Advanced Manufacturing of Gefarnate: A Safer, High-Yield Synthetic Route for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust manufacturing pathways for gastrointestinal therapeutics, and the synthesis of Gefarnate stands as a prime example of process evolution. As detailed in patent CN102146039A, a novel synthetic strategy has been developed that fundamentally alters the production landscape for this vital anti-ulcer agent. This innovative approach addresses critical historical bottlenecks related to solvent toxicity and purification complexity, offering a streamlined route that transitions from simple terpene precursors to the complex polyunsaturated ester structure required for clinical efficacy. By leveraging a combination of orthoester rearrangement chemistry and a highly controlled Mitsunobu esterification, this methodology delivers a product profile that meets the stringent purity demands of modern regulatory bodies while simultaneously enhancing operational safety. For global procurement teams and R&D directors, understanding the nuances of this specific patent technology is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering consistent quality at scale.
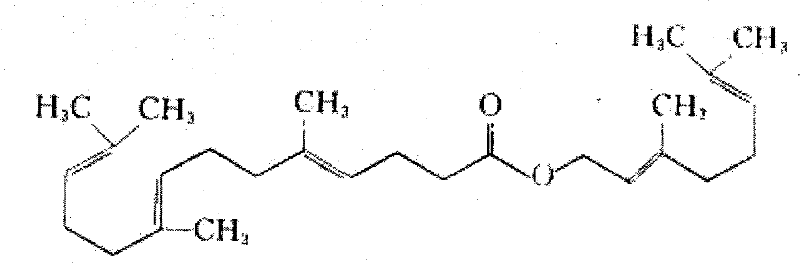
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Gefarnate has been plagued by significant environmental and safety challenges inherent to older synthetic protocols. Previous methodologies, such as those disclosed in patent CN200910000897, relied heavily on the use of methanol and N,N-dimethylformamide (DMF) as primary reaction media. Methanol presents severe volatility and toxicity risks to plant operators, necessitating expensive containment systems and rigorous air monitoring protocols to maintain workplace safety standards. Furthermore, DMF is notoriously difficult to remove completely from the final product due to its high boiling point and strong solvation properties, often leading to residual solvent levels that exceed International Council for Harmonisation (ICH) guidelines. These residual impurities not only compromise the safety profile of the active pharmaceutical ingredient but also require energy-intensive downstream processing steps, such as multiple high-vacuum distillations or complex chromatographic separations, which drastically inflate manufacturing costs and extend production lead times.
The Novel Approach
In stark contrast, the process outlined in CN102146039A introduces a greener and more efficient synthetic architecture that circumvents these legacy issues through strategic reagent selection. The new route initiates with the condensation of triethyl orthoacetate, nerolidol, and isobutyric acid, followed by a saponification step in ethanol, effectively eliminating the need for methanol in the early stages. The final coupling utilizes dichloromethane, which, while requiring careful handling, allows for much easier solvent removal compared to DMF, thereby simplifying the isolation of the crude product. This methodological shift not only reduces the toxicological burden on the manufacturing environment but also enhances the overall yield profile, with the final esterification step achieving yields as high as 90% under optimized conditions. By replacing problematic solvents with more manageable alternatives and optimizing reaction stoichiometry, this approach offers a compelling solution for cost reduction in API manufacturing while ensuring a cleaner impurity profile.
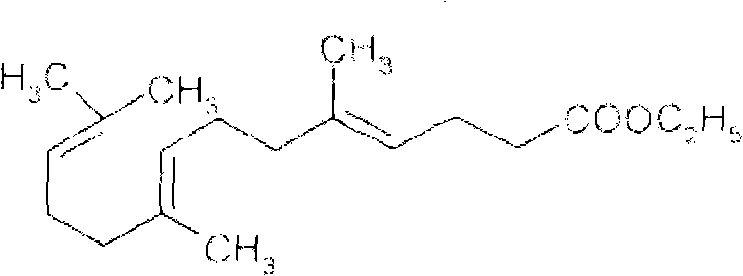
Mechanistic Insights into Orthoester Rearrangement and Mitsunobu Coupling
The core chemical innovation of this process lies in the precise execution of two distinct reaction mechanisms that preserve the delicate stereochemistry of the polyene chain. The initial phase involves an orthoester Claisen rearrangement, where triethyl orthoacetate reacts with nerolidol in the presence of isobutyric acid at elevated temperatures ranging from 120°C to 160°C. This thermal rearrangement constructs the carbon skeleton of the acid moiety while maintaining the geometric integrity of the double bonds, a critical factor for the biological activity of the final molecule. Following this, the intermediate undergoes alkaline hydrolysis using potassium hydroxide in ethanol at 55°C, a mild condition that prevents the isomerization of the sensitive conjugated diene systems. The resulting carboxylic acid (Intermediate 2) is then isolated through a rigorous extraction and acidification protocol, ensuring that inorganic salts and unreacted starting materials are thoroughly removed before the final coupling step.
The culmination of the synthesis is the Mitsunobu reaction, a powerful tool for forming ester bonds under mild conditions, which is particularly advantageous for sterically hindered or sensitive substrates. In this specific application, Intermediate 2 is reacted with geraniol in the presence of triphenylphosphine and diisopropyl azodicarboxylate (DIAD) at a strictly controlled low temperature of -5°C to 0°C. This低温 (low-temperature) regime is paramount; it suppresses side reactions and prevents the thermal degradation or cis-trans isomerization of the multiple double bonds present in both the acid and alcohol components. The reaction proceeds via the formation of a betaine intermediate, which activates the alcohol for nucleophilic attack by the carboxylate anion, resulting in the inversion of configuration at the alcohol center (though geraniol is primary, minimizing steric issues) and the formation of the target ester. The use of DIAD over DEAD (diethyl azodicarboxylate) often provides better solubility and handling characteristics in large-scale operations, further supporting the scalability of this route.
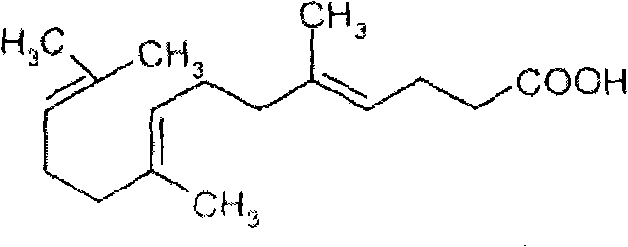
How to Synthesize Gefarnate Efficiently
Implementing this synthesis requires strict adherence to the thermal and stoichiometric parameters defined in the patent to ensure reproducibility and high purity. The process begins with the preparation of the acid fragment, followed by the critical low-temperature coupling with the terpene alcohol. Operators must monitor the reaction progress closely, typically using Thin Layer Chromatography (TLC) with a petroleum ether and isopropyl ether system, to determine the exact endpoint and prevent over-reaction or decomposition. The workup procedure involves multiple washing steps with sodium bicarbonate and water to remove the triphenylphosphine oxide byproduct, which is a common challenge in Mitsunobu chemistry, ensuring the final oil is free from phosphorus contaminants. For a detailed breakdown of the specific reagent quantities, mixing rates, and distillation cuts required for pilot and commercial batches, please refer to the standardized operating procedures below.
- Condense triethyl orthoacetate with nerolidol and isobutyric acid under heating (120-160°C) followed by vacuum distillation to obtain Intermediate 1.
- Hydrolyze Intermediate 1 using potassium hydroxide in ethanol at 55°C, followed by acidification and extraction to isolate the carboxylic acid Intermediate 2.
- Perform a Mitsunobu reaction between Intermediate 2 and geraniol using triphenylphosphine and DIAD at -5°C to 0°C to yield the final Gefarnate product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route translates into tangible strategic advantages beyond mere chemical elegance. The elimination of high-boiling, toxic solvents like DMF directly impacts the bottom line by reducing the energy consumption associated with solvent recovery and waste treatment. Furthermore, the reliance on naturally derived terpene starting materials such as nerolidol and geraniol ensures a stable and sustainable supply chain, as these feedstocks are widely available from the fragrance and flavor industries, mitigating the risk of raw material shortages that often plague synthetic petrochemical-derived intermediates. The robustness of the reaction conditions, particularly the high yields observed in the hydrolysis (93%) and esterification (90%) steps, means that less raw material is wasted per kilogram of finished product, driving significant efficiency gains in material utilization.
- Cost Reduction in Manufacturing: The streamlined purification process significantly lowers operational expenditures by removing the need for complex chromatographic columns or extensive high-vacuum stripping required to purge DMF residues. By utilizing ethanol and dichloromethane, which are cheaper and easier to recycle than specialized polar aprotic solvents, manufacturers can achieve substantial cost savings in utility and waste disposal budgets. Additionally, the high conversion rates minimize the volume of unreacted starting materials that must be recovered or disposed of, further optimizing the cost-per-kilogram metric for this high-value intermediate.
- Enhanced Supply Chain Reliability: The use of commodity chemicals like triethyl orthoacetate and isobutyric acid, combined with widely sourced terpenes, creates a resilient supply network that is less susceptible to geopolitical disruptions or single-source vendor failures. The simplified workflow, which avoids exotic catalysts or sensitive organometallic reagents, allows for production across a broader range of qualified contract manufacturing organizations (CMOs), thereby increasing supply redundancy and reducing lead times for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns better with modern green chemistry principles by reducing the generation of hazardous waste streams. The absence of methanol and DMF simplifies the permitting process for new production lines and reduces the liability associated with volatile organic compound (VOC) emissions. This compliance advantage facilitates faster commercial scale-up of complex terpenoid esters, allowing companies to bring products to market more rapidly without facing regulatory hurdles related to solvent residuals or worker safety exposure limits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Gefarnate synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on how this method outperforms traditional routes in terms of safety, yield, and purity. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this process for their own manufacturing portfolios or for sourcing partners who utilize this specific methodology.
Q: How does this new synthesis process improve upon previous methods regarding solvent toxicity?
A: Unlike prior art methods (e.g., CN200910000897) that utilized highly toxic and volatile methanol and difficult-to-remove DMF, this optimized process employs ethanol and dichloromethane, significantly reducing operator exposure risks and simplifying solvent recovery.
Q: What represents the critical control point for stereochemistry in this Gefarnate synthesis?
A: The stereochemical integrity is primarily maintained during the final Mitsunobu coupling step, where strict temperature control between -5°C and 0°C ensures the preservation of the E-configuration in the polyene chain, preventing isomerization.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the process demonstrates high yields (up to 90% in the final step) and utilizes robust, scalable unit operations such as vacuum distillation and standard liquid-liquid extraction, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gefarnate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate balance of temperature and stoichiometry required for the Mitsunobu coupling in Gefarnate synthesis is maintained perfectly at every batch size. We operate stringent purity specifications and utilize rigorous QC labs equipped with advanced analytical instrumentation to verify that every shipment meets the exacting standards required for gastrointestinal pharmaceutical applications, guaranteeing a product free from the toxic solvent residues associated with older methods.
We invite you to collaborate with us to leverage this advanced synthetic route for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your product quality while driving down total landed costs.
