Advanced Metal-Free Synthesis of Gem-Difluoroolefins for Scalable Pharmaceutical Manufacturing
Advanced Metal-Free Synthesis of Gem-Difluoroolefins for Scalable Pharmaceutical Manufacturing
The rapid evolution of modern medicinal chemistry has placed gem-difluoroolefins at the forefront of bioisostere design, owing to their unique ability to mimic carbonyl groups while enhancing metabolic stability and lipophilicity in drug candidates. A groundbreaking development in this sector is detailed in patent CN114409515A, which discloses a highly efficient preparation method for gem-difluoroolefin compounds via a novel hydrodefluorination strategy. This technology represents a significant paradigm shift from classical carbonyl-based syntheses, offering a robust pathway that utilizes trifluoromethyl olefin precursors under remarkably mild, metal-free conditions. For R&D directors and procurement specialists alike, this innovation promises not only superior chemical purity but also a streamlined supply chain for complex fluorinated intermediates essential in the development of next-generation active pharmaceutical ingredients.
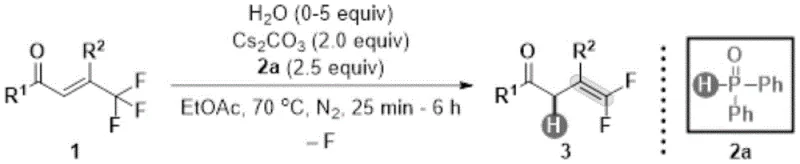
Traditionally, the construction of gem-difluoroolefin motifs has relied heavily on classical olefination protocols such as the Wittig or Horner-Wadsworth-Emmons reactions, which often necessitate harsh strongly basic environments and exhibit narrow substrate scopes. These conventional methods frequently struggle with sensitive functional groups, leading to decomposition or side reactions that compromise the overall yield and purity of the final API intermediate. Furthermore, the requirement for stoichiometric phosphorus ylides or specialized phosphonates can introduce significant cost burdens and waste disposal challenges in large-scale manufacturing settings. The limitations of these legacy technologies have long hindered the rapid exploration of fluorinated chemical space, creating a bottleneck for pharmaceutical companies seeking to optimize the pharmacokinetic profiles of their lead compounds through fluorine incorporation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The reliance on strong bases in traditional synthetic routes poses severe compatibility issues with electrophilic functional groups commonly found in advanced drug intermediates, such as esters, nitriles, or halogens. When attempting to synthesize complex molecules, chemists often face low regioselectivity and poor yields, forcing them to engage in tedious protection-deprotection sequences that inflate production timelines and costs. Additionally, the generation of stoichiometric amounts of phosphine oxide byproducts in Wittig-type reactions creates significant downstream purification hurdles, requiring extensive chromatographic separation that is difficult to translate from benchtop to kilogram-scale production. These inefficiencies underscore the critical industry need for a more tolerant and operationally simple methodology that can deliver high-purity fluorinated building blocks without compromising process safety or environmental compliance standards.
The Novel Approach
In stark contrast to these archaic methods, the technology described in patent CN114409515A introduces a transformative hydrodefluorination protocol that converts readily available trifluoromethyl olefins into valuable gem-difluoroolefins with exceptional efficiency. As illustrated in the structural transformation below, this method employs diphenylphosphine oxide as a benign reducing agent and cesium carbonate as a mild base, operating effectively in ethyl acetate at a moderate temperature of 70°C. This approach completely bypasses the need for cryogenic conditions or pyrophoric reagents, thereby enhancing operational safety and reducing energy consumption. The broad substrate tolerance allows for the successful conversion of diverse aryl and heteroaryl-substituted precursors, making it an ideal candidate for the late-stage functionalization of complex molecular scaffolds in drug discovery pipelines.
Mechanistic Insights into Diphenylphosphine Oxide Promoted Hydrodefluorination
The mechanistic elegance of this transformation lies in the synergistic interaction between the phosphine oxide reductant and the cesium carbonate base, which facilitates the selective cleavage of the carbon-fluorine bond without affecting other sensitive moieties. The reaction proceeds through a concerted pathway where the phosphine oxide serves as a hydrogen source, enabling the defluorinative reduction of the trifluoromethyl group to a gem-difluoromethylene unit. Crucially, the inclusion of water as a reaction promoter plays a pivotal role in activating the catalytic cycle; experimental optimization revealed that adding 5.0 equivalents of water boosts the yield to an impressive 87%, compared to only 62% in its absence. This subtle yet powerful modulation of the reaction environment highlights the sophistication of the process design, ensuring high conversion rates while maintaining excellent regioselectivity for the desired E-isomer products.
From an impurity control perspective, the metal-free nature of this chemistry is a distinct advantage for pharmaceutical manufacturing, as it eliminates the risk of heavy metal contamination that plagues transition-metal catalyzed cross-couplings. The use of cesium carbonate ensures a homogeneous reaction medium that minimizes the formation of polymeric byproducts or elimination side-reactions often seen with stronger alkali bases. Furthermore, the choice of ethyl acetate as the solvent not only provides excellent solubility for both organic substrates and inorganic bases but also aligns with green chemistry principles by avoiding chlorinated or high-boiling polar aprotic solvents. This combination of factors results in a crude product profile that is significantly cleaner, thereby simplifying the final purification steps and ensuring that the resulting high-purity pharmaceutical intermediates meet stringent regulatory specifications for residual impurities.
How to Synthesize Gem-Difluoroolefin Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric ratios and environmental controls to maximize the economic and chemical potential of the process. The standard protocol involves charging a reactor with the trifluoromethyl olefin substrate, diphenylphosphine oxide (2.5 equivalents), and cesium carbonate (2.0 equivalents) under an inert nitrogen atmosphere to prevent oxidation of the phosphine species. The detailed standardized synthesis steps, including specific workup procedures and purification parameters, are outlined in the technical guide below to ensure reproducibility and safety during scale-up operations.
- Combine trifluoromethyl olefin substrate with diphenylphosphine oxide (2.5 equiv) and cesium carbonate (2.0 equiv) in ethyl acetate solvent.
- Add water (5.0 equiv) as a reaction promoter and stir the mixture under nitrogen atmosphere at 70°C for 3 hours.
- Quench with saturated ammonium chloride, extract with ethyl acetate, dry over sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this hydrodefluorination technology offers compelling strategic advantages that directly impact the bottom line and operational resilience of chemical sourcing. By shifting away from precious metal catalysts and hazardous reagents, manufacturers can achieve substantial cost savings in raw material acquisition and waste management, while simultaneously mitigating the risks associated with supply chain disruptions for critical catalytic metals. The robustness of the reaction conditions allows for flexible production scheduling and easier technology transfer between sites, ensuring a continuous and reliable supply of high-value fluorinated intermediates even in volatile market conditions.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for expensive metal scavenging resins and rigorous testing for heavy metal residues, which are significant cost drivers in API manufacturing. Furthermore, the use of commodity chemicals like cesium carbonate and ethyl acetate, combined with the high atom economy of the hydrodefluorination step, leads to a drastically simplified bill of materials. This streamlined process architecture reduces the overall cost of goods sold (COGS) by minimizing solvent usage and shortening cycle times, allowing for more competitive pricing strategies in the global pharmaceutical intermediate market.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents such as diphenylphosphine oxide ensures that production is not vulnerable to the logistical complexities often associated with air-sensitive or pyrophoric reagents. The broad functional group tolerance means that a single manufacturing platform can service a wide variety of customer projects without requiring extensive retooling or custom equipment, thereby enhancing the agility of the supply chain. This versatility allows suppliers to respond rapidly to changing demand signals from R&D departments, reducing lead times for high-purity pharmaceutical intermediates and securing long-term partnerships with key stakeholders.
- Scalability and Environmental Compliance: Operating at a moderate temperature of 70°C in a green solvent like ethyl acetate significantly lowers the energy footprint of the process compared to cryogenic or high-temperature alternatives. The absence of toxic heavy metals simplifies wastewater treatment and solid waste disposal, ensuring full compliance with increasingly stringent environmental regulations across major manufacturing hubs. This eco-friendly profile not only reduces regulatory risk but also aligns with the sustainability goals of multinational corporations, making the commercial scale-up of complex fluorinated intermediates both economically viable and environmentally responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this hydrodefluorination methodology, derived directly from the experimental data and beneficial effects reported in the patent literature. Understanding these nuances is critical for process chemists aiming to integrate this technology into their existing workflows for the synthesis of bioactive molecules.
Q: What are the advantages of this hydrodefluorination method over traditional Wittig reactions?
A: Unlike traditional Wittig or Horner-Wadsworth-Emmons reactions that require strong basic conditions and have limited substrate tolerance, this metal-free hydrodefluorination method operates under mild conditions (70°C) with excellent functional group compatibility and regioselectivity.
Q: Is transition metal removal required for this synthesis?
A: No, the process is entirely metal-free, utilizing diphenylphosphine oxide as a reductant and cesium carbonate as a base, which eliminates the need for expensive and time-consuming heavy metal scavenging steps typically required in transition-metal catalyzed processes.
Q: What is the optimal water equivalent for maximizing yield in this reaction?
A: Experimental data indicates that adding 5.0 equivalents of water as a reaction promoter significantly enhances the yield to 87%, whereas omitting water or using excessive amounts (20 equiv) drastically reduces efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gem-Difluoroolefin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free hydrodefluorination technology in accelerating the development of fluorinated drugs and agrochemicals. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from gram-scale discovery to industrial manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of gem-difluoroolefin intermediate meets the highest quality standards required for clinical and commercial applications.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can optimize your specific project requirements and drive down development costs. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your molecule, along with specific COA data and route feasibility assessments. Let us help you secure a sustainable and cost-effective supply chain for your next-generation fluorinated compounds.
