contrast, the innovative method disclosed in patent CN111333494B streamlines the entire synthesis into a single, continuous operation that dramatically enhances efficiency and safety. By utilizing anisole as the starting material and reacting it directly with a specific acylating agent in the presence of a Lewis acid, the process achieves cyclization and ketone formation in one pot without the need for intermediate isolation. The reaction scheme below highlights this elegant transformation, where the methoxy group directs the acylation to the para-position, followed by intramolecular alkylation to close the ring.
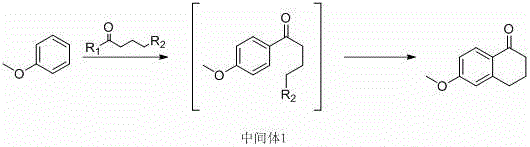
This novel approach eliminates the need for toxic chromium reagents and dangerous hydrogenation steps entirely, replacing them with standard organic synthesis conditions that are far easier to manage on a commercial scale. The result is a substantial improvement in yield, with the patent reporting figures exceeding 90 percent under optimized conditions, alongside a product purity that consistently surpasses 99 percent. By collapsing multiple unit operations into a single reactor vessel, the method not only reduces capital expenditure on equipment but also significantly shortens the overall production cycle time, offering a compelling value proposition for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this technological advancement lies in the precise control of Friedel-Crafts acylation and subsequent alkylation mechanisms, orchestrated by a robust Lewis acid catalyst system. The process initiates with the activation of the acylating agent, such as 4-chlorobutyryl chloride, by the Lewis acid (preferably aluminum trichloride), generating a highly reactive acylium ion species. This electrophile then attacks the electron-rich aromatic ring of anisole. Due to the strong electron-donating nature of the methoxy group, the reaction is highly regioselective for the para-position, especially when conducted at controlled low temperatures between -10°C and 40°C. This temperature control is critical; operating above 50°C can lead to the formation of unwanted ortho-isomers, which are difficult to separate and can degrade the quality of the final steroid API.
Following the initial acylation, the reaction mixture is heated to a higher temperature range of 70°C to 120°C to trigger the second mechanistic phase: intramolecular Friedel-Crafts alkylation. In this step, the alkyl halide tail of the intermediate undergoes cyclization, closing the six-membered ring to form the tetralone structure. The beauty of this mechanism is that the same Lewis acid catalyst facilitates both steps, allowing the reaction to proceed seamlessly without quenching or workup between stages. This continuity prevents the exposure of the sensitive intermediate to moisture or air, thereby minimizing side reactions and ensuring that impurity profiles remain exceptionally clean. The ability to tune the leaving groups on the acylating agent (such as using chlorides or bromides) further allows chemists to optimize reaction kinetics for specific scale-up requirements.
How to Synthesize 6-Methoxy-1-Tetralone Efficiently
Implementing this synthesis route requires careful attention to thermal management and addition rates to maximize the benefits of the one-pot design. The protocol begins by dissolving anisole in a suitable solvent like dichloroethane and cooling the mixture to near 0°C before the slow introduction of the Lewis acid. Once the catalyst is complexed, the acylating agent is added dropwise over a period of 2.0 to 2.5 hours to maintain strict temperature control and prevent exothermic runaway. After the acylation is complete, the temperature is ramped up to induce cyclization, followed by a straightforward aqueous quench and solvent recrystallization to isolate the high-purity product. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized guide below.
- Dissolve anisole in a solvent such as dichloroethane and cool to 0-15°C, then slowly add a Lewis acid like aluminum trichloride.
- Slowly dropwise add the acylating agent (e.g., 4-chlorobutyryl chloride) over 2.0-2.5 hours while maintaining low temperature to form Intermediate 1.
- Raise the temperature to 80-100°C to induce cyclization without isolating the intermediate, then quench with water and purify via solvent recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates into tangible strategic advantages that extend far beyond simple chemical yield improvements. The elimination of hazardous reagents like chromium trioxide removes a major regulatory burden and significantly lowers the costs associated with waste disposal and environmental compliance. Furthermore, the simplification of the process from a multi-step sequence to a one-pot reaction drastically reduces labor hours, energy consumption, and solvent usage, leading to a leaner and more agile manufacturing operation. These efficiencies collectively contribute to a more resilient supply chain capable of meeting fluctuating market demands without the volatility associated with complex, multi-vendor precursor sourcing.
- Cost Reduction in Manufacturing: The transition to this one-pot methodology fundamentally alters the cost structure by removing the need for expensive heavy metal catalysts and the specialized equipment required for high-pressure hydrogenation. By consolidating reaction steps, manufacturers can achieve significant savings in utility costs and reactor occupancy time, effectively lowering the overhead per kilogram of produced intermediate. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting materials to byproducts, ensuring that raw material spend is converted into saleable product with maximum efficiency.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the primary raw materials, anisole and simple acyl chlorides, are commodity chemicals available from a broad global supplier base, reducing dependency on niche or monopolized precursors. The robustness of the reaction conditions means that production is less susceptible to minor variations in feedstock quality or environmental factors, ensuring consistent batch-to-batch availability. This reliability is critical for long-term supply agreements with major pharmaceutical companies that require guaranteed continuity of supply for their steroid drug portfolios.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing standard glass-lined or stainless steel reactors commonly found in fine chemical plants, which facilitates rapid technology transfer from pilot to commercial scale. The absence of toxic chromium waste streams simplifies the environmental permitting process and reduces the risk of production shutdowns due to regulatory non-compliance. This green chemistry advantage not only aligns with corporate sustainability goals but also future-proofs the supply chain against increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for integration into their existing supply chains, we have compiled answers to common questions regarding the process specifics and quality attributes. These insights are derived directly from the experimental data and technical disclosures within the patent literature, providing a transparent view of the method's capabilities and limitations. Understanding these details is essential for conducting accurate feasibility studies and risk assessments prior to scaling.
Q: How does this new synthesis method compare to traditional chromium-based routes?
A: Unlike traditional methods that utilize toxic chromium trioxide and dangerous hydrogenation steps with yields around 35%, this novel one-pot Friedel-Crafts approach eliminates heavy metal waste, simplifies the process, and achieves yields exceeding 90% with purity greater than 99%.
Q: What are the critical parameters for controlling isomer formation?
A: Controlling the reaction temperature between -10°C and 40°C during the acylation phase and strictly managing the dropwise addition rate of the acylating agent are essential to prevent ortho-substitution and ensure the formation of the desired para-intermediate.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses readily available raw materials like anisole and common solvents, avoids complex separation steps through a one-pot design, and utilizes standard equipment, making it highly adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Methoxy-1-Tetralone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced pharmaceutical intermediates requires more than just a patented recipe; it demands deep process engineering expertise and a commitment to quality excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 6-methoxy-1-tetralone meets the exacting standards required for steroid hormone synthesis.
We invite global partners to collaborate with us to leverage this cutting-edge synthesis technology for their specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, along with specific COA data and route feasibility assessments. Let us help you optimize your supply chain and reduce your time-to-market with a partner dedicated to innovation, reliability, and mutual growth in the fine chemical sector.
