Advanced Imide-Structured Aromatic Diamine Monomers for Next-Generation Electronic Materials Manufacturing
The landscape of high-performance polymer manufacturing is undergoing a significant transformation driven by the demand for materials that combine exceptional thermal stability with enhanced processability. Patent CN103145581A introduces a groundbreaking class of aromatic diamine compounds containing imide structures, specifically designed to address the longstanding solubility issues associated with traditional polyimides. These novel monomers possess a unique rigid non-planar twisted structure that disrupts molecular packing, thereby facilitating dissolution in common organic solvents without compromising their inherent thermal resistance. This technological advancement is pivotal for the development of next-generation electronic chemicals, particularly in the realms of microelectronics and optoelectronics where material precision is paramount.
As a reliable electronic chemical supplier, understanding the structural nuances of these monomers is critical for R&D teams aiming to push the boundaries of polymer performance. The compounds disclosed in this patent not only serve as robust monomers for synthesizing polyamides, polyimides, and polyester-imides but also exhibit intrinsic photoelectric properties. For instance, specific derivatives demonstrate aggregation-induced luminescence, opening new avenues for their application as active layers in organic light-emitting diodes (OLEDs). This dual functionality positions these materials at the forefront of both structural and functional material science.
![Chemical structure of 4-(Bis(4-aminophenyl)amino)-N,N-bis(4'-(trifluoromethyl)-[1,1'-biphenyl]-4-yl)-benzamide showing trifluoromethyl groups for enhanced solubility](/insights/img/imide-aromatic-diamine-monomers-polyimide-supplier-20260305161021-09.png)
The limitations of conventional polyimide synthesis often stem from the rigid planar structure of traditional dianhydride and diamine monomers, which leads to strong intermolecular interactions and poor solubility. This necessitates the use of harsh processing conditions or expensive fluorinated solvents, driving up the cost reduction in display material manufacturing. Furthermore, the infusibility of standard polyimides restricts their molding and shaping capabilities, limiting their application in complex microelectronic components. The industry has long sought a balance between the mechanical strength provided by aromatic rings and the processability granted by flexible linkages, a balance that traditional methods struggle to achieve efficiently.
The novel approach detailed in the patent overcomes these hurdles by strategically incorporating imide linkages and bulky aromatic substituents into the diamine backbone. By utilizing the high reactivity of acid chlorides with active hydrogen, the synthesis creates a pre-organized imide structure before polymerization. This method allows for the introduction of diverse functional groups, such as trifluoromethyl or biphenyl units, via Suzuki coupling or nucleophilic substitution. The resulting molecules maintain the thermal integrity required for aerospace and military applications while offering the solubility needed for solution processing, effectively bridging the gap between performance and manufacturability.
Mechanistic Insights into Imide Formation and Functionalization
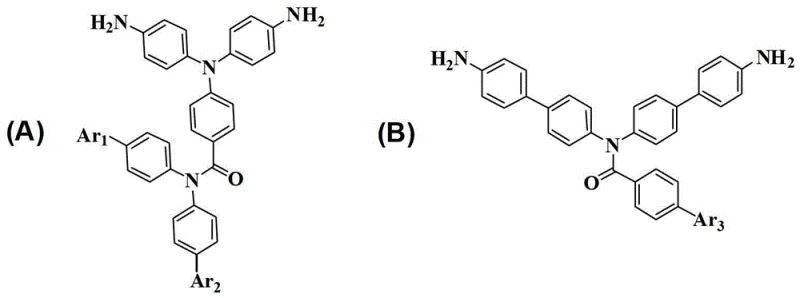
The synthesis mechanism relies on a precise sequence of acylation, reduction, and coupling reactions that ensure high purity and structural fidelity. Initially, the reaction between p-nitrobenzoyl chloride and diphenylamine under alkaline conditions forms a nitro-containing imide intermediate. This step is crucial as it establishes the core imide linkage which contributes to the thermal stability of the final polymer. Subsequent reduction of the nitro group to an amine using hydrazine hydrate or catalytic hydrogenation activates the molecule for further functionalization. This activation step is performed under controlled temperatures to prevent side reactions, ensuring a clean intermediate profile.
Following activation, the introduction of specific aromatic groups is achieved through either nucleophilic substitution with halonitrobenzenes or palladium-catalyzed Suzuki coupling with boronic acids. This flexibility allows for the fine-tuning of the monomer's electronic and steric properties. For example, the introduction of trifluoromethyl groups enhances solubility and lowers the dielectric constant, which is vital for high-purity OLED material applications. The final reduction step converts the dinitro precursors into the target diamines. The entire pathway is designed to minimize impurities, with column chromatography and recrystallization steps integrated to meet stringent purity specifications required for electronic grade materials.
How to Synthesize Imide-Structured Aromatic Diamines Efficiently
The synthesis of these high-value monomers involves a multi-step protocol that balances reaction efficiency with product quality. The process begins with the formation of the imide core, followed by sequential functionalization and reduction steps. Each stage requires careful control of reaction parameters such as temperature, solvent choice, and catalyst loading to maximize yield and minimize byproduct formation. The detailed standardized synthesis steps below outline the critical operational parameters derived from the patent examples, providing a roadmap for laboratory and pilot-scale production.
- Synthesize the nitro-containing imide intermediate by reacting acid chloride with active hydrogen sources like diphenylamine under alkaline conditions.
- Reduce the nitro group to an amino group using catalytic hydrogenation or hydrazine hydrate to form the amino-imide intermediate.
- Perform nucleophilic substitution with halonitrobenzene or Suzuki coupling with boronic acids to introduce functional groups, followed by final reduction to the diamine.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this synthetic route offers substantial cost savings and supply chain resilience. The reliance on commercially available starting materials such as diphenylamine, acid chlorides, and boronic acids reduces dependency on exotic reagents that often suffer from supply volatility. Furthermore, the reaction conditions utilize standard solvents like tetrahydrofuran and dimethylformamide, which are widely accessible and cost-effective. This accessibility translates directly into a more stable supply chain and reduced raw material costs, making the final polyimide products more competitive in the global market.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive transition metal catalysts in the initial imide formation step, relying instead on base-mediated acylation. This simplification removes the costly and time-consuming heavy metal removal processes typically required in pharmaceutical and electronic grade syntheses. Additionally, the high yields reported in the patent examples indicate efficient atom economy, reducing waste disposal costs and maximizing the output per batch of raw materials invested.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for the substitution of various aromatic groups without altering the core process flow. This flexibility means that if a specific substituent becomes unavailable, alternative groups with similar steric properties can be utilized without revalidating the entire manufacturing process. Such adaptability ensures continuous production capability even when facing fluctuations in the availability of specific fine chemical intermediates.
- Scalability and Environmental Compliance: The process operates at moderate temperatures and pressures, avoiding the extreme conditions that often necessitate specialized high-pressure reactors. This makes the technology highly scalable from kilogram to tonnage production using standard stainless steel equipment. Moreover, the use of hydrazine hydrate for reduction, while requiring careful handling, avoids the generation of heavy metal waste associated with other reduction methods, aligning better with increasingly strict environmental regulations in chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this technology. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and application potential of these imide-structured diamines for potential partners and licensees.
Q: What are the key advantages of these imide-structured diamines over traditional monomers?
A: These compounds feature a rigid non-planar twisted structure that prevents tight chain packing, significantly enhancing solubility in organic solvents while maintaining high thermal stability and unique fluorescence properties.
Q: Can these monomers be used for OLED applications?
A: Yes, specific derivatives exhibit aggregation-induced emission (AIE) effects and dual-wavelength fluorescence, making them highly suitable for photoelectric materials and OLED device fabrication.
Q: Is the synthesis process scalable for industrial production?
A: The patented method utilizes standard organic synthesis techniques such as acylation and Suzuki coupling with readily available reagents, ensuring straightforward purification and high yields suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Diamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance monomers play in the advancement of electronic and polymer industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of aromatic diamine meets the exacting standards required for semiconductor and display applications.
We invite you to collaborate with us to leverage this patented technology for your specific product lines. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in complex organic synthesis can drive innovation and efficiency in your supply chain.
