Advanced Electrochemical Strategy for Scalable Alpha-Ketoamide Pharmaceutical Intermediates Manufacturing
Advanced Electrochemical Strategy for Scalable Alpha-Ketoamide Pharmaceutical Intermediates Manufacturing
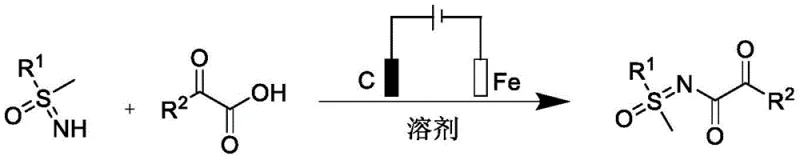
The landscape of organic synthesis is undergoing a paradigm shift towards greener, more sustainable methodologies, particularly in the production of high-value pharmaceutical intermediates. A groundbreaking development in this sector is detailed in patent CN115896824A, which discloses a novel electrochemical method for preparing α-ketoamide compounds. This technology represents a significant departure from conventional transition-metal-catalyzed pathways, offering a robust alternative that utilizes electricity as a clean oxidant. By leveraging an undivided cell setup with inexpensive electrode materials, this process achieves high yields under remarkably mild conditions, specifically at room temperature and in an open air atmosphere. For R&D directors and process chemists, this innovation promises not only enhanced purity profiles by eliminating metal residues but also a streamlined workflow that reduces the environmental footprint associated with complex amide bond formations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of α-ketoamides has relied heavily on the coupling of amines or amides with α-keto acids or acyl halides, often necessitating the use of stoichiometric amounts of chemical oxidants or transition metal catalysts such as palladium or copper. These conventional routes frequently suffer from significant drawbacks, including the generation of hazardous heavy metal waste that requires costly and time-consuming removal steps to meet stringent pharmaceutical purity standards. Furthermore, many traditional protocols demand rigorous exclusion of moisture and oxygen, requiring inert gas protection and specialized equipment that increases capital expenditure and operational complexity. The reliance on expensive catalysts also introduces supply chain vulnerabilities, as the availability and price volatility of precious metals can directly impact manufacturing costs and lead times for critical drug intermediates.
The Novel Approach
In stark contrast, the electrochemical methodology described in the patent utilizes a simple yet effective setup where electric current serves as the sole oxidant, thereby obviating the need for external chemical oxidants or transition metal catalysts. The reaction proceeds smoothly in an electrolyte-free acetonitrile solution using a graphite anode and a stainless steel cathode, which are both abundant and cost-effective materials. This approach allows the reaction to be conducted under ambient conditions without the need for inert atmosphere protection, drastically simplifying the operational procedure and reducing energy consumption associated with heating or cooling. The versatility of this method is evidenced by its broad substrate scope, successfully accommodating various substituted phenyl groups and heterocyclic moieties, making it a highly adaptable platform for the diverse needs of modern medicinal chemistry.
Mechanistic Insights into Electrochemical Oxidative Coupling
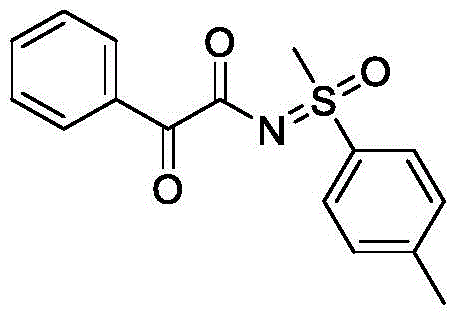
The core of this innovative process lies in the anodic oxidation of the sulfoximine substrate, which generates a reactive nitrogen-centered radical species capable of coupling with the α-keto acid. In the absence of supporting electrolytes, the acetonitrile solvent facilitates ion transport while the applied current drives the redox events at the electrode surfaces. The graphite anode provides a stable surface for the oxidation half-reaction, while the stainless steel cathode supports the corresponding reduction process, likely involving proton reduction to hydrogen gas. This electrochemical driving force ensures a continuous regeneration of the active species without the accumulation of reduced metal byproducts, leading to a cleaner reaction profile. The mechanism avoids the formation of metal-ligand complexes that often complicate downstream purification, resulting in a crude product that is significantly easier to isolate and purify via standard column chromatography techniques.
From an impurity control perspective, the elimination of transition metals is a critical advantage for pharmaceutical applications where residual metal limits are strictly regulated. The absence of metal catalysts means there is no risk of metal leaching into the final product, thereby reducing the burden on quality control laboratories and minimizing the need for specialized scavenging resins. Additionally, the mild reaction conditions help preserve sensitive functional groups that might otherwise degrade under harsh thermal or acidic conditions typical of traditional methods. The high functional group tolerance observed across various examples, including those with electron-donating and electron-withdrawing substituents, suggests a robust mechanistic pathway that is insensitive to minor variations in substrate electronics, ensuring consistent quality across different batches of production.
How to Synthesize Alpha-Ketoamide Efficiently
The practical implementation of this electrochemical synthesis is designed for ease of adoption in both laboratory and pilot-scale settings. The protocol involves mixing the sulfoximine and α-keto acid precursors in a specific molar ratio within a standard reactor, followed by the application of a constant low current. This simplicity allows for rapid screening of reaction conditions and quick optimization of parameters such as current density and reaction time. The detailed standardized synthetic steps provided below outline the precise quantities and operational parameters required to achieve optimal yields, serving as a foundational guide for process engineers looking to integrate this green chemistry solution into their existing manufacturing workflows.
- Prepare the reaction mixture by adding sulfoximine compounds and alpha-keto acids in a 1: 3 molar ratio to a reactor containing acetonitrile solvent under air atmosphere.
- Insert a graphite rod as the anode and a stainless steel sheet as the cathode into the undivided cell, then apply a constant current of 4mA for 6 hours at room temperature.
- Upon completion, evaporate the solvent under reduced pressure to isolate the crude product, followed by purification via column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this electrochemical technology offers substantial strategic benefits that extend beyond mere technical feasibility. By removing the dependency on precious metal catalysts and hazardous chemical oxidants, the overall cost structure of the manufacturing process is significantly optimized, leading to direct savings in raw material expenditures. The simplified operational requirements, such as the ability to run reactions in an open system at room temperature, reduce the need for specialized infrastructure and lower energy consumption, contributing to a more sustainable and cost-effective production model. Furthermore, the use of commodity electrode materials like graphite and stainless steel ensures a stable supply of consumables, mitigating risks associated with the volatility of the precious metals market.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and stoichiometric oxidants results in a drastic reduction in raw material costs, while the simplified workup procedure minimizes the consumption of purification media and solvents. This leaner process flow translates to lower operating expenses and improved profit margins for high-volume production runs of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Utilizing widely available and inexpensive starting materials such as sulfoximines and alpha-keto acids ensures a robust supply chain that is less susceptible to disruptions caused by the scarcity of specialized reagents. The straightforward nature of the electrochemical setup also allows for flexible manufacturing capabilities, enabling rapid scale-up to meet fluctuating market demands without significant lead time delays.
- Scalability and Environmental Compliance: The electrolyte-free nature of the reaction simplifies waste treatment protocols, as there are no heavy metal salts or toxic oxidant byproducts to manage, aligning perfectly with increasingly stringent environmental regulations. This green chemistry approach facilitates easier regulatory approval and supports corporate sustainability goals by reducing the overall environmental footprint of the chemical manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical aspects of adopting this technology for industrial applications. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this process into their current production lines.
Q: What are the primary advantages of this electrochemical method over traditional transition metal catalysis?
A: This method eliminates the need for expensive transition metal catalysts and external chemical oxidants, significantly reducing heavy metal contamination risks and simplifying downstream purification processes.
Q: Does this synthesis require specialized electrolytes or inert atmosphere conditions?
A: No, the process operates efficiently in an electrolyte-free environment using acetonitrile as the sole solvent and can be conducted under open air conditions at room temperature, greatly lowering operational complexity.
Q: What is the substrate scope for the sulfoximine and alpha-keto acid components?
A: The method demonstrates excellent functional group compatibility, accommodating various substituted phenyl groups, heterocycles like thiophene, and different alkyl substituents on both the sulfoximine and the alpha-keto acid moieties.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Ketoamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in the production of high-purity pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN115896824A can be seamlessly translated into efficient industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced electrochemical reactors, allowing us to maintain stringent purity specifications and deliver consistent quality for even the most complex molecular architectures required by the global pharmaceutical industry.
We invite you to collaborate with our technical team to explore how this cost-effective and environmentally friendly synthesis route can enhance your supply chain resilience. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can drive value and efficiency in your API intermediate manufacturing operations.
