Advanced Immobilized Enzyme Technology for Commercial Pyridoxal Phosphate Production
The pharmaceutical and nutritional industries are constantly seeking more sustainable and efficient pathways for producing essential vitamins and their active derivatives. A significant breakthrough in this domain is documented in patent CN115094102A, which details a novel method for preparing pyridoxal phosphate using immobilized enzyme technology. This innovation represents a paradigm shift from traditional harsh chemical synthesis to a biocatalytic approach that leverages the specificity of phosphotransferase and alcohol oxidase. By transitioning to this enzymatic route, manufacturers can achieve a process that is not only environmentally benign but also operationally superior in terms of control and reproducibility. The technical implications of this patent extend far beyond the laboratory, offering a robust framework for industrial scale-up that addresses critical pain points in the supply chain for high-purity vitamin intermediates. As a reliable vitamin B6 derivative supplier, understanding the nuances of this technology is essential for maintaining competitive advantage in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of pyridoxal phosphate has relied heavily on pure chemical synthesis methods that involve multiple strenuous steps and hazardous reagents. The conventional process typically requires the reaction of pyridoxamine dihydrochloride with anhydrous phosphoric acid, followed by oxidation using manganese dioxide, which introduces significant safety and environmental concerns. One of the most critical drawbacks of this chemical route is the necessity for evaporation dehydration to concentrate the product, a step that consumes excessive energy and exposes the thermally sensitive vitamin structure to high temperatures that can degrade quality. Furthermore, the chemical process generates a substantial volume of wastewater and consumes dangerous chemicals, creating a heavy burden on waste treatment facilities and increasing the overall operational cost. The low concentration of the product in the solution often necessitates complex downstream processing, which further elongates the production cycle and reduces the overall yield efficiency. These inherent limitations make the traditional chemical method increasingly unsustainable in the context of modern green chemistry standards and cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
In stark contrast to the cumbersome chemical routes, the novel immobilized enzyme method introduced in the patent offers a streamlined and mild alternative that fundamentally changes the production landscape. This approach utilizes a two-step biocatalytic conversion where pyridoxine is transformed into pyridoxal phosphate under physiological conditions, specifically at a temperature of 37°C and a pH range of 7.3 to 7.6. The use of immobilized enzymes allows for the catalyst to be fixed within a reactor, enabling a continuous flow process that eliminates the need for separating free enzymes from the product after each reaction cycle. This technological advancement drastically simplifies the operational workflow, reducing the time consumption associated with batch processing and enzyme recovery. By avoiding harsh oxidants and high-temperature concentration steps, the novel approach preserves the integrity of the final product while significantly lowering the environmental footprint. This method exemplifies how commercial scale-up of complex enzymatic reactions can be achieved with high efficiency and minimal resource consumption.
Mechanistic Insights into Immobilized Enzyme Catalytic Conversion
The core of this innovative process lies in the synergistic action of two specific enzymes: phosphotransferase and alcohol oxidase, which work in a coordinated cascade to transform the substrate. The reaction begins with the phosphorylation of pyridoxine, facilitated by phosphotransferase in the presence of ATP sodium salt and magnesium sulfate, which acts as a crucial cofactor for enzyme activity. Following this initial modification, the intermediate is subjected to oxidation by alcohol oxidase, which requires FMN as a coenzyme to successfully convert the hydroxymethyl group into the aldehyde functionality characteristic of pyridoxal phosphate. This dual-enzyme system is meticulously engineered to operate in a sequential manner, ensuring that each step proceeds with high specificity and minimal formation of byproducts. The immobilization of these enzymes onto a solid support, such as nickel ion metal chelate affinity chromatography medium or epoxy resin, stabilizes their structure and enhances their operational longevity. 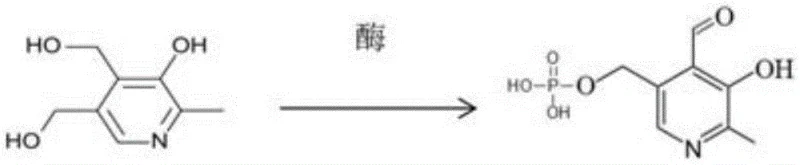
Controlling the impurity profile in vitamin synthesis is paramount for meeting the stringent quality standards required by regulatory bodies and end-users. The enzymatic nature of this reaction inherently limits the formation of non-specific byproducts that are common in chemical oxidation processes, thereby simplifying the purification workflow. The mild reaction conditions prevent the degradation of the pyridoxal phosphate molecule, which is susceptible to decomposition under acidic or high-temperature environments often found in traditional synthesis. By maintaining a precise pH of 7.3 to 7.6, the system ensures optimal enzyme activity while minimizing the risk of hydrolysis or other degradation pathways. This high level of control over the reaction environment translates directly into a cleaner crude product, reducing the load on downstream chromatography or crystallization steps. For R&D directors focused on purity and impurity profiles, this mechanism offers a compelling solution for producing high-purity pyridoxal phosphate with consistent batch-to-batch quality.
How to Synthesize Pyridoxal Phosphate Efficiently
Implementing this synthesis route requires a structured approach to enzyme preparation and reactor configuration to maximize catalytic efficiency and product yield. The process begins with the fermentation of genetically engineered Escherichia coli strains to produce the target enzymes, followed by cell lysis and purification to obtain crude enzyme solutions ready for immobilization. Once the enzymes are secured onto the chosen support medium, they are packed into column or tank reactors where the substrate solution is introduced under controlled flow rates. The detailed standardized synthesis steps involve precise management of cofactor concentrations, such as ATP and FMN, to sustain the catalytic cycle over extended periods. For a comprehensive understanding of the operational parameters and specific reactor setups, the detailed standardized synthesis steps are provided in the guide below.
- Ferment Escherichia coli strains containing phosphotransferase and alcohol oxidase genes to produce free enzymes, followed by cell lysis and supernatant extraction.
- Immobilize the extracted enzymes onto a nickel ion metal chelate affinity chromatography medium or epoxy resin to create reusable catalytic reactors.
- Flow a reaction solution containing pyridoxine, ATP, and FMN through the immobilized enzyme reactors at 37°C and pH 7.3-7.6 to generate pyridoxal phosphate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this immobilized enzyme technology presents substantial advantages that directly address the core concerns of procurement managers and supply chain heads. The elimination of hazardous chemical oxidants and the reduction in energy-intensive concentration steps lead to a significant decrease in raw material and utility costs. Moreover, the reusability of the immobilized enzymes means that the cost of the biocatalyst is amortized over multiple production batches, driving down the unit cost of the final product. This process enhancement also mitigates supply chain risks associated with the availability of specialized chemical reagents, as the biological components can be produced consistently through fermentation. By streamlining the production workflow, manufacturers can achieve faster turnaround times, effectively reducing lead time for high-purity vitamins and ensuring a more reliable supply for downstream customers.
- Cost Reduction in Manufacturing: The transition to an enzymatic process eliminates the need for expensive and hazardous chemical oxidants like manganese dioxide, which simplifies the procurement of raw materials and reduces safety compliance costs. Additionally, the mild reaction conditions remove the requirement for high-energy evaporation steps, leading to substantial savings in utility consumption and equipment maintenance. The ability to reuse the immobilized enzyme catalyst over multiple cycles further distributes the initial enzyme cost across a larger volume of product, significantly lowering the variable cost per kilogram. These combined factors result in a more economically viable production model that enhances profit margins without compromising on product quality.
- Enhanced Supply Chain Reliability: Relying on biological fermentation for enzyme production creates a more stable and scalable supply of catalysts compared to sourcing specialized chemical reagents that may be subject to market volatility. The robust nature of the immobilized enzyme reactors allows for continuous or semi-continuous operation, which minimizes downtime between batches and ensures a steady output of product. This operational consistency is crucial for maintaining long-term supply contracts and meeting the just-in-time delivery requirements of large-scale pharmaceutical clients. Furthermore, the reduced generation of hazardous waste simplifies logistics and disposal, removing potential bottlenecks that could disrupt the manufacturing schedule.
- Scalability and Environmental Compliance: The modular nature of the immobilized enzyme reactors facilitates easy scale-up from laboratory pilot studies to full commercial production without the need for complex process re-engineering. The environmentally friendly profile of the process, characterized by lower waste generation and milder conditions, ensures compliance with increasingly strict environmental regulations across different global jurisdictions. This sustainability advantage not only reduces the risk of regulatory fines but also aligns with the corporate social responsibility goals of major multinational customers. The simplified waste stream also lowers the cost and complexity of effluent treatment, making the facility more agile and responsive to production demands.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this immobilized enzyme technology for pyridoxal phosphate production. These insights are derived directly from the patent data and are intended to clarify the operational benefits and feasibility of adopting this route. Understanding these details is critical for stakeholders evaluating the potential integration of this technology into their existing manufacturing portfolios. The answers provided reflect the specific advantages of the enzymatic method over traditional chemical synthesis.
Q: How does the immobilized enzyme method compare to traditional chemical oxidation?
A: Unlike traditional chemical methods that require harsh oxidants like manganese dioxide and high-temperature concentration, the immobilized enzyme method operates at mild physiological temperatures (37°C) and neutral pH, significantly reducing energy consumption and preventing thermal degradation of the sensitive vitamin structure.
Q: Can the immobilized enzymes be reused for multiple production batches?
A: Yes, a key advantage of this technology is the reusability of the biocatalyst. The enzymes are fixed onto a solid support medium, allowing them to be retained within the reactor while the product flows through, enabling continuous operation over multiple batches without the need for fresh enzyme addition each time.
Q: What are the purity implications of using this enzymatic route?
A: The enzymatic specificity minimizes side reactions common in chemical synthesis, leading to a cleaner impurity profile. This reduces the burden on downstream purification steps, resulting in high-purity pyridoxal phosphate suitable for sensitive pharmaceutical and nutritional applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyridoxal Phosphate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this enzymatic route and possess the technical expertise to bring such complex pathways to industrial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust manufacturing processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of pyridoxal phosphate meets the highest international standards for pharmaceutical and nutritional use. Our commitment to technical excellence allows us to navigate the complexities of enzyme immobilization and continuous flow chemistry with precision and reliability.
We invite you to collaborate with us to optimize your supply chain and leverage these advanced manufacturing capabilities for your product portfolio. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate how we can support your long-term production goals. By partnering with us, you gain access to a supply chain that is not only cost-effective but also resilient and aligned with the future of green chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
