Advanced Isomerization Technology for High-Purity Liquid Crystal Monomer Commercialization
Advanced Isomerization Technology for High-Purity Liquid Crystal Monomer Commercialization
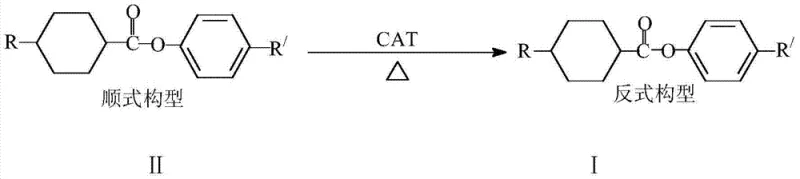
The landscape of electronic chemical manufacturing is constantly evolving, driven by the relentless demand for higher performance and lower costs in display technologies. Patent CN102633584B introduces a transformative approach to the synthesis of trans-cyclohexane carboxylate compounds, which serve as critical monomers in TN, STN, and TFT liquid crystal displays. This technology addresses a long-standing inefficiency in the industry where traditional synthesis routes generate substantial amounts of cis-isomer by-products that are often discarded. By leveraging a specific acid-catalyzed isomerization process, this method converts these low-value cis-configurations into high-value trans-configurations, thereby optimizing the entire production lifecycle. For R&D Directors and Supply Chain Heads, this represents a pivotal shift from linear consumption to circular efficiency, ensuring that every gram of raw material contributes to the final high-purity product required for advanced optical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of trans-cyclohexane carboxylate compounds has been plagued by inherent stereochemical challenges that limit overall process efficiency. The traditional synthetic pathway involves the esterification of cyclohexanecarboxylic acid derivatives, which inevitably results in a mixture of cis and trans isomers. Data indicates that in standard synthesis, the unwanted cis-isomer can account for approximately 30% of the reaction output. Since only the pure trans-structure possesses the necessary physical properties for liquid crystal display applications, this cis-fraction is typically separated and treated as waste mother liquor. This disposal not only represents a direct loss of expensive starting materials but also creates significant environmental burdens associated with chemical waste treatment. Furthermore, the low overall yield, often hovering around 50% for the target trans-product, necessitates larger reactor volumes and higher raw material procurement to meet production targets, driving up the cost of goods sold and complicating supply chain logistics for procurement managers seeking stability.
The Novel Approach
The innovative methodology outlined in the patent data fundamentally alters this economic equation by introducing a post-synthesis transformation step that recovers value from what was previously considered waste. Instead of discarding the cis-rich mother liquor, this novel approach subjects the cis-configuration cyclohexane carboxylate compounds to a controlled isomerization reaction. By utilizing p-toluenesulfonic acid as a catalyst within an organic solvent medium, the thermodynamic equilibrium is shifted to favor the more stable trans-configuration. This process allows manufacturers to recycle the cis-by-products back into the production stream, effectively boosting the total yield of the desired trans-isomer to greater than 69% from the recycled fraction. This shift from a discard-and-replace model to a recover-and-reuse model drastically reduces the volume of chemical waste requiring disposal and maximizes the utility of every kilogram of precursor purchased, offering a compelling value proposition for cost-conscious manufacturing operations.
Mechanistic Insights into Acid-Catalyzed Isomerization
At the core of this technological advancement lies a robust acid-catalyzed mechanism that facilitates the stereochemical inversion of the cyclohexane ring substituents. The reaction proceeds through a protonation event mediated by the p-toluenesulfonic acid catalyst, which temporarily increases the lability of the cyclohexane ring structure. Under the applied thermal conditions ranging from 35-200°C, with an optimal window of 60-100°C, the molecule gains sufficient energy to overcome the rotational barriers that lock the substituents in the cis-position. This allows the system to relax into the thermodynamically more stable trans-configuration, where steric hindrance between the substituents is minimized. The choice of p-toluenesulfonic acid is critical, as it provides the necessary acidity to drive the equilibrium without inducing unwanted side reactions such as ester hydrolysis or decomposition of the sensitive liquid crystal core structure, ensuring the integrity of the final electronic material.
Controlling the impurity profile is paramount for R&D Directors overseeing the quality of display materials, and this mechanism offers precise control over the final purity specifications. The reaction conditions are tuned to ensure that the isomerization proceeds to completion while minimizing the formation of degradation products. Following the reaction, standard workup procedures including water washing, extraction, and drying are employed to remove the catalyst and solvent residues. The patent data specifies that after purification, the resulting trans-cyclohexane carboxylate compounds achieve a purity level of ω > 99%. This high level of purity is essential for liquid crystal applications, where even trace impurities can affect the voltage holding ratio and response time of the display. The ability to consistently achieve this purity standard from recycled feedstocks demonstrates the robustness of the catalytic system and its suitability for producing high-performance electronic chemicals that meet stringent industry specifications.
How to Synthesize Trans-Cyclohexane Carboxylate Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize the conversion efficiency and product quality. The process begins with the selection of appropriate cis-4-R-cyclohexanecarboxylic acid-(4-R'-yl) ester compounds, which may be sourced from previous synthesis batches or purified waste streams. The reaction medium typically involves organic solvents such as toluene, xylene, n-heptane, or n-hexane, selected for their ability to dissolve the reactants and withstand the reflux temperatures. The ratio of solvent to raw material is maintained between 5-10ml per 1g to ensure adequate mixing and heat transfer. Once the catalyst is introduced, the system is heated to the target temperature range, and the reaction progress is monitored, often via gas chromatography, to determine the optimal endpoint before workup and purification steps are initiated to isolate the high-purity trans-product.
- Prepare the reaction system by mixing cis-4-R-cyclohexanecarboxylic acid-(4-R'-yl) ester compounds with an organic solvent such as toluene or xylene at a ratio of 5-10ml per 1g of raw material.
- Add p-toluenesulfonic acid as the catalyst with a mass ratio ranging from 1% to 20% relative to the raw material to initiate the isomerization process.
- Heat the reaction mixture to a temperature between 60-100°C and maintain reflux for 3-8 hours, followed by purification to achieve purity greater than 99%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this isomerization technology translates into tangible operational improvements and risk mitigation strategies. The primary advantage lies in the drastic reduction of raw material consumption per unit of finished product. By recovering value from the cis-isomer fraction, the effective yield of the manufacturing process is significantly enhanced without the need for additional precursor procurement. This efficiency gain acts as a buffer against raw material price volatility, allowing companies to maintain stable cost structures even when market prices for chemical intermediates fluctuate. Furthermore, the reduction in waste generation simplifies environmental compliance and lowers the costs associated with hazardous waste disposal, contributing to a more sustainable and economically viable supply chain operation that aligns with modern corporate responsibility goals.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the elimination of waste and the maximization of raw material utility. By converting the cis-isomer by-product, which traditionally represents a loss of approximately 30% of the reaction mass, into saleable trans-product, the effective cost per kilogram of the final liquid crystal monomer is substantially reduced. This process removes the need to purchase additional starting materials to compensate for low yields, thereby lowering the direct material costs. Additionally, the use of common, commercially available solvents and a simple organic acid catalyst avoids the expense of specialized or precious metal catalysts, further optimizing the operational expenditure profile for large-scale manufacturing facilities.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the availability of high-purity intermediates and the capacity to meet demand spikes. This method enhances reliability by creating an internal loop for material recovery, reducing dependence on external suppliers for every kilogram of product needed. The ability to process waste mother liquor into usable product means that production capacity is effectively increased without expanding physical infrastructure. This flexibility allows manufacturers to respond more agilely to market demands, ensuring that delivery schedules for critical display components are met consistently. The robustness of the reaction conditions also implies fewer production stoppages due to process sensitivity, leading to a more predictable and stable output of high-purity electronic chemicals.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces challenges related to heat management and waste handling, but this isomerization route is designed for industrial feasibility. The reaction operates at moderate temperatures and uses standard solvents, making it straightforward to scale from pilot batches to multi-ton production without complex engineering modifications. From an environmental perspective, the reduction in waste discharge is a significant compliance advantage. By minimizing the volume of cis-rich mother liquor that requires treatment or disposal, the facility reduces its environmental footprint and associated regulatory costs. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate image as a sustainable supplier of advanced electronic materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this isomerization technology in liquid crystal monomer production. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and technical feasibility for potential partners and stakeholders. Understanding these details is crucial for evaluating the integration of this process into existing manufacturing workflows and assessing its impact on overall product quality and cost efficiency.
Q: How does this isomerization process improve raw material utilization compared to traditional synthesis?
A: Traditional synthesis of trans-cyclohexane carboxylates often generates approximately 30% cis-isomer by-products which are typically discarded as waste. This patented method converts those cis-isomers back into the valuable trans-configuration, significantly increasing the overall yield from the initial raw materials and reducing waste discharge.
Q: What catalyst system is employed to ensure high purity in the final liquid crystal monomer?
A: The process utilizes p-toluenesulfonic acid as a robust organic catalyst. This system allows for precise control over the reaction conditions, enabling the production of trans-isomers with a purity exceeding 99% after standard purification steps like washing and distillation.
Q: Is this method scalable for industrial production of display materials?
A: Yes, the patent explicitly states that the method is simple to operate, easy to control, and suitable for industrial production. The use of common organic solvents like toluene and standard heating conditions facilitates easy scale-up for commercial manufacturing of electronic chemicals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-Cyclohexane Carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthesis routes in the competitive landscape of electronic materials. Our technical team has extensively analyzed the potential of isomerization technologies like CN102633584B to enhance the production of high-purity liquid crystal monomers. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs, guaranteeing that every batch of trans-cyclohexane carboxylate meets the exacting standards required for TN, STN, and TFT display applications.
We invite global partners to collaborate with us to optimize their supply chains and reduce manufacturing costs through advanced chemical engineering. By leveraging our expertise in process development, we can help you implement these efficient isomerization strategies to maximize yield and minimize waste. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term growth and stability in the electronic chemicals market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
