Revolutionizing Pyridoxal Phosphate Production: Advanced Biocatalytic Strategies for Commercial Scale-Up
The pharmaceutical and nutritional industries are constantly seeking more efficient and sustainable pathways for producing essential bioactive compounds, and Pyridoxal Phosphate (PLP), the active form of Vitamin B6, stands as a prime candidate for process innovation. As detailed in the groundbreaking patent CN113481253A, a novel biocatalytic method has been developed that fundamentally shifts the production paradigm from traditional harsh chemical synthesis to a sophisticated enzymatic approach. This technology leverages a dual-enzyme system involving phosphotransferase and alcohol oxidase to convert pyridoxine directly into Pyridoxal Phosphate with exceptional precision. For R&D Directors and Technical Procurement Managers, understanding the nuances of this patent is crucial, as it offers a pathway to higher purity intermediates while drastically reducing the environmental footprint associated with legacy manufacturing processes. The implications of this technology extend beyond mere laboratory curiosity; it represents a viable, scalable solution for the reliable Pyridoxal Phosphate supplier market, addressing critical pain points regarding cost, safety, and regulatory compliance in the production of high-purity nutritional ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Pyridoxal Phosphate has relied heavily on purely chemical synthetic routes that are fraught with significant technical and economic inefficiencies. The traditional method typically involves the reaction of pyridoxamine dihydrochloride with anhydrous phosphoric acid, followed by a rigorous oxidation step using manganese dioxide. This legacy approach presents multiple severe drawbacks that hinder modern manufacturing efficiency. Firstly, the concentration of the product in the reaction solution is often inherently low, necessitating energy-intensive evaporation and dehydration steps to concentrate the final product. These thermal processes not only consume substantial amounts of energy but also pose a risk of degrading the heat-sensitive vitamin derivative, potentially compromising the quality and stability of the final API intermediate. Furthermore, the reliance on strong acids and heavy metal oxidants generates a substantial volume of hazardous wastewater, creating a complex and costly waste management burden for chemical plants. The multi-step nature of the chemical synthesis also introduces more opportunities for side reactions, leading to a broader impurity spectrum that requires extensive and expensive downstream purification to meet the stringent purity specifications demanded by the pharmaceutical industry.
The Novel Approach
In stark contrast to the cumbersome chemical routes, the biocatalytic method disclosed in the patent introduces a streamlined, eco-friendly alternative that capitalizes on the exquisite specificity of biological enzymes. By utilizing a holoenzyme catalytic system, this new approach facilitates the conversion of pyridoxine to Pyridoxal Phosphate under remarkably mild conditions, specifically at a physiological temperature of 37°C and a neutral pH range of 7.3 to 7.6. This shift eliminates the need for extreme temperatures and corrosive reagents, thereby preserving the structural integrity of the sensitive vitamin molecule. The process is characterized by its high selectivity, which inherently minimizes the formation of by-products and simplifies the purification workflow. Moreover, the patent highlights the versatility of this method, offering both a step-by-step reaction protocol and a consolidated one-step reaction strategy. This flexibility allows manufacturers to optimize their production lines based on specific capacity and equipment constraints, ultimately leading to a process that is not only environmentally superior but also operationally more convenient and cost-effective for the commercial scale-up of complex nutritional ingredients.
Mechanistic Insights into Dual-Enzyme Catalytic Conversion
To fully appreciate the technical robustness of this synthesis route, one must delve into the mechanistic details of the enzymatic cascade. The core of this innovation lies in the synergistic action of phosphotransferase and alcohol oxidase, supported by essential cofactors such as ATP sodium salt and FMN (Flavin Mononucleotide). The reaction initiates with the phosphorylation of pyridoxine, where phosphotransferase catalyzes the transfer of a phosphate group from ATP to the hydroxymethyl group of the pyridoxine substrate. This step is critically dependent on the presence of inorganic salts, such as anhydrous magnesium sulfate or zinc chloride, which act as stabilizers and activators for the enzyme system. Following phosphorylation, the intermediate undergoes oxidation mediated by alcohol oxidase. This enzyme specifically targets the hydroxymethyl group, converting it into the aldehyde functionality characteristic of Pyridoxal Phosphate. The precise control of reaction parameters, including the molar ratios of enzymes to substrates and the maintenance of the pH within the narrow window of 7.3 to 7.6, ensures that the catalytic cycle proceeds with maximal efficiency. This level of control is unattainable in non-specific chemical oxidation, highlighting why this biocatalytic route is superior for generating high-purity OLED material precursors or pharmaceutical intermediates where structural fidelity is paramount.
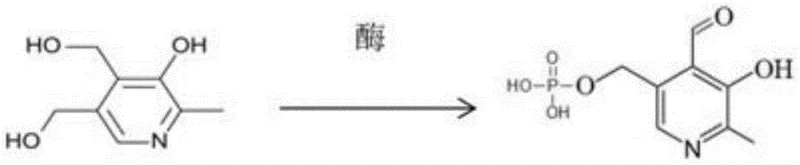
Furthermore, the impurity control mechanism inherent in this enzymatic process is a significant advantage for quality assurance teams. In chemical synthesis, over-oxidation or non-specific phosphorylation can lead to a complex mixture of analogues that are difficult to separate. However, enzymes possess active sites that are stereospecific and regioselective, meaning they only react with the intended functional groups on the pyridoxine molecule. This biological specificity drastically reduces the formation of structurally related impurities, resulting in a cleaner crude reaction mixture. Consequently, the downstream processing burden is significantly lightened, reducing the need for multiple recrystallization or chromatography steps. For the production of high-purity Pyridoxal Phosphate, this means a higher overall yield of the target molecule and a final product that consistently meets rigorous pharmacopoeial standards. The ability to tune the enzyme ratios, as demonstrated in the patent examples where varying the mass of alcohol oxidase impacts conversion rates, provides process engineers with powerful levers to optimize performance without altering the fundamental chemistry of the synthesis.
How to Synthesize Pyridoxal Phosphate Efficiently
Implementing this biocatalytic route in a production environment requires a clear understanding of the operational parameters defined in the patent literature. The process is designed to be adaptable, allowing for both batch and potentially continuous flow configurations depending on the reactor setup. The foundational step involves the preparation of the aqueous reaction matrix, ensuring that the phosphate buffer capacity is sufficient to maintain the pH stability throughout the exothermic enzymatic reactions. Detailed standard operating procedures regarding the sequential addition of cofactors like FMN and ATP are critical to prevent enzyme inhibition or substrate degradation. While the general concept is straightforward, the precise execution determines the success of the scale-up. For a comprehensive breakdown of the exact reagent volumes, incubation times, and workup procedures validated by experimental data, please refer to the standardized synthesis guide below.
- Prepare the reaction system by adding ddH2O, phosphate buffer (pH 7.3-7.6), ATP sodium salt, inorganic salt, and pyridoxine solution.
- Introduce phosphotransferase and maintain the temperature at 37°C for 1.5 hours to facilitate phosphorylation.
- Add alcohol oxidase and FMN solution to the supernatant, stir for 40-60 minutes to complete the oxidation to Pyridoxal Phosphate.
Commercial Advantages for Procurement and Supply Chain Teams
For Procurement Managers and Supply Chain Heads, the transition to this biocatalytic methodology offers compelling strategic advantages that go beyond simple technical metrics. The primary value proposition lies in the substantial cost savings driven by process simplification and resource efficiency. By eliminating the requirement for hazardous oxidizing agents like manganese dioxide and strong mineral acids, the facility reduces its expenditure on specialized safety equipment, hazardous waste disposal, and regulatory compliance measures. The milder reaction conditions also translate to lower energy consumption, as there is no need for high-temperature heating or extensive cooling systems to manage violent exotherms. These operational efficiencies accumulate to create a significantly reduced cost base for the manufacturing of Pyridoxal Phosphate, allowing suppliers to offer more competitive pricing structures without compromising on margin. Additionally, the aqueous nature of the reaction reduces the reliance on volatile organic solvents, further enhancing the sustainability profile of the supply chain and aligning with the increasing corporate mandates for green chemistry practices.
- Cost Reduction in Manufacturing: The economic benefits of this enzymatic route are derived from the elimination of expensive and dangerous chemical reagents traditionally used in oxidation steps. By replacing heavy metal catalysts with biocatalysts, the process removes the costly downstream unit operations required to strip trace metals from the final product to meet safety regulations. This simplification of the purification train not only saves on consumables and filtration media but also increases the overall throughput of the production line. The high conversion yields reported in the patent data suggest that raw material utilization is maximized, minimizing the loss of valuable pyridoxine starting material. Consequently, the total cost of goods sold (COGS) is optimized, providing a robust buffer against fluctuations in raw material market prices and ensuring long-term price stability for buyers seeking cost reduction in nutritional ingredients manufacturing.
- Enhanced Supply Chain Reliability: From a logistics and supply continuity perspective, the biocatalytic process offers distinct advantages regarding safety and storage. The absence of hazardous chemicals reduces the regulatory burden associated with the transportation and warehousing of raw materials, mitigating the risk of supply disruptions caused by stricter transport regulations or safety incidents. Furthermore, the enzymes used in this process, being biological entities, can often be sourced from stable, renewable supply chains, reducing dependency on petrochemical-derived reagents that are subject to volatile market dynamics. The robustness of the reaction conditions (37°C, neutral pH) also means that the manufacturing process is less susceptible to deviations caused by minor utility fluctuations, ensuring consistent batch-to-batch quality. This reliability is critical for reducing lead time for high-purity Pyridoxal Phosphate derivatives, as it minimizes the need for re-processing or rejecting out-of-specification batches that could otherwise delay shipments to global clients.
- Scalability and Environmental Compliance: Scaling a chemical process often amplifies its environmental risks, but this biocatalytic method scales with a significantly lower environmental penalty. The wastewater generated is primarily aqueous with biodegradable organic content, making it far easier and cheaper to treat compared to the heavy-metal-laden effluent from chemical oxidation. This ease of waste treatment facilitates faster regulatory approvals for plant expansions or new production lines, accelerating the time-to-market for increased capacity. The process is inherently designed for industrial application, with patent examples demonstrating efficacy in standard glass reaction vessels that can be directly translated to stainless steel bioreactors. This seamless scalability ensures that suppliers can rapidly respond to surges in demand for pharmaceutical intermediates without the need for massive capital investment in specialized corrosion-resistant infrastructure, thereby securing the supply chain against future market volatility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this biocatalytic technology. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing procurement strategies or R&D pipelines. The answers reflect the consensus on how this technology resolves historical bottlenecks in Vitamin B6 derivative production.
Q: What are the primary advantages of the biocatalytic method over chemical synthesis for Pyridoxal Phosphate?
A: The biocatalytic method operates under significantly milder conditions (37°C, neutral pH) compared to the harsh chemical oxidation processes. It eliminates the need for dangerous oxidizing agents like manganese dioxide, resulting in a cleaner impurity profile and reduced environmental waste.
Q: How does the enzyme ratio affect the conversion yield in this process?
A: Optimization of the phosphotransferase to alcohol oxidase ratio is critical. Patent data indicates that specific ratios, such as 8mg phosphotransferase to 0.8mg alcohol oxidase, can achieve conversion rates as high as 93% in one-step reactions, maximizing raw material efficiency.
Q: Is this biocatalytic process suitable for large-scale industrial manufacturing?
A: Yes, the process demonstrates high industrial application value due to its operational simplicity and high selectivity. The avoidance of complex purification steps required for removing heavy metal catalysts makes it highly scalable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyridoxal Phosphate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced biocatalytic technologies like the one described in patent CN113481253A requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial tank is seamless and efficient. Our facilities are equipped with state-of-the-art bioreactors and stringent purity specifications are enforced through our rigorous QC labs, guaranteeing that every batch of Pyridoxal Phosphate meets the highest international standards. We understand the critical nature of supply chain continuity for global pharmaceutical and nutritional companies, and our robust quality management systems are designed to mitigate risk and ensure consistent delivery of high-quality intermediates.
We invite you to collaborate with us to leverage these technological advancements for your specific product needs. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your current procurement volumes and quality requirements. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data rather than assumptions. By partnering with us, you gain access to a supply chain that is not only cost-effective but also aligned with the future of sustainable and efficient chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
