Scalable Microbial Asymmetric Reduction for High-Purity (R)-Mandelic Acid Production
Introduction to Advanced Biocatalytic Synthesis of Chiral Intermediates
The global demand for optically pure pharmaceutical intermediates has necessitated a paradigm shift from traditional chemical synthesis to more sustainable biocatalytic processes. Patent CN100376681C introduces a groundbreaking method for the preparation of (R)-mandelic acid, a critical chiral building block used in the synthesis of antibiotics, antitumor drugs, and slimming agents. This technology leverages the specific strain Saccharomyces cerevisiae AS2.150 to perform an asymmetric reduction of benzoylformic acid, achieving optical purities exceeding 90% e.e. without the need for hazardous chemical resolving agents. By utilizing whole-cell catalysis, this approach not only simplifies the downstream purification process but also aligns with modern green chemistry principles, offering a robust solution for the reliable pharmaceutical intermediate supplier seeking to optimize their supply chain for high-value chiral compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of single-enantiomer mandelic acid relied heavily on the chemical resolution of racemic mixtures, a process fraught with significant economic and environmental inefficiencies. Traditional methods typically employ optically active amine compounds, such as alpha-methyl benzylamine or (-)-ephedrine, to form diastereomeric salts that can be separated. However, these chiral separation agents are notoriously expensive, often costing an arm and a leg relative to the substrate, and they frequently possess high toxicity profiles that complicate waste disposal and worker safety. Furthermore, the maximum theoretical yield of such resolution processes is inherently capped at 50%, meaning half of the synthesized material is wasted or requires energy-intensive racemization and recycling steps. The reliance on organic solvents and the generation of large volumes of saline wastewater further exacerbate the environmental footprint, making these legacy processes increasingly untenable for cost-conscious cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
In stark contrast, the novel microbial asymmetric reduction method described in the patent utilizes a highly selective biological catalyst to convert achiral benzoylformic acid directly into the desired (R)-enantiomer. This biocatalytic route bypasses the need for stoichiometric amounts of chiral amines, replacing them with a catalytic amount of whole yeast cells and inexpensive glucose as a co-substrate. The process operates under mild aqueous conditions, typically at temperatures between 25°C and 40°C and neutral pH, which drastically reduces energy consumption compared to the harsh conditions often required for chemical synthesis. By leveraging the inherent stereoselectivity of the enzymes within Saccharomyces cerevisiae AS2.150, the method achieves near-quantitative conversion with exceptional enantiomeric excess, effectively doubling the potential yield compared to traditional resolution while simultaneously eliminating the toxicity associated with heavy metal catalysts or toxic resolving agents.
Mechanistic Insights into Microbial Asymmetric Reduction
The core of this technology lies in the sophisticated enzymatic machinery of the Saccharomyces cerevisiae AS2.150 strain, which facilitates the stereospecific reduction of the ketone group in benzoylformic acid. The reaction is driven by oxidoreductases that require the cofactor NADH to transfer a hydride ion to the prochiral carbon, establishing the (R)-configuration with high fidelity. A critical innovation in this patent is the efficient in-situ regeneration of the NADH cofactor, which is consumed during the reduction and converted to NAD+. Instead of adding expensive external cofactors, the system utilizes glucose as a sacrificial co-substrate; the yeast's metabolic pathways oxidize glucose to gluconolactone, simultaneously regenerating NADH from NAD+ to sustain the catalytic cycle. This self-sustaining loop ensures that a small amount of biological catalyst can process large quantities of substrate, making the process economically viable for large-scale operations.
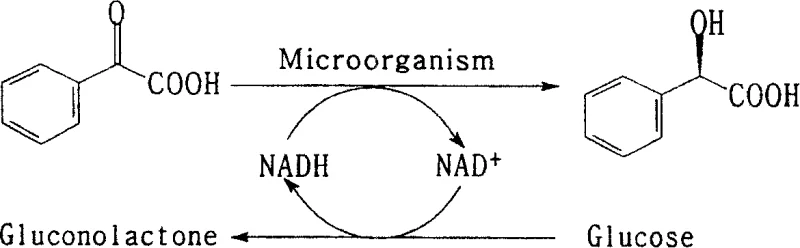
Beyond the primary reduction mechanism, the patent highlights the importance of optimizing the reaction microenvironment to maximize impurity control and catalyst stability. The presence of specific metal ions, such as Cobalt (Co), Aluminum (Al), or Calcium (Ca), has been shown to significantly enhance the transformation efficiency and optical purity of the final product. These ions likely act as stabilizers for the enzyme structure or facilitate substrate binding within the active site, ensuring that the competing formation of the unwanted (S)-enantiomer is minimized. The rigorous control of pH between 6.0 and 8.0 is also paramount, as deviations can lead to enzyme denaturation or non-specific background reactions that lower the enantiomeric excess. By fine-tuning these parameters, the process consistently delivers high-purity pharmaceutical intermediates with optical purity values reaching up to 99.91% e.e., meeting the stringent requirements of regulatory bodies for chiral drug substances.
How to Synthesize (R)-Mandelic Acid Efficiently
The synthesis of (R)-mandelic acid via this microbial route involves a streamlined sequence of cultivation, biotransformation, and isolation steps that are amenable to standard fermentation infrastructure. The process begins with the aerobic cultivation of the yeast strain in a nutrient-rich medium optimized for biomass and enzyme expression, followed by the harvesting of whole cells which serve as the biocatalyst. These cells are then suspended in a buffered solution containing the substrate benzoylformic acid and glucose, where the asymmetric reduction proceeds under controlled agitation and temperature. For a detailed breakdown of the specific media compositions, incubation times, and work-up procedures including extraction and crystallization, please refer to the standardized protocol below.
- Cultivate Saccharomyces cerevisiae AS2.150 in an optimized medium containing glucose, yeast extract, and specific metal salts at 25-35°C for 24-72 hours to generate active biomass.
- Harvest the whole cells via centrifugation and wash with physiological saline, then suspend them in a phosphate buffer system containing the substrate benzoylformic acid.
- Initiate the asymmetric reduction by adding glucose as a co-substrate for cofactor regeneration, maintaining pH 6.0-8.0 and 25-40°C until conversion is complete, followed by acidification and extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this microbial asymmetric reduction technology represents a strategic opportunity to de-risk the supply of critical chiral intermediates while driving down overall manufacturing costs. The shift from chemical resolution to biocatalysis eliminates the dependency on volatile markets for chiral amines and toxic solvents, replacing them with stable, commodity-grade raw materials like glucose and yeast extract. This transition not only simplifies the sourcing strategy but also mitigates regulatory risks associated with the handling and disposal of hazardous chemicals, ensuring a more resilient and compliant supply chain for long-term production campaigns.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the drastic reduction in raw material costs associated with chiral resolution. By eliminating the need for stoichiometric amounts of expensive resolving agents like alpha-methyl benzylamine, the variable cost per kilogram of the final product is significantly lowered. Furthermore, the ability to recycle the whole-cell biocatalyst for multiple batches enhances the effective turnover number of the enzyme, spreading the fixed cost of biomass production over a much larger volume of product. The high optical purity achieved directly from the reactor also minimizes the need for costly downstream purification steps such as recrystallization or chiral chromatography, leading to substantial overall process savings.
- Enhanced Supply Chain Reliability: The robustness of the Saccharomyces cerevisiae AS2.150 strain ensures a consistent and reliable supply of the active catalyst, reducing the risk of batch-to-batch variability that often plagues complex chemical syntheses. The whole cells demonstrate excellent stability, capable of being stored and reused over extended periods without significant loss of activity, which allows manufacturers to maintain strategic reserves of biocatalyst. This operational flexibility enables faster response times to market demand fluctuations and reduces the lead time for high-purity chiral intermediates, ensuring that downstream API production schedules are met without interruption.
- Scalability and Environmental Compliance: From a sustainability perspective, this biocatalytic process offers a clear path to scalable, green manufacturing that aligns with increasingly strict environmental regulations. The use of water as the primary solvent and the absence of heavy metal catalysts or toxic organic amines significantly reduce the burden on wastewater treatment facilities and lower the cost of waste disposal. The mild reaction conditions also translate to lower energy requirements for heating and cooling, contributing to a reduced carbon footprint. These factors make the technology highly attractive for commercial scale-up of complex pharmaceutical intermediates, facilitating easier regulatory approval and enhancing the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this microbial reduction technology. These insights are derived directly from the experimental data and optimization studies presented in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these nuances is essential for R&D teams planning technology transfer and for procurement teams assessing the long-term viability of the supply source.
Q: What is the primary advantage of using Saccharomyces cerevisiae AS2.150 over chemical resolution for (R)-mandelic acid?
A: The primary advantage is the elimination of expensive and toxic chiral resolving agents (such as alpha-methyl benzylamine) required in traditional chemical splitting methods. The microbial method utilizes inexpensive glucose for cofactor regeneration, significantly lowering raw material costs and environmental toxicity while achieving optical purities exceeding 90% e.e.
Q: How does the process ensure high optical purity and yield in industrial applications?
A: The process optimizes the reaction system by controlling pH between 6.0 and 8.0 and temperature between 25°C and 40°C. Furthermore, the addition of specific metal ions like Cobalt (Co) and the use of glucose as a co-substrate enhance the stereoselectivity of the enzyme within the whole cell, consistently delivering yields up to 100% and optical purity above 99% e.e. in optimized embodiments.
Q: Is the biocatalyst stable enough for repeated batch processing?
A: Yes, the whole cells of Saccharomyces cerevisiae AS2.150 demonstrate excellent operational stability. Patent data indicates that the cells can be recycled and reused for multiple consecutive batches (demonstrated up to 11 batches in specific examples) while maintaining high optical purity and consistent conversion rates, which is critical for continuous manufacturing efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-Mandelic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial fermenter is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC capabilities to guarantee that every batch of (R)-mandelic acid meets the exacting standards required by global pharmaceutical regulators.
We invite you to collaborate with us to leverage this advanced biocatalytic technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized microbial reduction process can deliver superior value, purity, and reliability for your critical manufacturing projects.
