Advanced Microbial Resolution of (R)-Mandelic Acid for Scalable Pharmaceutical Manufacturing
Advanced Microbial Resolution of (R)-Mandelic Acid for Scalable Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient, environmentally benign, and cost-effective methods for producing high-value chiral intermediates. A pivotal development in this domain is documented in patent CN100385007C, which discloses a novel method for the preparation of (R)-mandelic acid through asymmetric separation utilizing microorganisms. This technology represents a significant leap forward from traditional chemical synthesis, leveraging the inherent stereoselectivity of biological systems to achieve superior optical purity. Specifically, the patent details the screening and application of the strain Brevibacterium flavum AS 1.818, which acts as a highly efficient biocatalyst. By employing whole-cell catalysis, this method transforms racemic mandelic acid into the desired (R)-enantiomer with an optical purity consistently exceeding 90% e.e. This breakthrough not only addresses the critical demand for high-purity chiral building blocks but also aligns with global trends towards greener manufacturing processes.
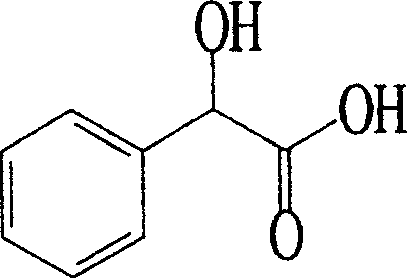
(R)-Mandelic acid, chemically known as α-hydroxyphenylacetic acid, serves as a vital chiral module in the synthesis of a diverse array of bioactive compounds. Its applications span the production of semi-synthetic antibiotics such as penicillins and cephalosporins, as well as anti-tumor agents, antidepressants, and weight-loss pharmaceuticals. Furthermore, it acts as a crucial chiral resolving agent for other racemic compounds. The structure shown above highlights the chiral center that dictates its biological activity, making the separation of enantiomers a non-negotiable requirement for drug safety and efficacy. The microbial resolution technique described in the patent offers a robust pathway to access this valuable intermediate, ensuring that supply chains for reliable pharmaceutical intermediate suppliers can meet the stringent quality standards required by regulatory bodies worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of optically pure mandelic acid has relied heavily on chemical resolution methods, which present substantial drawbacks in terms of cost, safety, and environmental impact. The traditional approach typically involves the use of expensive and toxic optically active amine compounds, such as α-methylbenzylamine, (-)-ephedrine, or cinchonine, to form diastereomeric salts. These salts must then be separated through fractional crystallization, a process that is inherently inefficient, often resulting in low yields and requiring multiple recrystallization steps to achieve acceptable purity. Moreover, the chiral resolving agents used are not only costly but also pose significant toxicity risks, leading to severe environmental pollution during disposal. Alternative methods like preparative chiral chromatography, while effective for analytical purposes, are economically unviable for large-scale production due to the high cost of chiral stationary phases and the limited loading capacity of the columns. Consequently, the chemical industry has long suffered from high energy consumption, complex production workflows, and the generation of hazardous waste streams associated with these legacy technologies.
The Novel Approach
In stark contrast to these cumbersome chemical processes, the microbial resolution method introduced in patent CN100385007C offers a streamlined and sustainable alternative. By utilizing the whole cells of Brevibacterium flavum AS 1.818, the process capitalizes on the natural enzymatic machinery of the microorganism to selectively recognize and metabolize the unwanted (S)-enantiomer of mandelic acid. This biological specificity eliminates the need for toxic chiral amines and complex salt formation steps, drastically simplifying the workflow. The reaction proceeds under mild aqueous conditions, typically at temperatures between 25°C and 40°C and neutral pH levels, which significantly reduces energy requirements compared to the harsh conditions often needed for chemical catalysis. Furthermore, the biocatalytic system demonstrates high regioselectivity and chemoselectivity, minimizing the formation of by-products and ensuring a cleaner reaction profile. This shift from chemical to biological catalysis not only enhances the optical purity of the final product but also fundamentally alters the economic and environmental footprint of cost reduction in chiral intermediate manufacturing.
Mechanistic Insights into Microbial Asymmetric Resolution
The core of this technology lies in the unique metabolic capabilities of the screened strain, Brevibacterium flavum AS 1.818. Mechanistically, the process functions as a dynamic kinetic resolution where the microbial cells act as a sink for the (S)-enantiomer. Upon exposure to racemic mandelic acid, the enzymes within the bacterial cells—likely specific dehydrogenases or oxidases induced during the culture phase—preferentially bind to and degrade the (S)-mandelic acid. This selective degradation leaves the (R)-mandelic acid largely untouched in the reaction medium. The patent data indicates that the (R)-enantiomer remains stable until the (S)-enantiomer is completely consumed, preventing the erosion of optical purity in the later stages of the reaction. This mechanism is far superior to simple equilibrium-based resolutions because it drives the reaction towards the accumulation of the desired product rather than a 50:50 mixture. The optimization of the culture medium, including the precise balance of carbon sources like glucose and nitrogen sources like peptone, ensures that the cells express the necessary enzymatic activity at high levels, thereby maximizing the conversion efficiency and the final enantiomeric excess.
Furthermore, the patent elucidates the critical role of reaction conditions and additives in modulating this enzymatic activity. The presence of specific metal ions in the transformation system was found to be a key factor in enhancing catalytic performance. Ions such as Magnesium (Mg), Manganese (Mn), Iron (Fe), and notably Copper (Cu) were observed to significantly boost the conversion rate, likely by acting as cofactors for the active enzymes or stabilizing the protein structure. Conversely, the absence of these ions or the presence of inhibitory substances could stall the reaction. The process also benefits from the addition of structural analogues like glycolic acid or lactic acid during the cultivation phase, which act as inducers to upregulate the expression of the relevant metabolic pathways. This level of mechanistic control allows for fine-tuning the process to achieve optical purities ranging from 90% to over 96% e.e., providing a robust platform for the commercial scale-up of complex pharmaceutical intermediates where consistency is paramount.
How to Synthesize (R)-Mandelic Acid Efficiently
Implementing this microbial resolution strategy requires a disciplined approach to fermentation and biocatalysis to ensure reproducible high-quality results. The process begins with the rigorous cultivation of the Brevibacterium flavum strain under optimized conditions to generate a highly active biomass. Following harvest, the cells are subjected to a controlled transformation phase where the racemic substrate is introduced. The detailed protocol involves precise control over pH, temperature, and agitation to maintain cell viability and enzymatic activity throughout the reaction duration. For research and development teams looking to replicate or adapt this methodology, understanding the interplay between cell density, substrate concentration, and reaction time is essential. The following guide outlines the standardized operational framework derived from the patent examples, serving as a foundational reference for process development.
- Cultivate Brevibacterium flavum AS 1.818 in an optimized medium containing glucose, peptone, and yeast extract at 30°C for 48 hours to generate active biomass.
- Harvest the cells via centrifugation, wash with physiological saline, and resuspend in a phosphate buffer system adjusted to pH 7.0.
- Add racemic mandelic acid substrate to the cell suspension and maintain at 25-40°C with shaking for 24-72 hours to allow selective degradation of the (S)-enantiomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this microbial resolution technology offers compelling strategic advantages that extend beyond mere technical feasibility. The primary value proposition lies in the drastic simplification of the supply chain and the reduction of dependency on volatile raw material markets. By eliminating the need for expensive, imported chiral resolving agents like alpha-methylbenzylamine, manufacturers can decouple their production costs from the fluctuations of the specialty chemicals market. Instead, the process relies on ubiquitous and inexpensive fermentation substrates such as glucose, peptone, and inorganic salts, which are readily available globally. This shift fundamentally alters the cost structure of production, moving it from a high-cost, low-yield chemical model to a lower-cost, high-efficiency biological model. Additionally, the reduction in hazardous waste generation translates directly into lower disposal costs and reduced regulatory compliance burdens, further enhancing the overall economic viability of the operation.
- Cost Reduction in Manufacturing: The elimination of stoichiometric amounts of expensive chiral amines represents a direct and significant saving in raw material costs. In traditional chemical resolution, up to half of the starting material is wasted as the unwanted enantiomer, and the resolving agent often requires energy-intensive recovery processes or is discarded entirely. In contrast, the microbial method utilizes catalytic amounts of biomass to drive the reaction, and the unwanted enantiomer is biodegraded into harmless by-products like water and carbon dioxide. This atom-economic approach, combined with the use of cheap fermentation media, results in a substantially lower cost of goods sold (COGS). Furthermore, the simplified downstream processing, which avoids complex crystallization sequences, reduces utility consumption and labor hours, contributing to a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: Relying on biological fermentation provides a more resilient supply chain compared to multi-step chemical synthesis which may depend on scarce reagents. The strain Brevibacterium flavum is robust and can be maintained and propagated indefinitely, ensuring a consistent source of catalyst. The raw materials required for the medium are commodity chemicals with stable supply lines, reducing the risk of production stoppages due to material shortages. Moreover, the scalability of fermentation technology is well-established, allowing for rapid expansion of production capacity to meet surging demand without the need for constructing entirely new types of chemical plants. This reliability is crucial for maintaining reducing lead time for high-purity pharmaceutical intermediates and ensuring uninterrupted supply to downstream drug manufacturers.
- Scalability and Environmental Compliance: The aqueous nature of the biocatalytic reaction and the biodegradability of the waste streams make this process inherently environmentally friendly. Unlike chemical resolution which generates toxic organic waste requiring specialized treatment, the effluent from this microbial process is largely organic and biodegradable, simplifying wastewater treatment and reducing environmental fees. This alignment with green chemistry principles facilitates easier permitting and compliance with increasingly stringent environmental regulations. From a scalability perspective, the transition from shake flasks to industrial fermenters is a standard engineering practice, meaning the technology can be scaled from pilot batches to hundreds of tons annually with predictable results, supporting long-term growth strategies for reliable pharmaceutical intermediate suppliers.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of this technology for their specific applications, we have compiled a set of frequently asked questions based on the detailed findings of patent CN100385007C. These queries address common concerns regarding process efficiency, scalability, and product quality, providing clarity on how this microbial resolution method compares to existing industrial standards. Understanding these technical nuances is essential for integrating this innovative approach into your current manufacturing portfolio and leveraging its full potential for cost and quality optimization.
Q: What is the primary advantage of using Brevibacterium flavum for mandelic acid resolution?
A: The primary advantage is the high stereoselectivity and mild reaction conditions. Unlike traditional chemical resolution which requires toxic and expensive chiral amines, this microbial method utilizes whole cells to preferentially degrade the unwanted (S)-enantiomer, leaving the desired (R)-mandelic acid with an optical purity exceeding 90% e.e. without hazardous waste generation.
Q: How does the addition of metal ions affect the conversion efficiency?
A: According to the patent data, the addition of specific metal ions such as Copper (Cu), Magnesium (Mg), Manganese (Mn), and Iron (Fe) significantly enhances the catalytic activity of the bacterial cells. For instance, the presence of Copper ions was observed to increase conversion activity substantially compared to controls without metal additives, optimizing the overall process efficiency.
Q: Is this biological method suitable for large-scale industrial production?
A: Yes, the process is highly amenable to scale-up. The use of standard fermentation techniques, readily available carbon sources like glucose, and robust whole-cell catalysts allows for transition from laboratory shake flasks to large industrial fermenters. The elimination of complex chromatographic separation steps further simplifies the downstream processing required for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-Mandelic Acid Supplier
The technological advancements detailed in patent CN100385007C underscore the immense potential of biocatalysis in modernizing the production of critical chiral intermediates. At NINGBO INNO PHARMCHEM, we recognize the strategic value of such innovations and have positioned ourselves at the forefront of adopting these advanced manufacturing techniques. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of high-quality materials. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise enantiomeric excess measurements, guaranteeing that every batch of (R)-mandelic acid meets the exacting standards required for pharmaceutical synthesis.
We invite global partners to collaborate with us to leverage this cutting-edge microbial resolution technology for their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your sourcing strategy, reduce your environmental footprint, and secure a competitive advantage in the global market through the adoption of superior, sustainable manufacturing processes.
