Advanced Non-Halide Synthesis of Substituted Aromatic Amines for Commercial Scale Production
The chemical landscape for producing high-value aromatic amines is undergoing a significant transformation, driven by the urgent need for greener, more efficient synthetic pathways. Patent CN1046701C introduces a groundbreaking methodology for the preparation of substituted aromatic amines, specifically targeting critical intermediates like 4-aminodiphenylamine (4-ADPA). This technology represents a paradigm shift away from traditional halide-dependent synthesis, offering a robust alternative that aligns perfectly with modern environmental standards and cost-efficiency mandates. By leveraging a unique nucleophilic substitution mechanism involving azo-containing compounds, this process eliminates the reliance on corrosive halogenated starting materials, thereby solving long-standing issues related to equipment degradation and hazardous waste management. For R&D directors and procurement strategists alike, understanding the nuances of this patent is essential for securing a competitive edge in the global supply of pharmaceutical and agrochemical intermediates.
Furthermore, the versatility of this synthetic route extends beyond simple amines, enabling the production of alkylated derivatives and complex diamines through reductive alkylation steps. The core innovation lies in the precise control of reaction parameters, particularly the molar ratio of protic materials to bases, which dictates the selectivity and yield of the final product. As a reliable pharmaceutical intermediates supplier, recognizing the potential of such non-halide technologies allows us to offer clients not just a product, but a sustainable supply chain solution. The ability to synthesize these compounds under moderate conditions (10°C to 150°C) using commodity reagents ensures that production can be scaled rapidly without the bottlenecks associated with specialized, hazardous reagents. This report delves deep into the mechanistic advantages and commercial implications of adopting this advanced synthesis protocol.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of substituted aromatic amines, such as 4-ADPA, has relied heavily on nucleophilic aromatic substitution mechanisms involving halogenated precursors. The most common traditional route involves the reaction of aniline with parachloronitrobenzene in the presence of an acid acceptor. While chemically feasible, this method presents severe drawbacks that impact both operational expenditure and environmental compliance. The primary issue is the generation of stoichiometric amounts of halide salts as waste byproducts, which are highly corrosive to standard reactor materials and require expensive, energy-intensive disposal processes. Additionally, the starting material, parachloronitrobenzene, itself poses significant handling risks and supply chain volatility due to its classification as a hazardous substance. The corrosion caused by chloride ions often necessitates the use of exotic alloy reactors, driving up capital expenditure significantly for manufacturers attempting to scale this process.
Beyond the equipment costs, the purification of the final product from halide contaminants adds further complexity and cost to the manufacturing workflow. Residual halides can interfere with downstream reactions, particularly in sensitive pharmaceutical applications where impurity profiles are strictly regulated. Alternative methods, such as the decarboxylation of urethanes or the hydrogenation of nitroso compounds, have been explored but often suffer from unacceptable yields or require prohibitively expensive catalysts and conditions. These conventional limitations create a bottleneck for cost reduction in fine chemical manufacturing, forcing companies to absorb high waste treatment costs and face potential regulatory scrutiny. The inability to efficiently manage these halogenated waste streams has become a critical pain point for supply chain heads looking to optimize their production networks for long-term sustainability.
The Novel Approach
In stark contrast to these legacy methods, the process described in CN1046701C utilizes a non-halide pathway that fundamentally alters the reaction landscape. By employing an azo-containing compound (such as azobenzene) reacting with a nucleophilic compound (like aniline or its derivatives) in the presence of a base, the synthesis bypasses the need for halogenated leaving groups entirely. This approach not only eliminates the formation of corrosive chloride waste but also utilizes readily available, commodity-grade starting materials that are stable and easy to handle. The reaction proceeds through a confined reaction zone where the interaction between the nucleophile and the azo group is meticulously controlled by the presence of a specific amount of protic material. This novel mechanism allows for the direct formation of the aromatic amine backbone without the intermediate formation of nitro-compounds that require separate, energy-intensive reduction steps in traditional routes.
The strategic advantage of this method lies in its modularity and adaptability. The process can be tuned to produce various substituted derivatives by simply altering the R-groups on the azo or nucleophilic components, providing a flexible platform for synthesizing a wide range of high-purity aromatic amines. Furthermore, the subsequent reduction step to convert the azo-intermediate into the final amine can be performed using standard catalytic hydrogenation techniques, which are well-understood and easily scalable in existing infrastructure. This seamless integration into current manufacturing capabilities means that adopting this technology does not require a complete overhaul of plant facilities, thereby reducing lead time for high-purity aromatic amines to market. For procurement managers, this translates to a more resilient supply chain less susceptible to the regulatory crackdowns often associated with halogenated chemistry.
Mechanistic Insights into Non-Halide Nucleophilic Substitution
The core of this innovative synthesis lies in the intricate interplay between the nucleophilic compound, the azo-containing species, and the reaction medium. The mechanism initiates with the contact of a nucleophile—selected from anilines, aliphatic amines, or amides—with an azo compound represented by the general formula X-R1-N=N-R2-Y. This reaction occurs in a suitable solvent system, which can range from polar aprotic solvents like DMSO to fused amides, depending on the solubility requirements of the specific substrates. Crucially, the reaction is facilitated by a suitable base, such as alkali metal hydroxides or alkoxides, which activates the nucleophile for attack. However, the true genius of this patent is the recognition that the reaction environment must be strictly controlled regarding proton transfer materials. The molar ratio of protolyte (such as water or alcohol) to base is maintained between 0:1 and about 5:1. This precise balance is essential; too much protic material can quench the reactive species or shift the equilibrium unfavorably, while too little can hinder the solubility or mobility of the ionic intermediates.
Following the initial coupling, the resulting azo-intermediate undergoes a reduction step to yield the final substituted aromatic amine. This reduction can be achieved via catalytic hydrogenation using noble metal catalysts like palladium or platinum on carbon, or through chemical reduction using zinc powder. The flexibility in reduction methods allows manufacturers to choose the most cost-effective route based on their available infrastructure. When amides are used as the nucleophilic source, the process includes an additional hydrolysis or ammonolysis step to liberate the free amine, demonstrating the pathway's versatility in handling different nitrogen sources. This multi-step yet integrated approach ensures that impurities are minimized at each stage, resulting in a product with a superior impurity profile compared to halide-based alternatives. The ability to control the reaction at temperatures ranging from 10°C to 150°C further enhances safety and selectivity, preventing thermal runaway and side reactions that often plague exothermic nucleophilic substitutions.
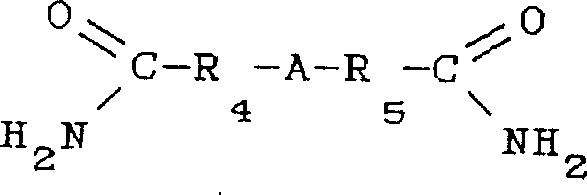
The scope of nucleophilic compounds applicable in this reaction is exceptionally broad, encompassing not just simple anilines but also complex amides and diamides as illustrated in the structural definitions within the patent. For instance, diamides with the structure shown above can serve as effective nucleophiles, expanding the utility of this process to the synthesis of polyfunctional amines. The presence of electron-withdrawing or electron-donating groups on the aromatic rings of the reactants can be tolerated, allowing for the fine-tuning of the electronic properties of the final amine product. This mechanistic robustness is vital for R&D teams aiming to develop new derivatives for specific pharmacological activities. By understanding that the reaction tolerates a wide variety of substituents (X and Y groups including halogens, nitro, amino, alkyl, etc.), chemists can design synthetic routes for complex molecules without being constrained by the incompatibility of functional groups often seen in harsher halide displacement reactions.
How to Synthesize 4-Aminodiphenylamine Efficiently
The synthesis of 4-aminodiphenylamine (4-ADPA) serves as the prime embodiment of this technology, demonstrating its practical viability for industrial applications. The process begins by contacting aniline with azobenzene in a solvent system such as aniline itself or DMSO, in the presence of a strong base like potassium tert-butoxide. The reaction mixture is heated to a moderate temperature, typically around 60°C to 80°C, and stirred under a controlled atmosphere. A critical operational parameter is the management of water content; in some embodiments, water is deliberately added or removed via distillation to maintain the optimal protolyte-to-base ratio, ensuring maximum conversion to the 4-(phenylazo)-pentanoic intermediate. Once the coupling is complete, the mixture is subjected to catalytic hydrogenation, often using platinum or palladium catalysts under hydrogen pressure, to reduce the azo linkage to the amine. Detailed standardized synthesis steps see the guide below.
- Contact a nucleophilic compound (such as aniline or amide derivatives) with an azo-containing compound in a suitable solvent system.
- React the mixture in the presence of a suitable base and a controlled amount of protic material at temperatures between 10°C and 150°C.
- Reduce the resulting reaction product under hydrogenation conditions to produce the final substituted aromatic amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this non-halide synthesis route offers transformative benefits that go beyond simple chemistry. The most immediate impact is seen in the drastic simplification of waste management protocols. By eliminating the generation of halogenated salts, manufacturers avoid the substantial costs associated with treating and disposing of hazardous chloride waste, which often constitutes a significant portion of the operational budget in traditional amine synthesis. This reduction in waste volume directly correlates to lower environmental compliance costs and reduces the risk of regulatory penalties. Furthermore, the absence of corrosive halides extends the lifespan of reactor vessels and piping, significantly lowering capital maintenance expenditures and reducing unplanned downtime due to equipment failure. These factors combine to create a leaner, more cost-efficient manufacturing model that enhances overall profitability.
- Cost Reduction in Manufacturing: The elimination of expensive halogenated starting materials like parachloronitrobenzene removes a major cost driver from the bill of materials. Additionally, the process avoids the need for specialized corrosion-resistant alloys in reactor construction, allowing for the use of standard stainless steel equipment which is far more economical. The streamlined purification process, resulting from fewer byproducts, reduces solvent consumption and energy usage during distillation and crystallization steps. Qualitative analysis suggests that the overall cost per kilogram of the final amine is significantly lowered due to these cumulative efficiencies, making the product more competitive in price-sensitive markets.
- Enhanced Supply Chain Reliability: The raw materials required for this process, primarily aniline and azobenzene derivatives, are commodity chemicals produced on a massive global scale. This ensures a stable and continuous supply, insulating manufacturers from the volatility often seen with specialized halogenated intermediates which may be subject to stricter transport regulations or limited production capacity. The robustness of the supply chain is further bolstered by the process's tolerance to varying grades of solvents and reagents, reducing the risk of production stoppages due to minor raw material specification deviations. This reliability is crucial for maintaining consistent delivery schedules to downstream pharmaceutical and agrochemical clients.
- Scalability and Environmental Compliance: The reaction conditions are mild and easily scalable from laboratory benchtop to multi-ton commercial production without significant re-engineering. The process operates at atmospheric or moderate pressures, reducing the safety risks associated with high-pressure reactors. From an environmental perspective, the 'green' nature of the chemistry—producing water and benign organic byproducts instead of toxic halides—aligns perfectly with increasingly stringent global environmental regulations. This forward-looking compliance ensures that the manufacturing facility remains operational and permitted for the long term, safeguarding the investment against future regulatory shifts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this non-halide synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this process into their existing production lines. The clarity provided here aims to bridge the gap between theoretical patent claims and practical industrial application.
Q: What are the primary advantages of this non-halide method over traditional chloronitrobenzene routes?
A: The primary advantage is the elimination of corrosive halogenated waste streams. Traditional methods using parachloronitrobenzene generate significant chloride waste requiring expensive disposal, whereas this azo-coupling route utilizes benign byproducts and avoids reactor corrosion entirely.
Q: How does the control of protic material impact the reaction yield?
A: Controlling the molar ratio of protic material to base (from 0:1 to about 5:1) is critical for selectivity. Excessive protic material can suppress the nucleophilic attack, while optimized levels ensure high conversion rates of the azo compound to the desired aromatic amine intermediate.
Q: Is this process scalable for industrial production of 4-ADPA?
A: Yes, the process operates at moderate temperatures (10°C to 150°C) and pressures suitable for standard stainless steel reactors. The use of commodity raw materials like aniline and azobenzene ensures supply continuity, making it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Aminodiphenylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced, non-halide synthesis routes is not just a chemical upgrade but a strategic business imperative. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN1046701C are fully realized in tangible product quality and volume. We are committed to delivering high-purity aromatic amines that meet the most stringent purity specifications required by the global pharmaceutical industry. Our rigorous QC labs employ state-of-the-art analytical techniques to verify impurity profiles, guaranteeing that every batch delivered is free from the halogenated contaminants that plague traditional supply chains. This dedication to quality and consistency makes us a trusted partner for companies seeking to secure their supply of critical intermediates.
We invite you to collaborate with us to explore how this innovative synthesis technology can optimize your specific manufacturing requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your current production metrics, highlighting exactly where efficiencies can be gained. We encourage you to reach out to request specific COA data and route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that is not only cost-effective and reliable but also aligned with the future of sustainable chemical manufacturing.
