Advanced Catalytic Synthesis of 1,5-Naphthalenediamine for High-Purity Pharmaceutical Manufacturing
Advanced Catalytic Synthesis of 1,5-Naphthalenediamine for High-Purity Pharmaceutical Manufacturing
The global demand for high-purity aromatic diamines, particularly 1,5-naphthalenediamine, has surged due to their critical role in synthesizing advanced polyurethanes and pharmaceutical intermediates. Traditional manufacturing routes often struggle with isomer separation and harsh reaction conditions, creating bottlenecks for reliable supply chains. Patent CN1483017A introduces a transformative methodology that constructs the naphthalene core from basic benzene derivatives rather than modifying existing naphthalene structures. This approach fundamentally shifts the paradigm from separation-intensive processes to construction-intensive synthesis, offering a pathway to significantly higher purity and operational efficiency. By leveraging specific catalytic cycles involving alkylation, cyclization, and selective hydrogenation, this technology addresses the longstanding challenges of isomer contamination that have plagued the industry for decades.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 1,5-naphthalenediamine has relied heavily on the nitration of naphthalene or the reduction of dinitronaphthalenes. These conventional pathways suffer from a fundamental chemical limitation: the electrophilic substitution on the naphthalene ring rarely yields a single isomer. Instead, manufacturers are forced to deal with complex mixtures containing significant amounts of the 1,8-isomer and other positional variants. Separating these isomers requires energy-intensive distillation or crystallization steps, which drastically reduce overall yield and increase waste generation. Furthermore, methods involving sulfonation and alkaline fusion, as noted in older literature like DE-C1-3840618, necessitate extremely corrosive conditions that compromise equipment longevity and pose severe safety risks. The inherent lack of regioselectivity in these traditional routes translates directly into higher production costs and inconsistent quality, making them increasingly unsustainable for modern high-specification applications.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes o-nitrotoluene and acrylic acid derivatives as primary building blocks to construct the second ring of the naphthalene system with precise regiocontrol. Since the starting material already possesses the necessary substitution pattern, the resulting cyclization inherently favors the formation of the 1,5-substituted framework without generating significant quantities of unwanted isomers. This bottom-up synthetic strategy eliminates the need for difficult downstream purification steps associated with isomer removal. The process flow involves a base-catalyzed alkylation followed by an acid-catalyzed cyclization, allowing for modular optimization of each step. By avoiding the statistical distribution of isomers typical of electrophilic aromatic substitution on naphthalene, this route ensures a much cleaner crude product profile, thereby streamlining the entire manufacturing workflow and enhancing the economic viability of producing high-purity 1,5-naphthalenediamine.
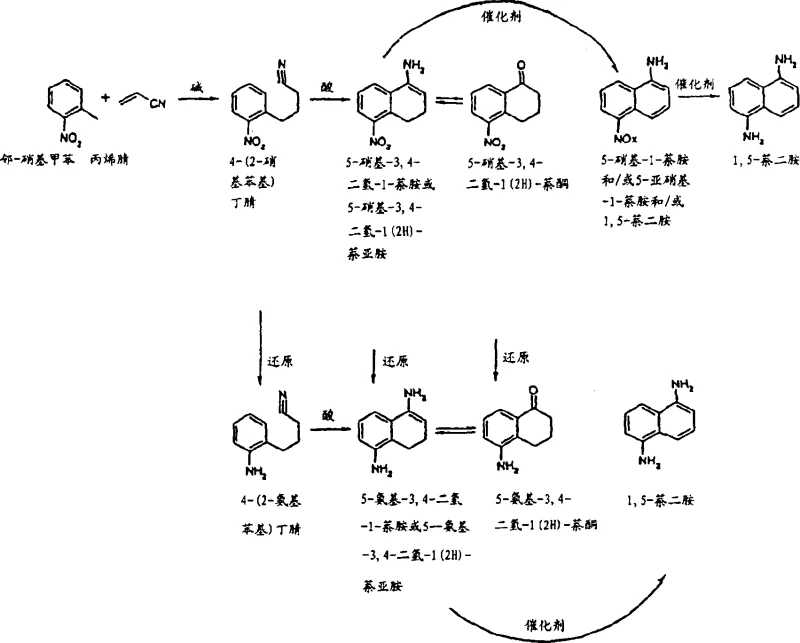
Mechanistic Insights into Base-Catalyzed Alkylation and Acid-Catalyzed Cyclization
The core of this innovative synthesis lies in the initial Michael addition of o-nitrotoluene to acrylonitrile, facilitated by phase transfer catalysts and strong bases such as sodium or potassium hydroxide. This step generates 4-(2-nitrophenyl)butyronitrile, a crucial linear precursor that sets the stage for ring closure. The subsequent cyclization is driven by strong Lewis or Bronsted acids, including sulfuric acid, polyphosphoric acid, or trifluoromethanesulfonic acid, which activate the nitrile or ester group for intramolecular electrophilic attack on the aromatic ring. This cyclization forms a tetrahydronaphthalene intermediate, specifically 5-nitro-3,4-dihydro-1(2H)-naphthalenone or its imine tautomer. The precision of this acid-catalyzed step is paramount, as it dictates the integrity of the newly formed ring system. Following cyclization, the process employs transition metal catalysts from Groups 8-10, such as palladium, platinum, or ruthenium, to effect dehydrogenation and aromatization. This dual-function catalytic capability allows for the conversion of the saturated ring into the aromatic naphthalene system while simultaneously or subsequently reducing the nitro group to an amine.
Impurity control is intrinsically built into the mechanistic design of this pathway. Because the carbon skeleton is assembled from defined precursors rather than modified via random substitution, the formation of structural isomers like 1,8-diaminonaphthalene is chemically precluded. The primary impurities arise from incomplete reactions or over-reduction, which are manageable through standard process controls. For instance, the patent highlights that the aromatization step can produce trace amounts of 5-nitroso-1-naphthylamine alongside the desired 5-nitro-1-naphthylamine, but these are readily converted to the final diamine during the subsequent hydrogenation stage. The use of supported metal catalysts further enhances selectivity, minimizing side reactions such as ring hydrogenation which could lead to tetralin derivatives. This robust mechanistic framework ensures that the final product meets stringent purity specifications required for sensitive applications in polymer chemistry and pharmaceutical synthesis, reducing the burden on quality control laboratories.
How to Synthesize 1,5-Naphthalenediamine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory scale to commercial production. The process begins with the alkylation of o-nitrotoluene, where temperature control between 20°C and 75°C is critical to maximize the yield of the butyronitrile intermediate while minimizing polymerization of the acrylonitrile. Following isolation, the cyclization step requires careful management of acid concentration and temperature, typically ranging from 60°C to 110°C, to ensure complete ring closure without degradation. The final stages involve catalytic hydrogenation, which can be performed in batch autoclaves or continuous trickle bed reactors depending on the desired throughput. Detailed standardized operating procedures regarding catalyst loading, pressure settings, and workup protocols are essential for reproducibility. For a comprehensive guide on the specific molar ratios, solvent choices, and safety parameters required for each unit operation, please refer to the structured synthesis guide below.
- React o-nitrotoluene with acrylonitrile under base catalysis to form 4-(2-nitrophenyl)butyronitrile.
- Cyclize the nitrile intermediate using strong acid catalysts to generate nitro-imine or nitro-ketone derivatives.
- Perform catalytic dehydrogenation followed by hydrogenation to yield the final 1,5-naphthalenediamine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers profound strategic advantages beyond mere technical feasibility. The reliance on o-nitrotoluene and acrylonitrile as feedstocks leverages widely available commodity chemicals, insulating the supply chain from the volatility associated with specialized naphthalene derivatives. This shift to basic chemical building blocks significantly reduces raw material costs and mitigates the risk of supply disruptions caused by niche manufacturer dependencies. Furthermore, the elimination of isomer separation steps translates directly into reduced processing time and lower energy consumption per kilogram of product. By simplifying the purification train, manufacturers can achieve faster batch turnover rates and higher overall equipment effectiveness. The ability to run key steps, such as hydrogenation and aromatization, in a continuous mode further enhances production flexibility, allowing suppliers to respond more agilely to fluctuating market demands without the need for massive inventory buffers.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the downstream processing requirements. Traditional methods incur substantial costs related to the separation of 1,5 and 1,8 isomers, often involving multiple crystallization cycles or complex distillation columns that consume significant utility resources. By inherently preventing the formation of these isomers, the new method removes these cost centers entirely. Additionally, the use of heterogeneous catalysts that can be recovered and reused minimizes the consumption of expensive precious metals like palladium and platinum. The reduction in waste generation also lowers disposal costs and environmental compliance burdens, contributing to a leaner and more cost-effective manufacturing model that improves margin potential for buyers.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, non-proprietary starting materials that are produced on a global scale. Unlike specialized intermediates that may have limited supplier bases, o-nitrotoluene and acrylic derivatives are manufactured by numerous chemical companies worldwide, ensuring competitive pricing and consistent availability. The process tolerance for various solvent systems and catalyst types further diversifies the sourcing options for critical inputs. This flexibility reduces the risk of single-source bottlenecks that can halt production lines. Moreover, the scalability of the continuous processing options means that capacity can be ramped up incrementally to match demand growth, ensuring a steady flow of high-purity 1,5-naphthalenediamine to downstream customers without the long lead times associated with building new batch facilities.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this methodology aligns well with modern green chemistry principles. The avoidance of corrosive sulfonation agents and the reduction in solvent usage for purification decrease the facility's environmental footprint. The process generates fewer hazardous byproducts, simplifying wastewater treatment and solid waste management. Scalability is enhanced by the compatibility of the reaction steps with standard industrial reactor designs, such as stirred tank reactors and tubular flow systems. This allows for seamless technology transfer from pilot plants to full-scale commercial production units. The ability to operate at moderate temperatures and pressures in many steps also reduces the capital expenditure required for high-specification pressure vessels, making it an attractive option for expanding production capacity in a cost-efficient and environmentally responsible manner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is crucial for R&D teams evaluating process feasibility and procurement officers assessing supplier capabilities. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance. These insights cover aspects ranging from catalyst selection to impurity profiles, providing a comprehensive overview of what stakeholders can expect when adopting this manufacturing route. For further technical clarification or specific data sheets, direct consultation with the technical team is recommended.
Q: How does this method avoid isomer contamination compared to traditional nitration?
A: Unlike direct nitration of naphthalene which produces mixed isomers like 1,8-dinitronaphthalene, this method constructs the naphthalene ring from o-nitrotoluene, ensuring the substituents are fixed in the 1,5-position inherently.
Q: What are the key catalysts used in the cyclization step?
A: Strong Lewis or Bronsted acids such as aluminum trichloride, sulfuric acid, polyphosphoric acid, or trifluoromethanesulfonic acid are employed to facilitate the intramolecular cyclization.
Q: Is the process suitable for large-scale continuous production?
A: Yes, the patent explicitly states that all reaction steps, including alkylation and hydrogenation, can be adapted for continuous operation in tubular reactors or stirred tank reactors, enhancing scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Naphthalenediamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of consistent quality and supply security in the pharmaceutical and fine chemical industries. Our expertise extends beyond simple distribution; we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that the transition from patent literature to commercial reality requires rigorous process engineering and quality assurance. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the absence of critical isomers like 1,8-diaminonaphthalene. We are committed to delivering 1,5-naphthalenediamine that meets the highest international standards, ensuring that your downstream synthesis of polyurethanes or active pharmaceutical ingredients proceeds without interruption or quality deviations.
We invite you to collaborate with us to optimize your supply chain for this vital intermediate. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage potential partners to contact us to request specific COA data and route feasibility assessments that demonstrate how our implementation of this advanced catalytic technology can benefit your operations. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable source of high-purity chemicals backed by deep technical knowledge and a commitment to long-term supply stability.
