Scalable Synthesis of Chiral Benzocyclobutane Amines for High-Purity Ivabradine Manufacturing
Scalable Synthesis of Chiral Benzocyclobutane Amines for High-Purity Ivabradine Manufacturing
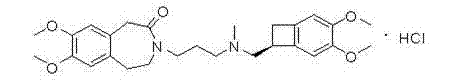
The pharmaceutical industry continuously seeks robust and safe manufacturing routes for critical cardiovascular therapeutics, with Ivabradine Hydrochloride standing out as a pivotal treatment for chronic stable angina. As detailed in patent CN102372642A, a significant technological breakthrough has been achieved in the preparation of its key chiral building block, (1S)-4,5-dimethoxy-1-[(methylamino)methyl]benzocyclobutane. This specific intermediate is essential for constructing the complex bicyclic core of the final active pharmaceutical ingredient, ensuring the necessary stereochemical purity required for biological efficacy. The structural complexity of Ivabradine, as illustrated in the chemical diagram below, demands precise synthetic control to avoid racemization and ensure high optical purity throughout the production chain.
This patent introduces a novel methodology that fundamentally shifts the paradigm from hazardous, low-yield historical processes to a streamlined, industrially viable protocol. By leveraging orthoformate chemistry coupled with selective borohydride reduction, the inventors have addressed long-standing bottlenecks regarding safety and scalability. For R&D directors and procurement specialists alike, understanding this shift is critical, as it represents a move away from exotic, dangerous reagents toward commodity chemicals that are readily available in the global supply chain. The ability to produce this high-value intermediate with improved safety profiles and simplified work-up procedures directly translates to enhanced supply chain reliability and reduced operational risk for downstream API manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this chiral benzocyclobutane amine has been plagued by severe inefficiencies and safety concerns that hindered large-scale commercial adoption. Early approaches, such as those described in European patent EP0534859, relied on the resolution of racemic mixtures using camphorsulfonic acid, a process notoriously inefficient with reported yields as abysmal as 2% to 3%. Such low throughput is economically unsustainable for modern pharmaceutical manufacturing, leading to excessive waste generation and inflated raw material costs. Furthermore, alternative routes documented in Chinese patent CN1699331A utilized Lithium Aluminum Hydride (LiAlH4) as the reducing agent, introducing extreme safety hazards due to its pyrophoric nature and violent reactivity with moisture.
The operational difficulties associated with these legacy methods extend beyond mere safety; they impose significant engineering constraints on reactor design and process control. The vigorous reflux required for LiAlH4 reductions creates challenges in heat dissipation and poses a high risk of "flushing" or runaway reactions, limiting the feasible batch size to small laboratory-scale reactors. Additionally, the quenching of aluminum salts generates substantial volumes of hazardous solid waste, complicating environmental compliance and increasing disposal costs. These factors collectively render conventional methods unsuitable for the reliable, multi-ton production volumes required by the global market for cardiovascular medications, necessitating a safer and more efficient alternative.
The Novel Approach
In stark contrast to these hazardous precedents, the method disclosed in CN102372642A employs a mild and highly controllable two-step sequence that utilizes orthoformates and borohydrides. This innovative route begins with the condensation of the starting amine with an orthoformate reagent, such as trimethyl or triethyl orthoformate, to form a stable alkoxy methyleneimine intermediate. This activation step is crucial as it protects the amine functionality and sets the stage for a highly selective reduction, avoiding the side reactions and racemization issues common in direct alkylation or harsh reduction protocols. The subsequent reduction utilizes sodium borohydride or potassium borohydride, reagents that are significantly safer, cheaper, and easier to handle than lithium aluminum hydride.
The practical advantages of this new approach are manifold, offering a pathway that is inherently safer and more amenable to industrial scale-up. The reaction conditions are mild, typically proceeding in alcohol solvents like ethanol or isopropanol at temperatures that do not require extreme cooling or heating beyond standard reflux. Moreover, the process allows for a telescoped operation where the crude intermediate oil obtained from the first step can be directly charged into the reduction vessel without purification. This elimination of isolation steps not only reduces solvent consumption and processing time but also minimizes product loss, thereby significantly boosting the overall process mass intensity and economic viability of the synthesis.
Mechanistic Insights into Orthoformate Condensation and Borohydride Reduction
The core chemical innovation lies in the strategic use of orthoformate reagents to generate an imine-like intermediate, which serves as a superior substrate for subsequent hydride reduction. In the first stage, the primary amine group of the (1S)-4,5-dimethoxy-1-(aminomethyl)benzocyclobutane reacts with the orthoformate to eliminate an alcohol molecule and form the corresponding N-alkoxymethylimine. This transformation effectively masks the nucleophilicity of the nitrogen atom, preventing unwanted polymerization or over-alkylation during the reaction. The stability of this intermediate allows for the removal of excess orthoformate and generated alcohol under reduced pressure, yielding a brown oil that is sufficiently pure for the next stage without requiring chromatographic purification or crystallization.

The second stage involves the stereoselective reduction of the imine double bond using a borohydride source in an alcoholic medium. Unlike the aggressive and non-selective nature of aluminum hydrides, borohydrides offer a more controlled delivery of hydride ions, which attack the electrophilic carbon of the imine bond. The presence of the alcohol solvent facilitates proton transfer, completing the reduction to the secondary amine while maintaining the integrity of the chiral center at the benzocyclobutane ring. This mechanism ensures that the optical purity established in the starting material is preserved throughout the synthesis, a critical quality attribute for the final API. The mildness of this reduction also means that sensitive functional groups elsewhere in the molecule remain untouched, resulting in a cleaner impurity profile and simplifying downstream purification efforts.
How to Synthesize (1S)-4,5-dimethoxy-1-[(methylamino)methyl]benzocyclobutane Efficiently
Implementing this synthesis requires careful attention to stoichiometry and temperature control to maximize yield and safety. The process begins by charging the chiral aminomethyl benzocyclobutane starting material into a reactor with a slight excess of orthoformate reagent, typically in a molar ratio ranging from 1:1.5 to 1:10, with a preferred range of 1:4 to 1:7 for optimal conversion. The mixture is heated to reflux to drive the condensation reaction to completion, monitored by TLC until the starting amine is fully consumed. Upon completion, the volatile components are stripped under vacuum to afford the crude imine intermediate, which is immediately redissolved in an anhydrous alcohol solvent such as ethanol or isopropanol for the reduction step.
- Condense (1S)-4,5-dimethoxy-1-(aminomethyl)benzocyclobutane with trimethyl or triethyl orthoformate under reflux to form the alkoxy methyleneimine intermediate.
- Cool the reaction mixture to below 0°C in an alcohol solvent and add sodium borohydride or potassium borohydride in portions.
- Warm to room temperature and then reflux for 3 to 8 hours to complete the reduction, followed by solvent removal and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented methodology offers substantial strategic benefits centered around cost reduction and risk mitigation. By replacing hazardous and expensive reagents like Lithium Aluminum Hydride with commodity borohydrides and orthoformates, the direct material costs of the synthesis are drastically lowered. These reagents are produced on a massive global scale, ensuring consistent availability and shielding the supply chain from the volatility often associated with specialized fine chemical suppliers. Furthermore, the elimination of complex resolution steps and the ability to telescope the reaction sequence reduce the number of unit operations, leading to shorter cycle times and higher throughput in existing manufacturing facilities.
- Cost Reduction in Manufacturing: The transition to safer, commodity-grade reagents eliminates the need for specialized handling equipment and expensive safety infrastructure required for pyrophoric materials. The high yields demonstrated in the patent examples, reaching up to 85% in optimized conditions, mean that less raw material is wasted per kilogram of product, directly improving the cost of goods sold. Additionally, the simplified work-up procedure reduces solvent usage and waste disposal fees, contributing to a leaner and more cost-effective manufacturing process that enhances overall profit margins.
- Enhanced Supply Chain Reliability: Relying on widely available chemicals like sodium borohydride and ethanol mitigates the risk of supply disruptions that can occur with niche reagents. The robustness of the reaction conditions allows for production in a wider range of manufacturing sites, including those with standard chemical processing capabilities rather than specialized high-hazard zones. This flexibility enables diversification of the supplier base and ensures business continuity, which is critical for meeting the rigorous delivery schedules of major pharmaceutical clients.
- Scalability and Environmental Compliance: The mild reaction profile and absence of heavy metal catalysts or toxic aluminum waste streams simplify the environmental permitting process for scale-up. The process generates significantly less hazardous waste compared to traditional methods, aligning with modern green chemistry principles and reducing the regulatory burden on the manufacturer. This environmental compatibility facilitates smoother technology transfer from pilot plant to commercial scale, accelerating the time to market for the final drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific technical disclosures and comparative data found within the patent documentation, providing clarity on the operational benefits and chemical rationale behind the new method. Understanding these details is essential for technical teams evaluating the feasibility of adopting this process for their own supply chains.
Q: Why is the borohydride reduction method preferred over Lithium Aluminum Hydride for this intermediate?
A: The borohydride method eliminates the severe safety hazards associated with Lithium Aluminum Hydride, such as pyrophoric risks and violent exotherms during quenching, while offering milder reaction conditions that are easier to control on a large industrial scale.
Q: What is the typical yield range for this novel synthetic route?
A: Experimental data from the patent indicates yields ranging significantly higher than previous resolution methods, with optimized examples demonstrating yields up to 85%, compared to the extremely low 2-3% yields of older resolution-based processes.
Q: Can the intermediate crude oil be used directly in the next step?
A: Yes, the process allows for a telescoped operation where the brown oil obtained after removing the orthoformate solvent can be directly subjected to the reduction step without intermediate purification, streamlining the workflow.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (1S)-4,5-dimethoxy-1-[(methylamino)methyl]benzocyclobutane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful manufacture of life-saving cardiovascular medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering this key Ivabradine intermediate with stringent purity specifications, utilizing rigorous QC labs to verify every batch against the highest international standards. Our state-of-the-art facilities are equipped to handle the specific requirements of chiral amine synthesis, guaranteeing consistency and reliability for your long-term projects.
We invite you to engage with our technical procurement team to discuss how this advanced synthetic route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this safer, more efficient methodology. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your unique production needs, ensuring a partnership built on transparency, technical excellence, and mutual success.
