Advancing Fluorinated Pharmaceutical Intermediates via Nickel-Catalyzed Migration Defluorination Allylation
The landscape of modern medicinal chemistry is increasingly defined by the strategic incorporation of fluorine atoms into drug candidates, where gem-difluoroolefins serve as exceptional carbonyl bioisosteres due to their enhanced metabolic stability and electronic properties. Patent CN115850111A introduces a groundbreaking preparation method for nickel-catalyzed aliphatic amines containing gem-difluoroolefin structures, addressing critical challenges in the synthesis of these valuable motifs. This technology leverages a migration defluorination allylation reaction that operates under remarkably mild conditions, utilizing ethylene glycol dimethyl ether nickel bromide as a cost-effective catalyst system. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, this patent represents a significant leap forward in accessing complex fluorinated scaffolds that were previously difficult or expensive to manufacture. The ability to synthesize these structures efficiently opens new avenues for drug discovery programs focused on enhancing the pharmacokinetic profiles of lead compounds through precise fluorine placement.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of gem-difluoroolefin moieties has relied heavily on methods that present substantial operational and economic hurdles for large-scale manufacturing. Traditional cross-coupling reactions often necessitate the use of pre-prepared organometallic reagents, which are not only sensitive to air and moisture but also exhibit poor tolerance towards diverse functional groups commonly found in advanced intermediates. Furthermore, many existing protocols depend on expensive noble metal catalysts such as palladium or rhodium, which drastically inflate the cost of goods sold and complicate the removal of trace metal impurities to meet stringent regulatory standards for active pharmaceutical ingredients. The requirement for harsh reaction conditions or specialized directing groups that need subsequent installation and removal steps further diminishes the atom economy and overall efficiency of these legacy processes. Consequently, the pharmaceutical industry has long sought a more robust, economical, and versatile synthetic route that can accommodate a broad range of substrates without compromising on yield or purity.
The Novel Approach
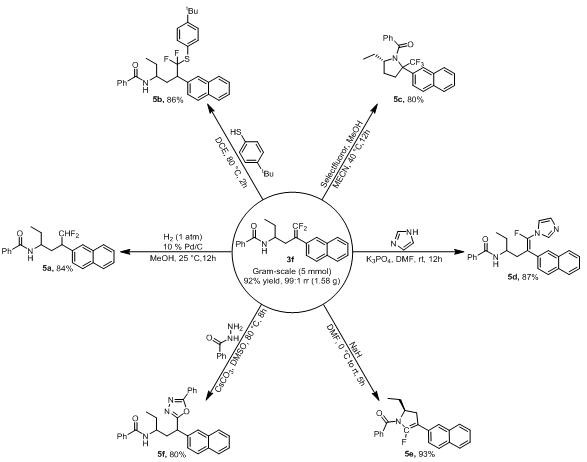
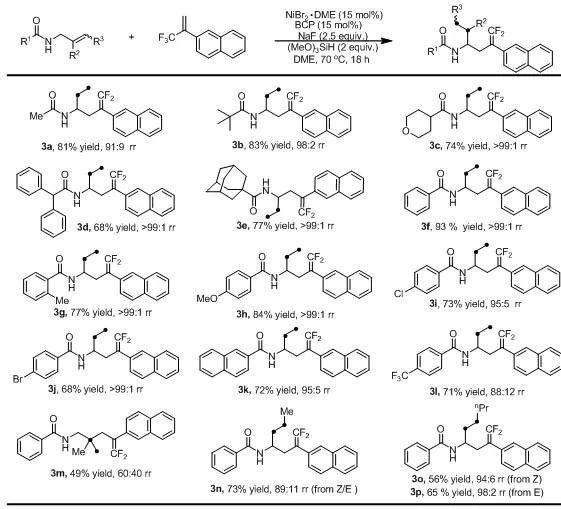
The methodology disclosed in CN115850111A fundamentally shifts the paradigm by employing a nickel-hydride catalytic system that enables the direct transformation of unactivated olefins and trifluoromethyl-substituted olefins. This novel approach eliminates the need for pre-functionalized organometallic nucleophiles, thereby simplifying the synthetic workflow and reducing the generation of hazardous waste. By utilizing cheap and readily available nickel salts combined with bathocuproine ligands, the process achieves high regioselectivity and impressive isolated yields, reaching up to 93 percent in optimized examples. The reaction proceeds smoothly at a moderate temperature of 70°C in ethylene glycol dimethyl ether, demonstrating exceptional compatibility with various functional groups including esters, nitriles, and halides. This advancement signifies a major step towards cost reduction in pharmaceutical intermediates manufacturing, offering a streamlined pathway that aligns perfectly with the principles of green chemistry and sustainable production practices demanded by modern supply chains.
Mechanistic Insights into Ni-Catalyzed Migration Defluorination Allylation
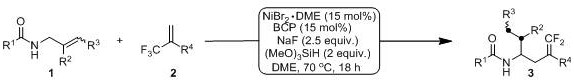
The core innovation of this technology lies in the intricate mechanistic pathway involving nickel-hydride species that facilitate remote C-H bond functionalization through a chain-walking mechanism. Initially, the nickel catalyst generates a reactive Ni-H species in situ using trimethoxysilane as a hydrogen source, which then inserts into the unactivated olefin substrate. Through a series of reversible beta-hydride elimination and re-insertion steps, the metal center migrates along the carbon chain until it reaches the thermodynamically favored position directed by the amide group. This precise positioning allows for the subsequent activation of the trifluoromethyl-substituted olefin, triggering a defluorinative cross-coupling event. The use of sodium fluoride as a base plays a crucial role in facilitating the beta-fluorine elimination step, which is the key transformation that converts the trifluoromethyl group into the desired gem-difluoroolefin motif. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters for commercial scale-up of complex fluorinated intermediates, as it highlights the importance of ligand selection and base stoichiometry in controlling the selectivity and efficiency of the catalytic cycle.
The exceptional functional group tolerance observed in this system can be attributed to the mild nature of the nickel-hydride species and the specific electronic environment created by the bathocuproine ligand. Unlike more aggressive transition metal catalysts, this system does not promote unwanted side reactions such as homocoupling or over-reduction of sensitive moieties like aldehydes or ketones. The amide directing group not only guides the regioselectivity but also stabilizes the intermediate complexes, ensuring that the reaction proceeds cleanly to the desired product. This level of control is particularly beneficial for the synthesis of high-purity gem-difluoroolefin compounds intended for biological evaluation, where even minor impurities can skew assay results. For supply chain heads concerned with reducing lead time for high-purity fluorinated intermediates, this robustness translates to fewer purification steps and higher overall throughput, enabling faster delivery of critical building blocks to drug discovery teams.
How to Synthesize Gem-Difluoroolefin Compounds Efficiently
The practical implementation of this synthesis route is designed to be accessible for both laboratory-scale optimization and industrial production environments. The protocol involves combining the nickel catalyst, ligand, base, silane, and olefin substrates in a dry reaction vessel under an inert argon atmosphere to prevent catalyst deactivation. Following the reaction period, standard workup procedures involving vacuum concentration and silica gel chromatography allow for the isolation of the target molecules with high purity. Detailed standardized synthesis steps see the guide below.
- In an argon-filled glove box, combine NiBr2•DME catalyst, bathocuproine ligand, unactivated olefin substrate, trifluoromethyl-substituted olefin, sodium fluoride base, and trimethoxysilane in dry DME solvent.
- Stir the reaction mixture at 70°C for 18 hours to facilitate the migration defluorination allylation process.
- Upon completion, concentrate the solution in vacuo and purify the crude product via silica gel column chromatography using ethyl acetate and n-hexane.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this nickel-catalyzed methodology offers profound benefits for procurement managers and supply chain leaders tasked with optimizing the cost and reliability of raw material sourcing. The shift from precious metal catalysts to base metal nickel represents a substantial cost savings opportunity, as nickel salts are orders of magnitude cheaper than palladium or rhodium counterparts. Additionally, the elimination of pre-formed organometallic reagents reduces the complexity of the supply chain, as fewer specialized starting materials need to be sourced and stored under strict conditions. The mild reaction conditions also contribute to lower energy consumption during the manufacturing process, further enhancing the overall economic viability of producing these high-value intermediates. For organizations seeking a reliable pharmaceutical intermediates supplier, partnering with a manufacturer capable of executing this technology ensures a stable supply of critical fluorinated building blocks without the volatility associated with precious metal markets.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts with inexpensive nickel salts drastically lowers the direct material costs associated with catalysis. Furthermore, the simplified operational procedure reduces labor hours and equipment usage time, leading to significant overhead reductions. The high atom economy of the reaction minimizes waste disposal costs, contributing to a leaner and more profitable manufacturing model that supports competitive pricing strategies for downstream clients.
- Enhanced Supply Chain Reliability: By utilizing commercially available and stable reagents such as sodium fluoride and trimethoxysilane, the risk of supply disruptions is significantly mitigated. The robustness of the reaction against moisture and air, relative to traditional organometallic methods, allows for more flexible manufacturing schedules and reduces the need for specialized containment infrastructure. This reliability ensures consistent delivery timelines, which is crucial for maintaining the momentum of clinical development programs and meeting commercial launch deadlines.
- Scalability and Environmental Compliance: The protocol has been demonstrated to be scalable to gram-level production with maintained efficiency, indicating strong potential for multi-kilogram or ton-scale operations. The use of less hazardous solvents and the generation of simpler waste streams align with increasingly stringent environmental regulations, reducing the compliance burden on manufacturing facilities. This sustainability profile enhances the long-term viability of the supply chain and appeals to environmentally conscious stakeholders within the pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this nickel-catalyzed synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity for potential partners and collaborators.
Q: What are the advantages of using nickel catalysis over traditional noble metals for gem-difluoroolefin synthesis?
A: Nickel catalysis offers a significantly more cost-effective alternative to precious metals like palladium or rhodium. The patent demonstrates that nickel systems can achieve high regioselectivity and yield under mild conditions (70°C), reducing energy consumption and catalyst costs while maintaining excellent functional group tolerance.
Q: How does this method improve regioselectivity compared to conventional cross-coupling reactions?
A: The method utilizes an amide directing group which effectively controls the position of the nickel-hydride species during the chain-walking process. This ensures precise migration defluorination allylation, avoiding the poor regioselectivity often seen in traditional organometallic nucleophile approaches.
Q: Is this synthesis protocol scalable for commercial production of fluorinated intermediates?
A: Yes, the patent explicitly mentions scalability to gram-scale production. The use of commercially available reagents, mild reaction temperatures, and standard purification techniques like silica gel chromatography indicates strong potential for commercial scale-up of complex fluorinated intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gem-Difluoroolefin Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting innovative synthetic technologies to deliver high-value chemical solutions to the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries like the nickel-catalyzed gem-difluoroolefin synthesis can be successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediates meets the exacting standards required for drug substance manufacturing. Our commitment to technical excellence allows us to navigate the complexities of fluorine chemistry with precision, providing our clients with a secure and dependable source of critical building blocks.
We invite you to engage with our technical procurement team to discuss how this advanced methodology can be integrated into your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this nickel-catalyzed route for your fluorinated intermediates. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your target molecules, ensuring that your supply chain is optimized for both performance and profitability in an increasingly competitive landscape.