Advanced Isomerization Technology for High-Purity Trans-Cyclohexane Carboxylate Liquid Crystal Monomers
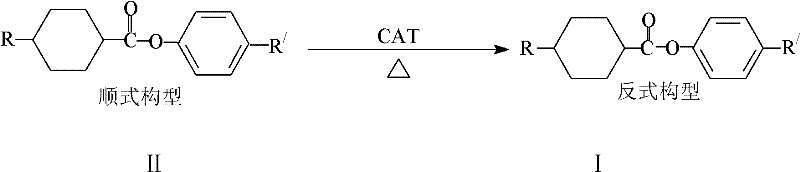
The landscape of electronic chemical manufacturing is constantly evolving, driven by the relentless demand for higher performance display technologies. At the forefront of this innovation is the synthesis of liquid crystal monomers, specifically trans-cyclohexane carboxylate compounds, which are critical components in TN, STN, and TFT liquid crystal displays. Patent CN102633584A introduces a groundbreaking methodology that addresses a longstanding inefficiency in this sector: the management of cis-isomer by-products. Traditionally, the synthesis of these vital monomers results in a mixture of cis and trans configurations, with the cis-form often deemed as waste. This patent delineates a robust chemical pathway to convert these cis-configuration compounds into the commercially valuable trans-configuration, thereby transforming a waste stream into a revenue-generating asset. This technological leap not only enhances the economic viability of liquid crystal production but also aligns with global sustainability goals by minimizing chemical waste discharge.
For R&D Directors and technical leaders, the implications of this patent extend beyond mere waste reduction; it represents a fundamental shift in process design. By integrating this isomerization step, manufacturers can drastically improve the atom economy of their synthesis routes. The ability to recycle up to 30% of the reaction output that was previously discarded allows for a more circular production model. This report analyzes the technical nuances of this conversion process, evaluating its feasibility for large-scale adoption and its potential to redefine cost structures in the display material supply chain. We will explore the mechanistic underpinnings, the operational parameters, and the strategic commercial advantages that this technology offers to forward-thinking chemical enterprises.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of trans-cyclohexane carboxylate compounds has been plagued by inherent stereochemical challenges. The conventional synthetic route involves the esterification of cyclohexanecarboxylic acid derivatives with phenolic compounds. However, this reaction is not stereoselective, inevitably generating a significant proportion of cis-isomers alongside the desired trans-products. In many standard processes, the cis-isomer content can reach approximately 30% of the total reaction mixture. Since only the pure trans-structure possesses the necessary physical properties for liquid crystal applications, such as appropriate birefringence and clearing points, the cis-fraction is traditionally separated and discarded. This separation process is not only costly, requiring extensive chromatography or crystallization steps, but it also results in a substantial loss of raw materials. The disposal of these cis-rich mother liquors creates an environmental burden, contributing to chemical waste volumes that modern regulations increasingly frown upon. Furthermore, the low overall yield, often hovering around 50% when accounting for the discarded cis-fraction, inflates the cost of goods sold and limits the scalability of production.
The Novel Approach
The methodology disclosed in patent CN102633584A offers a transformative solution to these inefficiencies by introducing a post-synthesis isomerization step. Instead of discarding the cis-isomer rich fractions, this novel approach subjects them to a catalytic transformation that converts the cis-configuration into the thermodynamically more stable trans-configuration. By utilizing p-toluenesulfonic acid as a catalyst in an organic solvent medium, the process effectively 'recycles' the unwanted isomers back into the product stream. This means that the effective yield of the process is no longer limited by the initial stereoselectivity of the esterification reaction. The ability to process waste mother liquors and convert them into high-purity trans-products fundamentally alters the mass balance of the manufacturing process. This approach not only recovers valuable chemical intermediates but also simplifies the purification workflow, as the need to strictly separate cis-isomers prior to conversion is eliminated. The result is a more streamlined, cost-effective, and environmentally responsible manufacturing protocol.

Mechanistic Insights into Acid-Catalyzed Isomerization
The core of this technological advancement lies in the acid-catalyzed stereochemical inversion of the cyclohexane ring. The reaction mechanism relies on the ability of the p-toluenesulfonic acid catalyst to facilitate the equilibration between the cis and trans isomers. Under thermal conditions, the cyclohexane ring can undergo conformational changes, and the presence of the acid catalyst lowers the energy barrier for this interconversion. The trans-isomer is thermodynamically favored due to reduced steric hindrance between the substituents on the cyclohexane ring compared to the cis-isomer. By maintaining the reaction system at elevated temperatures, typically between 35-200°C, the equilibrium is driven towards the trans-configuration. The choice of solvent plays a crucial role in this mechanism; solvents such as toluene, xylene, n-heptane, or n-hexane provide the necessary medium for the reactants to interact effectively with the catalyst while withstanding the required thermal conditions. The ratio of solvent to raw material, optimized between 5-10ml/1g, ensures adequate solubility and heat transfer, which are critical for uniform isomerization throughout the reaction bulk.
Controlling impurities during this isomerization is paramount for maintaining the high quality required for electronic materials. The patent specifies that after the isomerization reaction, the mixture undergoes a rigorous workup procedure involving water washing, extraction, drying, and solid-liquid separation. This sequence is designed to remove the acid catalyst and any polar by-products that may have formed during the thermal treatment. The subsequent purification step, often involving distillation or recrystallization, ensures that the final product achieves a purity level of greater than 99%. This high purity is essential because even trace impurities can significantly degrade the performance of liquid crystal displays, causing issues such as image sticking or reduced response times. The robustness of this purification protocol ensures that the recycled trans-isomers are indistinguishable in quality from those produced via direct synthesis, thereby validating the commercial viability of the recycling process for high-end display applications.
How to Synthesize Trans-Cyclohexane Carboxylate Efficiently
Implementing this isomerization technology requires precise control over reaction parameters to maximize yield and purity. The process begins with the selection of appropriate cis-rich feedstocks, which can be derived from the mother liquors of previous synthesis batches. These feedstocks are mixed with an organic solvent and the p-toluenesulfonic acid catalyst. The detailed standardized synthesis steps for this transformation are outlined in the guide below, providing a clear roadmap for technical teams to replicate the patent's success in a pilot or production setting. Adhering to the specified temperature ranges and reaction times is critical to ensuring complete conversion without degrading the sensitive ester functionality. This section serves as a technical bridge between the patent claims and practical laboratory or plant floor execution.
- Prepare the reaction mixture by combining cis-cyclohexane carboxylate raw materials with an organic solvent such as toluene or xylene.
- Add p-toluenesulfonic acid as the catalyst with a mass ratio ranging from 1% to 20% relative to the raw material.
- Heat the reaction system to a temperature between 35-200°C for 2-12 hours to facilitate the stereochemical transformation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this isomerization technology presents a compelling value proposition centered on cost efficiency and supply security. By converting waste streams into sellable products, the effective cost of raw materials is significantly reduced. This process eliminates the need to purchase additional starting materials to compensate for the low yields associated with traditional stereoselective synthesis. Furthermore, the reduction in waste disposal costs contributes directly to the bottom line, as the handling and treatment of hazardous chemical waste represent a substantial operational expense in fine chemical manufacturing. The simplicity of the reaction conditions, utilizing common solvents and a readily available acid catalyst, ensures that the technology can be implemented without significant capital expenditure on specialized equipment. This ease of integration minimizes downtime and accelerates the time-to-market for cost-optimized products.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the drastic improvement in raw material utilization. By recovering cis-isomers that were previously discarded, the overall yield of the manufacturing process is enhanced without increasing the input of expensive precursors. This leads to a substantial reduction in the cost per kilogram of the final liquid crystal monomer. Additionally, the elimination of complex separation steps required to remove cis-isomers prior to disposal simplifies the process flow, reducing labor and energy consumption. The qualitative shift from a linear 'make-and-waste' model to a circular 'make-and-recycle' model ensures long-term cost stability, protecting the supply chain from volatility in raw material pricing.
- Enhanced Supply Chain Reliability: Implementing this recycling protocol increases the resilience of the supply chain by maximizing output from existing feedstock inventories. In scenarios where raw material availability is constrained, the ability to squeeze more product from the same amount of starting material provides a critical buffer against supply disruptions. The use of common organic solvents and standard catalysts further enhances reliability, as these materials are widely available from multiple suppliers, reducing the risk of single-source bottlenecks. This robustness ensures consistent delivery schedules for downstream display manufacturers, fostering stronger long-term partnerships and trust within the value chain.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are inherently scalable, utilizing standard reactor types and thermal management systems found in most fine chemical plants. The process does not require exotic high-pressure or cryogenic conditions, making it suitable for large-scale commercial production. From an environmental perspective, the reduction in waste discharge aligns with increasingly stringent global environmental regulations. By minimizing the volume of chemical waste requiring treatment, manufacturers can reduce their environmental footprint and avoid potential regulatory penalties. This proactive approach to sustainability enhances the corporate image and meets the growing demand from end-users for responsibly sourced electronic materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the isomerization of cyclohexane carboxylates. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent data. They are designed to provide clarity on the feasibility, safety, and economic impact of adopting this technology. Understanding these details is crucial for stakeholders evaluating the integration of this process into their existing manufacturing portfolios.
Q: What is the primary advantage of this isomerization method over traditional synthesis?
A: The primary advantage is the ability to recycle cis-isomer by-products, which traditionally constitute waste, into valuable trans-isomers, significantly improving overall raw material utilization and reducing environmental discharge.
Q: What catalyst is used in this transformation process?
A: The process utilizes p-toluenesulfonic acid as a catalyst, which effectively promotes the stereochemical conversion under controlled thermal conditions in organic solvents.
Q: What purity levels can be achieved with this method?
A: Following purification steps such as washing, extraction, and distillation, the method achieves a product purity of greater than 99%, meeting stringent requirements for liquid crystal display applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-Cyclohexane Carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes in the production of high-performance electronic chemicals. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the isomerization process described in CN102633584A can be seamlessly transitioned from the lab to the plant. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs which employ advanced analytical techniques to verify every batch. Our capability to handle complex stereochemical transformations positions us as a strategic partner for companies seeking to optimize their liquid crystal monomer supply chain.
We invite you to collaborate with us to explore how this isomerization technology can enhance your production efficiency and reduce costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this advanced recycling methodology into your operations. Together, we can drive the next generation of sustainable and cost-effective display material manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
