Optimizing Gefarnate Production: A Technical Breakthrough in Mild Wittig Chemistry for Commercial Scale
Introduction to Advanced Gefarnate Synthesis
The pharmaceutical landscape for gastrointestinal therapeutics relies heavily on the consistent supply of high-quality isoprenoid intermediates, among which Gefarnate stands out as a critical active ingredient for treating gastric and duodenal ulcers. The patent CN103012140A introduces a transformative preparation method that addresses long-standing inefficiencies in the industrial synthesis of this compound. Historically, the production of Gefarnate has been plagued by cumbersome synthetic routes that suffer from low yields, poor purity profiles, and complex post-treatment requirements that hinder large-scale manufacturing. This new technical disclosure pivots away from traditional high-energy pathways, proposing a streamlined approach centered around a mild Wittig reaction strategy. By fundamentally re-engineering the carbon-carbon bond formation steps, the methodology not only enhances the overall atomic economy but also drastically simplifies the purification workflow. For R&D directors and process chemists, this represents a significant opportunity to upgrade legacy manufacturing lines to more sustainable and efficient protocols that align with modern green chemistry principles.
Furthermore, the strategic implementation of specific polymerization inhibitors during the final esterification stage marks a crucial innovation in controlling product quality. In conventional processes, the tendency of unsaturated fatty acid derivatives to undergo unwanted polymerization under thermal stress often leads to significant material loss and the generation of difficult-to-remove impurities. The patented method mitigates this risk through the precise addition of agents such as resorcinol or phenol, which stabilize the reaction mixture during reflux. This attention to detail in reaction engineering ensures that the final distillation cuts, collected specifically between 186°C and 200°C, possess exceptional purity levels exceeding 98%. Such rigorous control over the impurity profile is essential for meeting the stringent regulatory standards required for pharmaceutical intermediates intended for human consumption, thereby securing a more reliable supply chain for downstream drug manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art technologies for synthesizing Gefarnate, such as those referenced in patents CN101475476A and CN101805260A, typically rely on the use of nerolidol as a starting material coupled with orthoformates or orthoacetates. These legacy routes are characterized by severe operational drawbacks that make them increasingly obsolete in a cost-sensitive market. The most glaring issue is the requirement for extreme reaction conditions, often necessitating temperatures as high as 155°C for extended periods ranging from 10 to 25 hours. This intense thermal demand not only results in substantial energy consumption but also promotes thermal degradation of the sensitive isoprenoid backbone, leading to a complex mixture of byproducts. Additionally, the use of trimethyl orthoformate generates methanol as a toxic byproduct, posing significant environmental and safety hazards regarding respiratory mucosa damage and volatility. The reliance on ether solvents and the subsequent necessity for column chromatography for purification further exacerbate the cost and complexity, rendering these methods unsuitable for efficient industrial scale-up.
The Novel Approach
In stark contrast, the novel approach detailed in CN103012140A leverages a Wittig reaction between a phosphorus ylide derived from ethyl 4-bromobutyrate and geranylacetone. This shift in synthetic strategy allows the critical carbon-chain elongation to proceed under remarkably mild conditions, specifically at room temperature, which preserves the integrity of the double bonds and minimizes side reactions. The elimination of high-temperature steps in the early stages of synthesis significantly reduces the energy footprint of the process. Moreover, the subsequent hydrolysis and esterification steps are optimized to avoid the use of toxic methanol and ether, replacing them with more manageable solvents like DMF and xylene. The process design inherently favors high conversion rates, with experimental data indicating yields for farnesyl ethyl acetate reaching over 90%, a substantial improvement over the roughly 72% observed in comparative examples using older methodologies. This efficiency gain translates directly into reduced raw material waste and lower production costs.
Mechanistic Insights into Wittig Olefination and Controlled Esterification
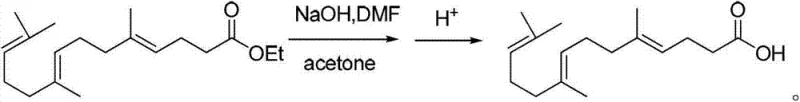
The core of this technological advancement lies in the precise execution of the Wittig olefination, which constructs the central carbon skeleton of the molecule with high stereochemical control. The process begins with the in situ generation of the phosphorus ylide from ethyl 4-bromobutyrate and triphenylphosphine in the presence of triethylamine within a tetrahydrofuran medium. This ylide then reacts with geranylacetone to form farnesyl ethyl acetate. The beauty of this mechanism is its operational simplicity; it proceeds efficiently at ambient temperatures, avoiding the thermal stress that typically causes isomerization or decomposition in polyunsaturated systems. The reaction pathway ensures that the resulting intermediate is predominantly in the desired trans-configuration, which is critical for the biological activity of the final API. Following this, the ester group is hydrolyzed using sodium hydroxide in DMF to yield farnesyl acetic acid, a key precursor that is isolated with high purity through a specialized extraction protocol involving pH adjustment and petroleum ether washing.

Following the formation of the acid intermediate, the final esterification with geraniol presents its own mechanistic challenges, primarily the risk of polymerization due to the multiple double bonds present in the structure. The patent elucidates a mechanism where the addition of specific polymerization inhibitors, such as resorcinol, interferes with the radical propagation steps that lead to oligomer formation. This allows the esterification to proceed to completion in xylene under reflux without significant degradation of the product. The use of anhydrous calcium chloride in the workup phase further aids in removing unreacted geraniol through complexation, ensuring that the final distillation yields a product of exceptional clarity and purity. This multi-faceted approach to reaction control demonstrates a deep understanding of the chemical stability issues inherent in terpenoid synthesis.
How to Synthesize Gefarnate Efficiently
The synthesis of Gefarnate via this patented route offers a robust framework for laboratory and pilot-scale production, emphasizing reproducibility and safety. The protocol is designed to minimize the need for specialized equipment, relying instead on standard chemical engineering unit operations such as reflux, distillation, and filtration. The initial formation of the ylide and subsequent coupling requires careful moisture control but avoids the cryogenic conditions often associated with sensitive organometallic reactions. For process chemists looking to implement this route, the key lies in the rigorous control of the stoichiometry during the ylide formation and the precise temperature management during the final esterification to maximize the efficacy of the polymerization inhibitor. Detailed standardized operating procedures for each step, including specific solvent ratios and workup sequences, are essential to replicate the high yields reported in the patent examples.
- Preparation of Phosphorus Ylide: React ethyl 4-bromobutyrate with triphenylphosphine and triethylamine in THF at room temperature to generate the active ylide species.
- Wittig Olefination and Hydrolysis: Couple the ylide with geranylacetone to form farnesyl ethyl acetate, followed by alkaline hydrolysis using NaOH/DMF to yield farnesyl acetic acid.
- Controlled Esterification and Purification: React farnesyl acetic acid with geraniol in xylene using a polymerization inhibitor, followed by attapulgite and activated carbon purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method offers compelling economic and logistical benefits that extend beyond simple yield improvements. The primary advantage is the drastic simplification of the purification train. By eliminating the need for column chromatography, which is a major bottleneck in batch processing and a significant source of solvent waste, the process becomes far more amenable to continuous or large-batch manufacturing. This reduction in downstream processing complexity directly correlates to a significant reduction in manufacturing costs, as it lowers both the capital expenditure on separation equipment and the operational expenditure on solvents and labor. Furthermore, the use of readily available and cost-effective starting materials like ethyl 4-bromobutyrate and geranylacetone ensures a stable supply chain that is less susceptible to the volatility associated with exotic or highly specialized reagents.
- Cost Reduction in Manufacturing: The transition to a room-temperature Wittig reaction eliminates the heavy energy loads associated with maintaining 155°C for extended periods in traditional methods. This energy efficiency, combined with the avoidance of toxic methanol byproducts which require expensive abatement systems, results in substantial cost savings. The high conversion rates mean less raw material is wasted, optimizing the cost per kilogram of the final active pharmaceutical ingredient. Additionally, the replacement of complex chromatographic purification with simple distillation and adsorption techniques using attapulgite and activated carbon significantly lowers the cost of goods sold.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as triphenylphosphine, sodium hydroxide, and xylene ensures that the supply chain is robust and resilient against disruptions. Unlike processes dependent on single-source specialty reagents, this method utilizes materials that are widely available from multiple global suppliers. The simplified workflow also reduces the lead time for production batches, allowing for more responsive inventory management and the ability to scale up production volume rapidly to meet market demand without the need for extensive process re-validation.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method is superior. The removal of methanol generation and the reduction in solvent usage align with increasingly strict global environmental regulations. The use of attapulgite and activated carbon for decolorization and purification generates solid waste that is easier to handle and dispose of compared to the liquid waste streams from column chromatography. This makes the process not only scalable to multi-ton production but also sustainable, reducing the overall environmental footprint of the manufacturing facility and ensuring long-term regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Gefarnate synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear picture of the method's capabilities and limitations. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term viability of the supply source.
Q: How does the new Wittig-based route improve upon traditional nerolidol synthesis methods?
A: Traditional methods often require harsh conditions (155°C) and produce toxic methanol byproducts. The novel Wittig route operates at room temperature for the key coupling step, significantly reducing energy consumption and eliminating toxic volatile byproducts, leading to higher purity and safer industrial handling.
Q: What role does the polymerization inhibitor play in the final esterification step?
A: During the esterification of farnesyl acetic acid with geraniol, polymeric byproducts can form, reducing yield and complicating purification. The addition of inhibitors like resorcinol or phenol suppresses these side reactions, ensuring higher conversion rates and simplifying the downstream distillation process.
Q: Is this synthesis method suitable for large-scale commercial manufacturing?
A: Yes, the process is specifically designed for industrial scalability. It avoids column chromatography, utilizes common solvents like xylene and DMF, and employs robust purification techniques such as attapulgite adsorption, making it cost-effective and feasible for multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gefarnate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of effective gastrointestinal therapies. Our technical team has extensively analyzed the synthetic pathways described in CN103012140A and possesses the expertise to execute this advanced Wittig-based protocol with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to industrial reactor is seamless. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the absence of polymeric byproducts and ensuring the correct stereochemistry of the final product, guaranteeing that every batch meets the highest international standards.
We invite pharmaceutical partners to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route. By optimizing the supply chain for Gefarnate, we can help you reduce lead time for high-purity pharmaceutical intermediates and secure a competitive advantage in the market. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your project goals and drive your product success.
