Advanced Catalytic Strategy For Commercial Scale Muscone Production And Supply Chain Optimization
Introduction To The Breakthrough In Macrocyclic Ketone Synthesis
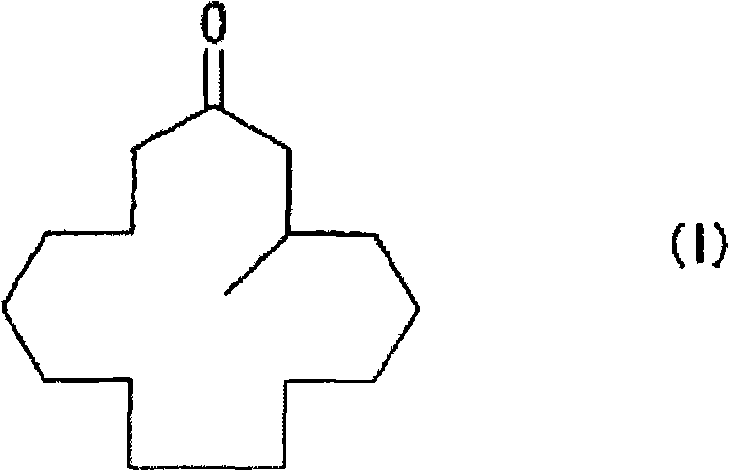
The chemical industry constantly seeks more efficient pathways for producing high-value fragrance intermediates, and the technology disclosed in patent CN101056843B represents a significant leap forward in the manufacturing of muscone. This specific intellectual property details a novel process for producing macrocyclic ketones through a 1,4-conjugated methyl addition reaction of 2-cyclopentadecenone, achieving high yields under practical and manageable conditions. Unlike traditional methods that often struggle with low efficiency or harsh requirements, this approach utilizes a copper or nickel catalyst alongside a specialized enol-based anion trapping agent to stabilize reaction intermediates. The result is a robust synthetic route that avoids the need for extremely low temperatures or dilute reaction conditions, which are typically cost-prohibitive in large-scale operations. By effectively managing the reactivity of the enolate anion, the process minimizes side reactions and maximizes the formation of the desired 3-methyl-1-cyclopentadecene derivatives. This technological advancement provides a solid foundation for producing high-purity muscone, a critical component in the fine chemical and fragrance sectors, with enhanced economic feasibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active muscone has been plagued by significant technical and economic hurdles that hinder efficient commercial production. Conventional methods often rely on chiral auxiliary groups or specific ligand systems that require reaction temperatures as low as -78°C to maintain stereocontrol and prevent decomposition. These extremely low-temperature conditions demand specialized cryogenic equipment and substantial energy inputs, driving up the operational costs significantly for any manufacturing facility. Furthermore, previous reports indicate that maintaining high yields often necessitates very low concentration conditions, sometimes requiring solvent-to-substrate ratios as high as 50:1, which drastically reduces reactor throughput and increases solvent recovery burdens. The formation of high molecular weight by-products is another persistent issue when attempting to run these reactions at higher concentrations, leading to complex purification steps and reduced overall material efficiency. Additionally, the use of stoichiometric amounts of chiral auxiliaries in older methods generates substantial chemical waste, conflicting with modern green chemistry principles and environmental compliance standards. These combined factors create a bottleneck that limits the scalability and cost-effectiveness of traditional muscone manufacturing processes.
The Novel Approach
The patented methodology introduces a transformative strategy by incorporating an enol-based anion trapping agent into the catalytic cycle, fundamentally altering the reaction landscape for macrocyclic ketone synthesis. This innovative approach allows the 1,4-conjugate methyl addition to proceed efficiently at much higher concentrations, thereby maximizing reactor utilization and minimizing solvent usage without compromising product quality. By trapping the reactive enolate anion intermediate immediately after formation, the process effectively suppresses the generation of unwanted polymeric by-products that typically plague high-concentration reactions. The operational temperature range is significantly relaxed to between -30°C and 30°C, eliminating the need for expensive cryogenic cooling systems and allowing for the use of standard industrial reactors. This shift not only reduces capital expenditure on specialized equipment but also lowers the ongoing energy costs associated with maintaining extreme thermal conditions. Consequently, the novel approach offers a streamlined pathway that enhances yield consistency and simplifies downstream processing, making it an attractive option for industrial adoption.
Mechanistic Insights into Cu-Catalyzed 1,4-Conjugate Addition
The core of this synthetic breakthrough lies in the precise mechanistic control exerted by the copper or nickel catalyst system in conjunction with the anion trapping agent. During the reaction, the methylated organometallic reagent undergoes a 1,4-conjugate addition to the 2-cyclopentadecenone substrate, generating a highly reactive enolate anion intermediate that is prone to side reactions if left unchecked. The introduction of the trapping agent, such as an acid anhydride or chlorosilane, immediately captures this enolate to form a stable 3-methyl-1-cyclopentadecene derivative, effectively freezing the stereochemistry and preventing further unwanted transformations. This stabilization step is crucial for maintaining high selectivity and yield, as it isolates the intermediate from the harsh reaction environment until it can be safely processed in the subsequent step. The choice of catalyst, often copper triflate or similar species, works synergistically with optically active ligands to induce the desired chirality at the 3-position of the macrocyclic ring. This level of mechanistic sophistication ensures that the final muscone product possesses the specific optical activity required for high-quality fragrance applications.
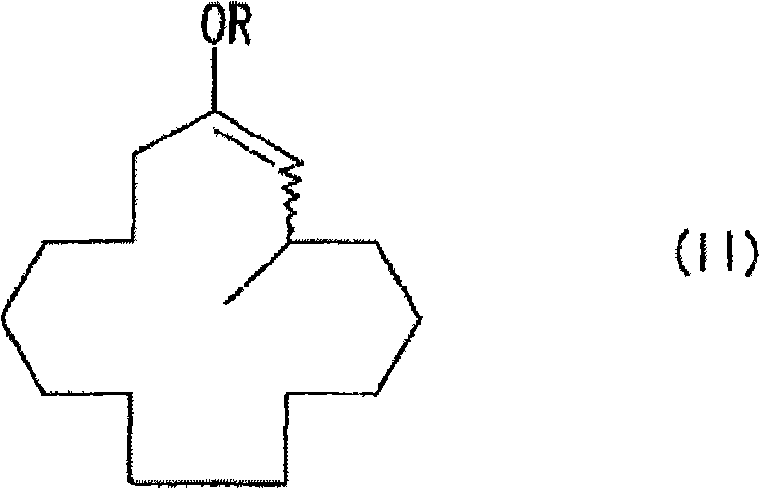
Controlling the impurity profile is another critical aspect of this mechanism, achieved through the careful selection of trapping agents and reaction conditions. The formation of the enol derivative acts as a protective measure, shielding the reactive center from nucleophilic attacks that could lead to racemization or degradation. Subsequent solvolysis of this protected intermediate under mild acidic or basic conditions releases the final ketone functionality without disturbing the established stereocenter. This two-step sequence, addition followed by trapping and then solvolysis, provides a robust handle on the impurity spectrum, ensuring that high molecular weight by-products are kept to a minimum. The ability to tune the trapping agent allows chemists to optimize the balance between reaction rate and selectivity, further refining the purity of the crude product before distillation. Such precise control over the chemical pathway is essential for meeting the stringent quality specifications demanded by the global fragrance and flavor industry.
How to Synthesize Muscone Efficiently
Implementing this synthesis route requires a clear understanding of the sequential steps involved in transforming the starting enone into the final macrocyclic ketone. The process begins with the preparation of the catalytic system, where the copper salt and chiral ligand are mixed under an inert atmosphere to ensure optimal activity and prevent oxidation. Following this, the 2-cyclopentadecenone substrate is introduced along with the methylating agent and the crucial enol anion trapping agent, initiating the conjugate addition and stabilization sequence. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction system with copper catalyst and optically active ligand under inert atmosphere.
- Conduct 1,4-conjugate methyl addition on 2-cyclopentadecenone in the presence of an enol anion trapping agent.
- Perform solvolysis on the resulting 3-methyl-1-cyclopentadecene derivative to obtain the final muscone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented process offers compelling advantages that directly address the pain points of cost volatility and production reliability in the fine chemical sector. The elimination of extreme low-temperature requirements translates into a significant reduction in energy consumption and infrastructure maintenance, lowering the overall cost of goods sold for the manufacturer. By enabling high-concentration reactions, the process maximizes the output per batch, effectively increasing asset utilization rates and reducing the time required to fulfill large volume orders. This efficiency gain is critical for supply chain heads who need to ensure consistent availability of key fragrance intermediates without facing bottlenecks caused by slow reaction kinetics or complex workups. Furthermore, the simplified purification profile reduces the demand for extensive chromatographic separations, speeding up the production cycle and minimizing solvent waste disposal costs. These operational improvements collectively enhance the resilience of the supply chain, making it less susceptible to disruptions caused by equipment failures or raw material shortages.
- Cost Reduction in Manufacturing: The economic benefits of this technology are driven primarily by the removal of energy-intensive cryogenic cooling steps and the reduction in solvent volumes required for dilute reactions. Operating at near-ambient temperatures allows facilities to utilize standard cooling systems rather than specialized low-temperature reactors, resulting in substantial capital and operational expenditure savings. Additionally, the high yield achieved through the trapping agent strategy means less raw material is wasted on by-products, improving the overall material balance and reducing the cost per kilogram of the final product. The ability to recycle solvents more efficiently due to higher concentrations further contributes to the long-term cost competitiveness of this manufacturing route. These factors combine to create a more economically sustainable production model that can withstand market fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: Reliability in the supply of high-purity muscone is paramount for fragrance houses that rely on consistent quality for their formulations. This process enhances reliability by simplifying the reaction conditions, thereby reducing the risk of batch failures due to temperature excursions or mixing issues common in cryogenic processes. The robustness of the catalytic system ensures consistent conversion rates, allowing for more accurate production planning and inventory management. Moreover, the use of commercially available catalysts and trapping agents reduces dependency on exotic or hard-to-source reagents, mitigating supply risk. This stability allows procurement managers to negotiate better terms with suppliers who can guarantee on-time delivery and consistent quality specifications. Ultimately, a more reliable production process leads to a more secure supply chain for downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to industrial production often reveals hidden challenges, but this methodology is inherently designed for scalability. The high-concentration capability means that reactor volumes can be utilized more effectively, facilitating a smoother transition from pilot plant to full-scale manufacturing without the need for disproportionate increases in equipment size. Environmental compliance is also improved as the process generates less waste solvent and avoids the use of stoichiometric chiral auxiliaries that contribute to heavy waste loads. The reduced energy footprint aligns with corporate sustainability goals, making the product more attractive to environmentally conscious buyers. This alignment with green chemistry principles ensures long-term regulatory compliance and reduces the risk of future environmental penalties or restrictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced muscone synthesis technology. These answers are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners and stakeholders. Understanding these details is crucial for evaluating the feasibility of integrating this process into existing manufacturing workflows. The responses highlight the key differentiators that set this method apart from legacy technologies in terms of efficiency and quality.
Q: How does the enol anion trapping agent improve muscone yield?
A: The trapping agent captures the enolate anion generated during the 1,4-conjugate addition, preventing the formation of high molecular weight by-products and allowing the reaction to proceed at higher concentrations with significantly improved yield.
Q: What are the temperature advantages of this new process compared to conventional methods?
A: Unlike conventional methods requiring extremely low temperatures like -78°C, this patented process operates effectively at temperatures ranging from -30°C to 30°C, drastically reducing energy consumption and equipment complexity.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the method eliminates the need for low-concentration reaction conditions and extreme cooling, making it highly scalable and economically viable for industrial manufacturing of high-purity muscone.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Muscone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting advanced synthetic routes like the one described in CN101056843B to maintain a competitive edge in the global fragrance market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into reliable industrial realities. We are committed to delivering high-purity muscone that meets stringent purity specifications through our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging this optimized catalytic technology, we can offer our clients a supply of fragrance intermediates that is both cost-effective and consistently high in quality. Our dedication to technical excellence ensures that every batch produced adheres to the highest standards of the industry.
We invite you to collaborate with us to explore how this innovative synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate the tangible benefits of partnering with us. Let us help you secure a stable and efficient supply of high-purity muscone for your future product developments.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
