Advanced Synthesis of Ivabradine Key Intermediate for Commercial Scale Production
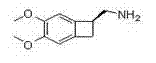
The pharmaceutical landscape for cardiovascular therapeutics has been significantly shaped by the introduction of Ivabradine Hydrochloride, a selective inhibitor of the cardiac pacemaker current. Central to the manufacturing of this vital medication is the chiral building block known as (1S)-4,5-dimethoxy-1-[(methylamino)methyl]benzocyclobutane. Recent intellectual property developments, specifically patent CN102372642B, have unveiled a transformative preparation method that addresses long-standing inefficiencies in the supply chain of this critical intermediate. This innovation represents a paradigm shift from hazardous, low-yielding legacy processes to a streamlined, industrially viable protocol that leverages orthoformate condensation followed by mild borohydride reduction. For global procurement and technical teams, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediates supplier capable of delivering consistent quality.
![Chemical structure of the target intermediate (1S)-4,5-dimethoxy-1-[(methylamino)methyl]benzocyclobutane](/insights/img/ivabradine-intermediate-synthesis-pharma-supplier-20260305014752-04.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this benzocyclobutane derivative has been plagued by severe safety hazards and economic inefficiencies that hindered commercial scale-up of complex pharmaceutical intermediates. Early methodologies, such as those described in European Patent EP0534859, relied on the resolution of racemic mixtures using camphorsulfonic acid, resulting in abysmal overall yields of merely 2-3%, which is economically unsustainable for high-volume API manufacturing. Furthermore, alternative routes documented in Chinese Patent CN1699331A necessitated the use of Lithium Aluminum Hydride (LiAlH4) as a reducing agent. This reagent is notoriously pyrophoric, posing extreme fire risks upon exposure to moisture and requiring stringent anhydrous conditions that complicate reactor operations and increase capital expenditure for safety infrastructure. These legacy processes often suffered from violent reflux conditions that were difficult to control, limiting production to small-scale reactors and creating significant bottlenecks in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to these perilous and inefficient antecedents, the methodology disclosed in CN102372642B introduces a robust two-step sequence that prioritizes operator safety and process scalability without compromising stereochemical integrity. The novel approach circumvents the need for dangerous hydride reagents by employing a condensation reaction with orthoformate esters to generate a stable imine intermediate, which is subsequently reduced using sodium borohydride or potassium borohydride. This strategic modification eliminates the risk of spontaneous combustion associated with aluminum hydrides and allows the reaction to proceed under much milder thermal conditions. By utilizing readily available and cost-effective reagents such as trimethyl orthoformate and ethanol, the new process drastically simplifies the operational workflow, thereby facilitating cost reduction in API manufacturing while ensuring that the chiral center remains intact throughout the transformation.
Mechanistic Insights into Orthoformate Condensation and Borohydride Reduction
The core chemical innovation lies in the formation of an alkoxy-methylene-imine intermediate, which serves as a protected and activated species for the subsequent reduction step. In the first stage, the primary amine group of the starting material reacts with the orthoformate reagent under reflux conditions to eliminate alcohol and form the imine bond. This step is crucial as it prevents potential side reactions and prepares the nitrogen atom for selective methylation during the reduction phase. The molar ratio of the starting amine to the orthoformate is carefully optimized, typically ranging from 1:1.5 to 1:10, with a preferred range of 1:4 to 1:7 to ensure complete conversion without excessive waste. This condensation creates a transient species that is highly susceptible to nucleophilic attack by the hydride source in the following step, setting the stage for a clean and efficient transformation.
Following the in-situ formation of the imine, the reaction mixture is cooled to below 0°C before the gradual addition of the borohydride reducing agent. This temperature control is vital for managing the exothermic nature of the reduction and preventing the decomposition of the intermediate. Once the borohydride is added, the system is allowed to warm to room temperature and then heated to reflux, driving the reduction to completion over a period of 3 to 8 hours. The use of alcohol solvents such as methanol, ethanol, or isopropanol provides an ideal medium for both the solubility of the intermediates and the stability of the borohydride species. This mechanistic pathway effectively suppresses racemization, a common pitfall in amine synthesis, ensuring that the final product retains the critical (1S) configuration required for biological activity in the final Ivabradine drug substance.
How to Synthesize (1S)-4,5-dimethoxy-1-[(methylamino)methyl]benzocyclobutane Efficiently
Implementing this synthesis route requires precise adherence to the reaction parameters outlined in the patent to maximize yield and purity. The process begins with the reflux of the chiral aminomethyl benzocyclobutane with an excess of orthoformate ester, followed by the removal of volatiles to isolate the crude imine oil. This crude material is then subjected to reduction in an alcoholic solvent with careful temperature modulation. The detailed standardized synthesis steps, including specific workup procedures and purification techniques to achieve pharmaceutical grade quality, are provided in the guide below.
- React (1S)-4,5-dimethoxy-1-(aminomethyl)benzocyclobutane with trimethyl orthoformate under reflux to form the imine intermediate.
- Cool the reaction mixture to below 0°C and add sodium borohydride portions in an alcohol solvent.
- Warm to room temperature and reflux to complete the reduction, followed by standard workup to isolate the target amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers profound strategic benefits that extend beyond mere technical feasibility. The elimination of Lithium Aluminum Hydride removes a major safety liability, significantly lowering insurance premiums and reducing the need for specialized hazardous material handling protocols. Furthermore, the substantial improvement in reaction yield, moving from single-digit percentages in older methods to over 80% in this new process, directly translates to a drastic reduction in raw material consumption per kilogram of finished product. This efficiency gain is a primary driver for cost reduction in API manufacturing, allowing for more competitive pricing structures in the final drug market. Additionally, the use of common solvents like ethanol and dichloromethane simplifies waste stream management and solvent recovery, further enhancing the environmental profile and operational sustainability of the production facility.
- Cost Reduction in Manufacturing: The transition from expensive and dangerous resolving agents and reducing chemicals to commodity reagents like orthoformates and sodium borohydride results in substantial cost savings. By avoiding the complex resolution steps that discarded up to 98% of the material in previous methods, the new process maximizes atom economy and minimizes the cost of goods sold. The simplified workflow also reduces labor hours and energy consumption associated with maintaining extreme anhydrous conditions, contributing to a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: Reliance on widely available industrial chemicals ensures that production schedules are not disrupted by the scarcity of specialized reagents. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, leading to more consistent batch-to-batch performance. This reliability is critical for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream API synthesis can proceed without interruption and meeting the rigorous demands of global regulatory bodies.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of pyrophoric reagents make this process inherently safer for scaling from pilot plant to multi-ton commercial production. The ability to run reactions at atmospheric pressure without specialized high-pressure equipment lowers capital investment barriers. Moreover, the cleaner reaction profile generates less hazardous waste, facilitating easier compliance with increasingly stringent environmental regulations and supporting the company's sustainability goals through greener chemistry practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on safety, yield, and scalability concerns.
Q: Why is the new orthoformate method safer than previous Lithium Aluminum Hydride routes?
A: Previous methods utilized Lithium Aluminum Hydride (LiAlH4), which poses severe fire hazards upon contact with moisture and requires rigorous anhydrous conditions. The new method employs Sodium Borohydride, a significantly safer and more manageable reducing agent suitable for large-scale operations.
Q: What yield improvements does patent CN102372642B offer over European Patent EP0534859?
A: While the older European patent reported extremely low yields of only 2-3% due to inefficient resolution steps, the new process achieves yields up to 85% by avoiding racemization and utilizing a direct reductive amination strategy.
Q: Is this synthesis route scalable for industrial API production?
A: Yes, the process is specifically designed for industrial amplification. It uses common solvents like ethanol and reagents like orthoformates, eliminating the need for specialized high-pressure equipment or dangerous pyrophoric reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (1S)-4,5-dimethoxy-1-[(methylamino)methyl]benzocyclobutane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving cardiovascular medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of (1S)-4,5-dimethoxy-1-[(methylamino)methyl]benzocyclobutane meets the exacting standards required for GMP API synthesis. Our facility is equipped to handle the specific solvent systems and reaction conditions outlined in CN102372642B, providing a secure and compliant source for your supply chain.
We invite you to collaborate with us to optimize your sourcing strategy for this vital intermediate. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this advanced synthesis method can enhance your project's timeline and budget.
