Advanced Solvent-Free Synthesis of Azoxystrobin Intermediates for Commercial Scale-Up
Advanced Solvent-Free Synthesis of Azoxystrobin Intermediates for Commercial Scale-Up
The global demand for high-efficiency fungicides continues to drive innovation in the synthesis of key agrochemical building blocks. Patent CN102241651A introduces a transformative preparation method for azoxystrobin intermediates, specifically targeting the critical bottleneck in benzofuranone synthesis. This technology addresses the longstanding challenges of incomplete conversion and solvent dependency that have plagued traditional manufacturing routes. By leveraging a solvent-free cyclization strategy combined with vacuum dehydration, this process achieves superior transformation efficiency and product purity. For R&D directors and procurement specialists, understanding the nuances of this patented route is essential for optimizing supply chains and reducing production costs in the competitive agrochemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
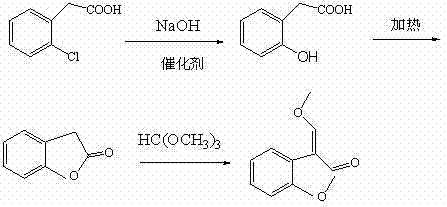
Historically, the industrial synthesis of benzofuranone, a pivotal precursor for azoxystrobin, has relied heavily on solvent-based reflux systems. Traditional protocols typically utilize toluene as a reaction medium, requiring extended reflux periods of 4 to 6 hours to drive the esterification and cyclization of o-hydroxyphenylacetic acid. Despite these prolonged reaction times, the conversion efficiency remains suboptimal, often leaving approximately 5% of the starting material unreacted. This residual o-hydroxyphenylacetic acid not only complicates downstream purification but also negatively impacts the overall yield of the final azoxystrobin active ingredient. Furthermore, the reliance on volatile organic solvents like toluene introduces significant environmental and safety liabilities, necessitating complex recovery systems and increasing the carbon footprint of the manufacturing process.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data revolutionizes this step by eliminating the solvent entirely. The process involves heating o-hydroxyphenylacetic acid with a minimal amount of catalyst, ranging from 0 to 3 percent, under vacuum conditions at temperatures between 125°C and 180°C. This solvent-free environment, coupled with continuous vacuum distillation, effectively removes the water generated during the reaction, shifting the chemical equilibrium decisively towards product formation. As a result, the reaction time is drastically shortened to just 2 to 3 hours, and the conversion rate is pushed to near completion, with residual starting material dropping to negligible levels. This methodological shift not only simplifies the operational workflow by removing solvent recovery steps but also significantly enhances the purity of the resulting benzofuranone, setting a new standard for efficiency in agrochemical intermediate manufacturing.
Mechanistic Insights into Acid-Catalyzed Solvent-Free Cyclization
The core chemical innovation lies in the intramolecular dehydration cyclization of o-hydroxyphenylacetic acid to form the lactone ring of benzofuranone. In the absence of a bulk solvent, the reactant molecules are in a highly concentrated state, which inherently increases the frequency of effective collisions. The addition of strong acid catalysts, such as sulfuric acid, methanesulfonic acid, or p-toluenesulfonic acid, serves to protonate the carboxylic acid group, making it a better electrophile for the nucleophilic attack by the ortho-hydroxyl group. Crucially, the application of vacuum plays a dual mechanistic role: it lowers the boiling point of the system to prevent thermal degradation of the sensitive organic molecules while simultaneously acting as a driving force to physically remove the water byproduct. According to Le Chatelier's principle, the continuous removal of water prevents the reverse hydrolysis reaction, thereby locking the equilibrium in favor of the cyclic benzofuranone product. This thermodynamic control is the key differentiator that allows for such high conversion rates without the need for excessive reagents or prolonged heating.
From an impurity control perspective, this mechanism offers distinct advantages over solution-phase chemistry. In traditional toluene reflux, the presence of solvent can sometimes stabilize transition states that lead to side reactions or polymerization, and the azeotropic removal of water is often less efficient than direct vacuum distillation. The patent data indicates that by maintaining the reaction temperature precisely between 135°C and 145°C, the formation of thermal degradation byproducts is minimized. Furthermore, the subsequent step involving the reaction of benzofuranone with trimethyl orthoformate in isobutyric anhydride is highly selective. The use of isobutyric anhydride as a reaction medium in the second step helps to solubilize the benzofuranone while participating in the activation of the orthoformate, ensuring that the methylene group is introduced cleanly at the 3-position of the furanone ring. This two-stage cascade ensures that the final intermediate possesses a GC purity exceeding 98%, which is critical for the efficacy of the final fungicide formulation.
How to Synthesize Azoxystrobin Intermediate Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing high-quality azoxystrobin intermediates suitable for commercial applications. The process begins with the preparation of the starting material, o-hydroxyphenylacetic acid, via the hydrolysis of o-chlorophenylacetic acid, followed by the critical solvent-free cyclization and final condensation steps. This sequence is designed to maximize atom economy and minimize waste generation, aligning with modern green chemistry principles. Operators must pay close attention to the vacuum levels and temperature gradients during the cyclization phase to ensure optimal water removal without charring the product. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are provided in the technical guide below to facilitate immediate implementation in pilot or production plants.
- Synthesize o-hydroxyphenylacetic acid by reacting o-chlorophenylacetic acid with aqueous sodium hydroxide and a copper catalyst under high temperature and pressure.
- Perform solvent-free cyclization of o-hydroxyphenylacetic acid at 125-180°C under vacuum with 0-3% acid catalyst to form benzofuranone, removing water continuously.
- React the resulting benzofuranone with trimethyl orthoformate in isobutyric anhydride at 90-95°C to generate the final azoxystrobin intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solvent-free technology translates directly into tangible operational improvements and risk mitigation. The elimination of toluene from the cyclization step removes a major variable from the supply chain, reducing dependency on volatile solvent markets and mitigating the risks associated with solvent transportation and storage regulations. Additionally, the simplified process flow, which bypasses the need for complex solvent recovery and distillation units, allows for a more streamlined production schedule. This efficiency gain means that manufacturing facilities can achieve higher throughput with existing infrastructure, effectively increasing capacity without significant capital expenditure. The reduction in reaction time from nearly 6 hours to roughly 2 hours further amplifies this capacity increase, allowing for more batches to be produced within the same operational window.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the removal of solvent costs and the associated energy consumption for solvent recovery. By operating under solvent-free conditions, the manufacturer eliminates the expense of purchasing, storing, and recycling large volumes of toluene. Furthermore, the shortened reaction time leads to substantial savings in utility costs, such as steam and electricity, required to maintain reflux conditions. The higher yield and purity also mean that less raw material is wasted, and downstream purification costs are significantly lowered, resulting in a more cost-effective overall production model for agrochemical intermediates.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain continuity by reducing the complexity of the manufacturing process. With fewer unit operations and no reliance on solvent azeotropes for water removal, the process is less prone to operational upsets or equipment failures. The raw materials required, such as o-chlorophenylacetic acid and trimethyl orthoformate, are commodity chemicals with stable global supply lines, ensuring consistent availability. This reliability is crucial for meeting the rigorous delivery schedules demanded by large-scale agrochemical companies, minimizing the risk of production delays that could disrupt the broader agricultural supply chain.
- Scalability and Environmental Compliance: Scaling up a solvent-free reaction is inherently safer and more straightforward than scaling a solvent-based one, as there is no risk of solvent flash points or explosion limits to manage in the reactor headspace. The reduced generation of hazardous waste, specifically spent solvent streams, simplifies environmental compliance and waste disposal logistics. This aligns perfectly with increasingly stringent global environmental regulations, positioning manufacturers who adopt this technology as leaders in sustainable chemical production. The ability to scale from laboratory quantities to multi-ton production runs with consistent quality makes this an ideal candidate for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on process parameters and expected outcomes. Understanding these details is vital for technical teams evaluating the feasibility of integrating this method into their existing production lines. The answers reflect the specific advantages of the solvent-free approach and its impact on product quality and process efficiency.
Q: What are the primary advantages of this solvent-free method over traditional toluene reflux?
A: The solvent-free method eliminates the need for toluene, simplifies the process, shortens reaction time from 4-6 hours to 2-3 hours, and significantly improves conversion rates, reducing residual starting material from approximately 5% to less than 1%.
Q: Is a catalyst strictly required for the benzofuranone cyclization step?
A: No, the patent specifies that the reaction can proceed with 0-3% catalyst. While catalysts like sulfuric acid, methanesulfonic acid, or p-toluenesulfonic acid can accelerate the reaction, the process is effective even without them under vacuum conditions.
Q: How does this process impact the purity of the final agrochemical intermediate?
A: By ensuring thorough conversion of o-hydroxyphenylacetic acid and effectively removing water via vacuum distillation, the process yields benzofuranone with content exceeding 97%, leading to a final intermediate purity of over 98% after crystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azoxystrobin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the efficacy of final agrochemical products. Our technical team has extensively analyzed the solvent-free cyclization technology described in patent CN102241651A and validated its potential for large-scale manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to industrial reactor is seamless and efficient. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of azoxystrobin intermediate we produce, consistently meeting the high standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this solvent-free route can optimize your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to deliver reliable, cost-effective, and high-quality agrochemical intermediates that drive success in the global market.
