Revolutionizing 1,5-Naphthalenediamine Production via Constructive Cyclization Technology
The global demand for high-purity aromatic diamines continues to surge, driven by their critical role in advanced polymer matrices and pharmaceutical scaffolding. Patent CN1275934C introduces a transformative methodology for the production of 1,5-naphthalenediamine, shifting away from traditional destructive nitration routes toward a constructive ring-building strategy. This intellectual property details a robust synthetic pathway originating from readily available commodity chemicals, specifically ortho-nitrotoluene and acrylic acid derivatives such as acrylonitrile. By fundamentally altering the bond construction sequence, this technology addresses the perennial challenge of isomeric purity that plagues conventional naphthalene functionalization. For R&D directors and procurement specialists alike, this represents a pivotal opportunity to secure a supply of 1,5-naphthalenediamine that is not only chemically superior but also economically optimized through simplified downstream processing. The following analysis dissects the technical merits and commercial implications of this patented process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 1,5-naphthalenediamine has relied heavily on the direct nitration of naphthalene followed by reduction, a process fraught with inherent selectivity issues. When naphthalene is subjected to nitration conditions, the electrophilic substitution occurs at multiple positions, inevitably generating a complex mixture containing the desired 1,5-isomer alongside significant quantities of the 1,8-isomer and other regioisomers. Separating these structural analogues requires energy-intensive fractional crystallization or chromatography, which drastically erodes overall yield and escalates production costs. Furthermore, alternative routes involving the sulfonation of naphthalene followed by amination often necessitate harsh corrosive conditions and generate substantial amounts of inorganic salt waste. These legacy methods impose a heavy burden on supply chain reliability, as the variability in isomer ratios from batch to batch can compromise the consistency of downstream polymerization or drug synthesis applications.
The Novel Approach
In stark contrast, the novel approach delineated in the patent constructs the naphthalene core de novo from a substituted benzene precursor, thereby bypassing the regioselectivity problems entirely. By initiating the synthesis with ortho-nitrotoluene, the position of the nitrogen functionality is pre-determined, ensuring that subsequent ring closure occurs exclusively at the desired positions to form the 1,5-substitution pattern. This constructive strategy utilizes a Michael addition followed by an intramolecular cyclization, effectively building the second ring onto the existing aromatic system. The result is a reaction profile that inherently favors the formation of the target molecule with minimal generation of positional isomers. This paradigm shift not only simplifies the purification train but also enhances the atom economy of the process, making it a far more attractive option for manufacturers seeking to reduce their environmental footprint while maximizing output efficiency.
Mechanistic Insights into Constructive Cyclization and Hydrogenation
The core of this synthetic innovation lies in the precise orchestration of four distinct chemical transformations, beginning with the base-catalyzed Michael addition of ortho-nitrotoluene to acrylonitrile. This initial step generates 4-(2-nitrophenyl)butyronitrile, a critical linear intermediate that sets the stage for ring closure. The subsequent cyclization is mediated by strong Lewis or Bronsted acids, such as sulfuric acid, polyphosphoric acid, or metal halides like aluminum chloride and antimony pentafluoride. These acidic conditions promote the intramolecular attack of the aromatic ring onto the nitrile or ester functionality, forming a tetrahydronaphthalene derivative. The patent highlights the versatility of this step, noting that the cyclization can proceed via imine or enamine intermediates depending on the specific substrate and conditions employed. This mechanistic flexibility allows process chemists to tune the reaction parameters to optimize for either kinetic speed or thermodynamic stability, ensuring high conversion rates even at commercial scales.
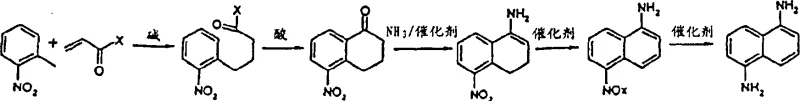
Following the formation of the partially saturated ring system, the process advances through a dehydrogenation step to establish full aromaticity, yielding 5-nitro-1-naphthylamine or its nitroso congeners. This aromatization is typically catalyzed by transition metals from Groups 8 to 10, such as palladium, platinum, or ruthenium, often supported on carbon or alumina. The final transformation involves the catalytic hydrogenation of the nitro group to the primary amine, completing the synthesis of 1,5-naphthalenediamine. Crucially, the patent discloses that the dehydrogenation and hydrogenation steps can potentially be telescoped or performed in the same reactor system using compatible catalysts, which significantly reduces equipment footprint and handling time. The ability to control the oxidation state of the intermediate through careful selection of catalysts and reaction atmospheres underscores the sophistication of this route, offering a level of process control that is unattainable with crude nitration methods.
How to Synthesize 1,5-Naphthalenediamine Efficiently
The implementation of this synthesis route requires careful attention to reaction conditions, particularly regarding the choice of base for the initial addition and the acid strength for cyclization. The patent provides extensive guidance on optimizing these parameters, suggesting the use of phase-transfer catalysts like tetrabutylammonium chloride to enhance the efficiency of the biphasic Michael addition. Detailed standard operating procedures for scaling this chemistry from gram to kilogram levels involve precise temperature control during the exothermic cyclization and rigorous safety protocols for handling high-pressure hydrogenation steps. For a comprehensive breakdown of the specific reagent stoichiometry, solvent choices, and workup procedures required to replicate this high-yielding pathway, please refer to the standardized synthesis guide below.
- Perform base-catalyzed Michael addition of o-nitrotoluene with acrylonitrile to form 4-(2-nitrophenyl)butyronitrile.
- Execute acid-catalyzed intramolecular cyclization using strong Lewis or Bronsted acids to generate nitro-tetralone derivatives.
- Conduct catalytic dehydrogenation followed by high-pressure hydrogenation to yield pure 1,5-naphthalenediamine.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this constructive synthesis route offers profound advantages for procurement managers and supply chain heads who are tasked with minimizing risk and controlling costs. The primary economic driver is the elimination of expensive and wasteful isomer separation processes. In traditional manufacturing, a significant portion of the raw material input is lost to unwanted isomers that must be separated and disposed of or sold at a discount. By virtually eliminating these byproducts at the source, the new process ensures that a much higher percentage of the input mass is converted into saleable product, leading to substantial cost reductions in 1,5-naphthalenediamine manufacturing. Additionally, the reliance on commodity feedstocks like ortho-nitrotoluene and acrylonitrile insulates the production cost from the volatility associated with specialized naphthalene derivatives, providing greater predictability in pricing models for long-term contracts.
- Cost Reduction in Manufacturing: The streamlined nature of this synthetic route directly translates to lower operational expenditures by removing the need for complex purification trains. Traditional methods often require multiple recrystallizations or distillation columns to achieve acceptable purity levels, which consume vast amounts of energy and solvents. In contrast, the high selectivity of the constructive cyclization means that the crude product is already of high purity, significantly reducing the load on downstream purification units. Furthermore, the potential for continuous processing, as noted in the patent, allows for smaller reactor volumes to achieve the same throughput, lowering capital expenditure requirements for new production facilities and reducing the overall cost of goods sold.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the bottlenecks inherent in separation-heavy processes. If a crystallization step fails or a distillation column fouls due to isomer buildup, the entire production line can halt. The robustness of this new method mitigates such risks by simplifying the unit operations involved. Because the process does not rely on the separation of close-boiling or similar-melting isomers, the likelihood of process upsets is markedly reduced. This stability ensures a more consistent delivery schedule for customers, allowing supply chain planners to maintain leaner inventory buffers without fear of stockouts. The use of widely available starting materials further secures the supply chain against raw material shortages that might affect niche naphthalene precursors.
- Scalability and Environmental Compliance: As regulatory pressures regarding waste disposal and emissions intensify, the environmental profile of a chemical process becomes a key factor in vendor selection. This synthesis route generates significantly less hazardous waste compared to sulfonation-based methods, which produce large quantities of acidic wastewater. The constructive approach is inherently cleaner, with fewer side reactions leading to tarry byproducts. Moreover, the scalability of the reaction steps, particularly the hydrogenation which can be run in continuous fixed-bed reactors, facilitates easy capacity expansion. Manufacturers can scale from pilot plant quantities to multi-ton annual production with minimal re-engineering, ensuring that supply can grow in lockstep with market demand while maintaining strict adherence to environmental compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 1,5-naphthalenediamine synthesized via this advanced pathway. These insights are derived directly from the experimental data and claims within the patent literature, providing a factual basis for evaluating the technology's viability. Understanding these nuances is essential for technical teams assessing the feasibility of integrating this material into their own formulations or synthesis pipelines. The answers reflect the specific capabilities and limitations of the described process.
Q: How does this process avoid isomer contamination compared to direct nitration?
A: Unlike direct nitration of naphthalene which produces complex mixtures of 1,5-, 1,8-, and other isomers requiring difficult separation, this constructive method builds the second ring regioselectively from o-nitrotoluene, inherently favoring the 1,5-substitution pattern without generating significant positional isomers.
Q: What catalysts are suitable for the final hydrogenation step?
A: The patent specifies that heterogeneous catalysts comprising Group 8-10 metals such as platinum, palladium, or ruthenium on carbon supports are highly effective. Raney nickel is also cited as a cost-effective alternative for large-scale reduction of the nitro-naphthylamine intermediates.
Q: Can this synthesis be adapted for continuous flow manufacturing?
A: Yes, the patent explicitly states that all reaction steps, including the initial Michael addition and the final hydrogenation, can be performed continuously in stirred-tank reactors or tubular reactors, offering significant advantages for process intensification and throughput scaling.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Naphthalenediamine Supplier
The technical elegance of this constructive synthesis route underscores the immense potential for producing 1,5-naphthalenediamine with unparalleled purity and efficiency. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory breakthroughs are successfully translated into industrial reality. Our engineering team is adept at managing the specific challenges of high-pressure hydrogenation and acid-catalyzed cyclizations, guaranteeing that every batch meets stringent purity specifications. With our rigorous QC labs and state-of-the-art manufacturing infrastructure, we are uniquely positioned to deliver high-purity 1,5-naphthalenediamine that satisfies the most demanding requirements of the global pharmaceutical and specialty polymer markets.
We invite forward-thinking organizations to collaborate with us to optimize their supply chains and reduce their dependency on legacy manufacturing methods. By leveraging our expertise in this specific synthetic technology, your organization can achieve significant competitive advantages through improved material quality and cost stability. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your volume requirements. Let us provide you with a Customized Cost-Saving Analysis that demonstrates exactly how switching to our advanced supply model can enhance your bottom line while securing your raw material future.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
