Advanced Ruthenium Complex Synthesis: Technical Insights for Commercial Scale-up of complex Pharmaceutical Intermediates
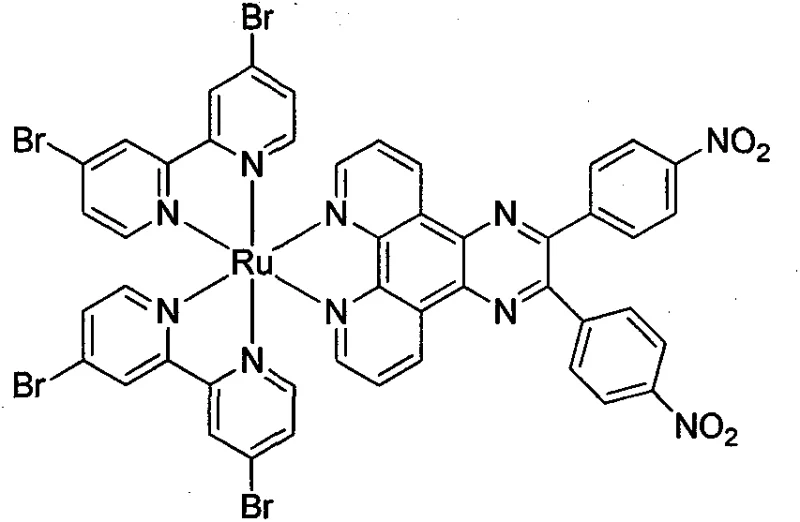
The pharmaceutical industry is constantly seeking novel metal complexes that can overcome the limitations of traditional platinum-based chemotherapeutics, and patent CN105884833B presents a significant breakthrough in this domain by detailing the preparation method of a novel ruthenium complex containing 4,4'-dibromo-2,2'-bipyridine. This specific Ru(II) complex is engineered to exhibit high DNA affinity, leveraging the planar aromatic heterocycle structure to facilitate effective intercalation into DNA base pairs, which is crucial for inhibiting cancer cell replication. The synthesis route described in the patent is characterized by mild reaction conditions and a straightforward procedural flow, which suggests a high degree of feasibility for industrial adaptation and scale-up. Furthermore, the incorporation of strong electron-withdrawing groups, specifically nitro and bromine substituents, significantly enhances the binding capacity of the complex to DNA, thereby potentially improving its antitumor efficacy compared to existing standards. For R&D directors and procurement specialists, understanding the structural nuances and synthetic accessibility of this compound is vital for evaluating its potential as a high-purity pharmaceutical intermediate. The detailed methodology provided offers a clear pathway for producing this advanced material, ensuring that supply chain stakeholders can assess the reliability of sourcing such specialized chemical entities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional anticancer agents like Cisplatin, while effective, are plagued by significant drawbacks including severe toxic side effects, the development of drug resistance in cancer cells, and poor water solubility which complicates formulation and delivery. These limitations often necessitate complex purification processes to remove toxic byproducts, thereby increasing the overall cost reduction in pharmaceutical intermediates manufacturing and extending the lead time for high-purity pharmaceutical intermediates. Moreover, the synthesis of many metal-based drugs often involves harsh conditions that can degrade sensitive functional groups, leading to lower yields and a broader impurity profile that requires extensive downstream processing. The inability of conventional platinum complexes to specifically recognize gene variation sites in cancer cells further limits their therapeutic index, often resulting in damage to healthy tissues. Consequently, there is a pressing need for alternative metal complexes that offer a better safety profile and more targeted mechanism of action without compromising on synthetic efficiency. The reliance on expensive and difficult-to-remove heavy metals in older protocols also poses environmental and supply chain challenges that modern green chemistry principles aim to resolve.
The Novel Approach
The novel approach outlined in patent CN105884833B addresses these challenges by utilizing a ruthenium polypyridine complex that boasts low toxicity, mature synthesis technology, and easy absorption by cancer tissues with quick excretion rates. This method allows for the adjustment of affinity, electron transfer direction, and substitution reaction rates by modifying the ligand, enabling photoelectric activation and targeted therapy. The specific use of 4,4'-dibromo-2,2'-bipyridine and pyrazino[1,10]phenanthroline ligands creates a structure with three planar aromatic heterocycles that have high affinity with DNA, surpassing the binding capabilities of many conventional agents. The synthesis steps are designed to be simple and easy to implement, with stable structures of intermediate reaction products that minimize the risk of decomposition during production. This stability is crucial for maintaining consistent quality across batches, a key concern for supply chain heads managing the commercial scale-up of complex pharmaceutical intermediates. By avoiding the severe side effects associated with cisplatin, this new complex represents a paradigm shift towards safer and more effective antitumor medications.
Mechanistic Insights into Ru(II) Complex DNA Intercalation
The mechanistic action of this Ru(II) complex is rooted in its ability to interact with DNA through electrostatic binding, groove binding, and most importantly, insertion binding, which is considered the optimal mode for damaging base pairs directly. The insertion binding mode allows the complex to insert into the base pairs of the DNA, influencing the DNA replication of cancer cells and effectively preventing them from copying. If the metal complex can recognize the gene variation site of a cancer cell, it inserts into the mismatched base pair to prevent replication, serving as a potent anticancer medicine to radically cure hereditary diseases. The presence of the planar aromatic heterocycle in the Ru(II) complex facilitates this intercalation process, ensuring a strong and stable interaction with the genetic material. This mechanism is superior to simple electrostatic interactions which may be reversible and less effective in halting cell proliferation. Understanding this mechanism is critical for R&D teams evaluating the biological potential of this compound for further drug development.
In the selection of the inserted ligand, the nitro and the bromine groups play a pivotal role due to their strong electron-withdrawing effects, which enhance the binding capacity of the complex to DNA. These substituents modify the electronic density of the aromatic system, making it more electrophilic and thus more attractive to the electron-rich DNA bases. This electronic tuning is a sophisticated strategy to maximize therapeutic efficacy without altering the core metal center, which remains stable throughout the biological interaction. The synthesis of the ligand involves a condensation reaction between 4,4'-dinitrobenzil and 5,6-diamine-1,10-phenanthroline, creating a rigid planar structure ideal for intercalation. The final coordination with the ruthenium center locks this geometry in place, ensuring that the electron-withdrawing groups are positioned optimally for DNA interaction. This level of molecular engineering demonstrates the high technical sophistication required to produce high-purity pharmaceutical intermediates with specific biological activities.
How to Synthesize Novel Ruthenium Complex Efficiently
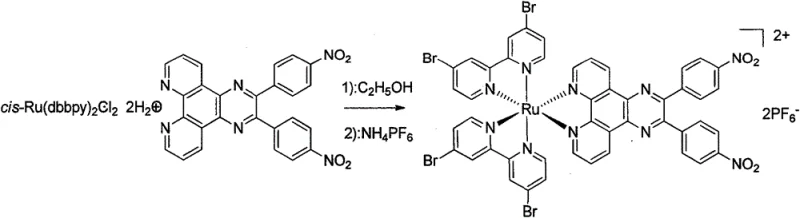
The synthesis of this novel ruthenium complex is a multi-step process that requires precise control over reaction conditions to ensure high yields and purity, starting from readily available starting materials like 4-nitrobenzaldehyde. The detailed standardized synthesis steps involve the initial formation of 4-nitrobenzoin, followed by oxidation to 4,4'-dinitrobenzil, and subsequent condensation to form the phenanthroline derivative ligand. The final stages involve the coordination of this ligand with a ruthenium precursor to form the target Ru(II) complex, which is then purified through column chromatography and recrystallization. Each step has been optimized to maximize yield, with reported yields ranging from 61.7% to 83.6%, indicating a robust and efficient synthetic route. The use of common solvents like ethanol, ethyl acetate, and dimethylformamide further simplifies the process, making it accessible for standard chemical manufacturing facilities. For a comprehensive guide on the specific operational parameters and safety precautions, please refer to the standardized protocol provided below.
- Synthesize 4-nitrobenzoin from 4-nitrobenzaldehyde and vitamin B1.
- Oxidize 4-nitrobenzoin to 4,4'-dinitrobenzil using NBS.
- Condense 4,4'-dinitrobenzil with 5,6-diamine-1,10-phenanthroline to form the ligand.
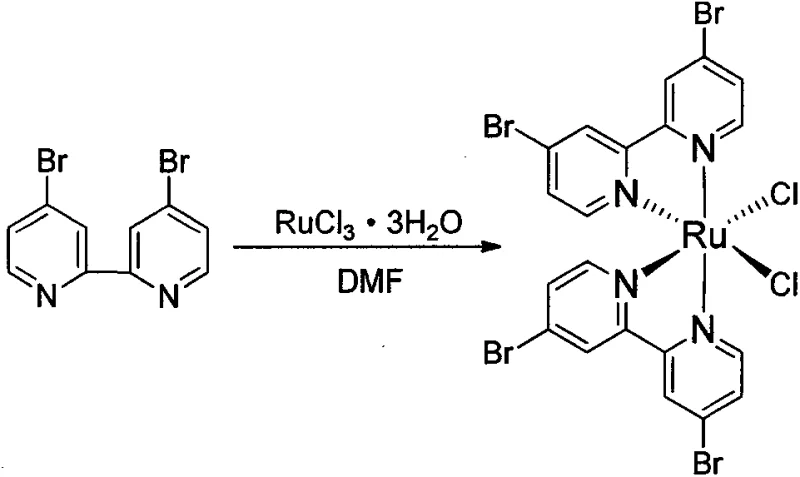
- Prepare cis-Ru(dbbpy)2Cl2 from ruthenium trichloride and 4,4'-dibromo-2,2'-bipyridine.
- React the Ru precursor with the ligand to form the final Ru(II) complex.
Commercial Advantages for Procurement and Supply Chain Teams
The commercial viability of this synthesis route is underpinned by its ability to address traditional supply chain and cost pain points through simplified processing and high-yield reactions. The use of stable intermediates reduces the risk of batch failure, ensuring a consistent supply of material which is critical for maintaining production schedules in a pharmaceutical setting. Furthermore, the mild reaction conditions reduce the energy consumption and equipment stress associated with high-temperature or high-pressure processes, contributing to overall operational efficiency. The purification methods described, such as recrystallization and column chromatography, are standard industry practices that do not require exotic or prohibitively expensive equipment, facilitating easier technology transfer. This accessibility makes the process attractive for contract development and manufacturing organizations looking to expand their portfolio of specialty chemical offerings. By streamlining the synthesis, the overall production timeline can be optimized, reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The synthesis route eliminates the need for extremely harsh conditions that often require specialized corrosion-resistant equipment, thereby lowering capital expenditure and maintenance costs. The high yields observed in each step, particularly the 83.6% yield in the oxidation step, minimize raw material waste and maximize the output per batch, leading to substantial cost savings. Additionally, the use of vitamin B1 as a catalyst in the initial step is a cost-effective alternative to more expensive transition metal catalysts, further driving down the cost of goods sold. The ability to recrystallize products from common solvents like methanol and ethanol reduces solvent recovery costs and simplifies waste management protocols. These factors collectively contribute to a more economical manufacturing process without compromising on the quality of the final product.
- Enhanced Supply Chain Reliability: The starting materials, such as 4-nitrobenzaldehyde and ruthenium trichloride, are commercially available from multiple global suppliers, reducing the risk of supply chain disruptions due to single-source dependency. The stability of the intermediate products allows for potential stockpiling or semi-finished goods inventory, providing flexibility in production planning and demand forecasting. The robust nature of the reaction conditions means that the process is less sensitive to minor fluctuations in temperature or pressure, ensuring consistent output even in varying manufacturing environments. This reliability is crucial for procurement managers who need to guarantee continuous supply to downstream drug formulation partners. The established synthetic pathway also allows for easier scaling from laboratory to pilot to commercial production, ensuring that supply can grow in tandem with market demand.
- Scalability and Environmental Compliance: The process utilizes solvents and reagents that are well-understood in terms of environmental impact, facilitating easier compliance with increasingly stringent global environmental regulations. The absence of highly toxic byproducts, common in some heavy metal syntheses, simplifies waste treatment and disposal, reducing the environmental footprint of the manufacturing facility. The high atom economy of the condensation reactions ensures that a significant portion of the reactant mass is incorporated into the final product, minimizing waste generation. Scalability is supported by the use of standard unit operations like reflux, filtration, and rotary evaporation, which are easily replicated in larger reactors without significant process redesign. This alignment with green chemistry principles not only meets regulatory requirements but also enhances the corporate social responsibility profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions and answers are derived directly from the technical details and beneficial effects described in patent CN105884833B, addressing common inquiries regarding the synthesis and application of this ruthenium complex. These insights are intended to clarify the technical feasibility and commercial potential of the material for stakeholders evaluating its integration into their supply chain. The answers reflect the specific advantages of the novel method over conventional approaches, highlighting the improvements in yield, purity, and biological activity. Understanding these details is essential for making informed decisions regarding the procurement and development of this specialized chemical entity. For further technical specifications or custom synthesis inquiries, direct consultation with the technical team is recommended.
Q: What is the primary advantage of this Ruthenium complex over Cisplatin?
A: The novel Ruthenium complex offers lower toxicity and higher water solubility compared to Cisplatin, along with enhanced DNA binding affinity due to electron-withdrawing nitro and bromine groups.
Q: How does the synthesis method ensure high purity?
A: The method utilizes mild reaction conditions and specific recrystallization steps, such as using absolute ethanol and methanol, which facilitate the removal of impurities and yield stable crystalline products.
Q: Is this process scalable for industrial production?
A: Yes, the patent describes a simple preparation method with stable intermediate products and high yields in each step, indicating strong potential for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Novel Ruthenium Complex Supplier
The synthesis route detailed in patent CN105884833B demonstrates significant technical potential for the production of advanced antitumor agents, and NINGBO INNO PHARMCHEM is well-positioned to support the commercialization of such complex molecules. As a CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from benchtop to market. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of the ruthenium complex meets the highest standards required for pharmaceutical applications. We understand the critical nature of supply continuity and quality consistency in the drug development lifecycle, and our team is dedicated to providing the reliability you need. Partnering with us means gaining access to a wealth of technical expertise and manufacturing capacity tailored to the unique demands of specialty chemical synthesis.
We invite you to initiate a dialogue with our technical procurement team to discuss how we can assist in optimizing your supply chain for this specific compound. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our manufacturing efficiencies can translate into tangible benefits for your project budget. We encourage you to reach out for specific COA data and route feasibility assessments to verify our capability to meet your exact requirements. Our goal is to be your strategic partner in bringing innovative therapies to market, leveraging our technical prowess to overcome synthesis challenges. Contact us today to explore the possibilities of collaborating on this high-potential ruthenium complex project.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
