Revolutionizing Alpha-Ketoamide Production: A Manganese-Catalyzed Breakthrough for Commercial Scale
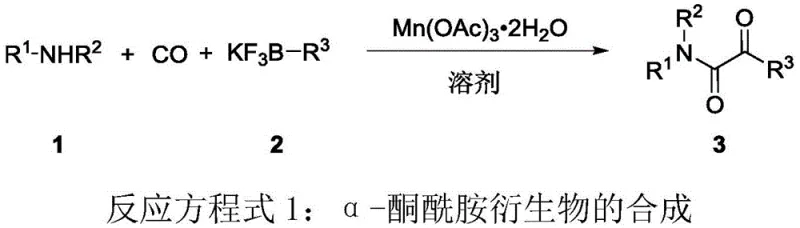
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing complex molecular architectures, particularly those containing bioactive motifs like the alpha-ketoamide scaffold. Patent CN115572239A introduces a transformative approach to synthesizing these valuable derivatives, shifting the paradigm from precious metal catalysis to base metal promotion. This innovation leverages trivalent manganese acetate to facilitate a double carbonylation amination reaction between simple amines and alkyl boron reagents under a carbon monoxide atmosphere. By replacing costly palladium systems with abundant manganese, this technology not only addresses economic constraints but also significantly expands the chemical space accessible to process chemists. For R&D directors and procurement strategists alike, this represents a critical opportunity to optimize supply chains for high-value intermediates used in drug discovery and agrochemical development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the alpha-ketoamide core has relied heavily on palladium-catalyzed double carbonylation reactions or the use of pre-functionalized alpha-keto acids and acid chlorides. While effective in specific contexts, these conventional pathways suffer from significant drawbacks that hinder large-scale adoption. The reliance on palladium-phosphine catalyst systems imposes a heavy financial burden due to the volatility of precious metal prices and the complexity of ligand synthesis. Furthermore, these Pd-catalyzed protocols often exhibit narrow substrate tolerance, frequently failing when electron-withdrawing groups are present on the electrophile or when attempting to utilize diverse amine sources beyond simple alkyl amines. Additionally, competing pathways often lead to the formation of undesired simple amide by-products, necessitating rigorous and yield-eroding purification steps that complicate the manufacturing process and reduce overall atom economy.
The Novel Approach
In stark contrast, the methodology described in CN115572239A utilizes a trivalent manganese-promoted system that elegantly circumvents these traditional bottlenecks. By employing manganese acetate dihydrate as a cheap and non-toxic oxidant, the process eliminates the need for expensive ligand systems while maintaining high catalytic efficiency. This novel route demonstrates exceptional versatility, successfully accommodating a wide array of aryl amines and alkyl boron reagents that were previously problematic. The reaction proceeds through a distinct radical mechanism that inherently suppresses the formation of simple amide side products, thereby delivering the target alpha-ketoamides with superior selectivity. This shift not only simplifies the operational workflow but also aligns perfectly with green chemistry principles by utilizing earth-abundant metals and reducing heavy metal waste in the final product stream.
Mechanistic Insights into Manganese-Promoted Double Carbonylation
The core of this technological advancement lies in the unique reactivity of trivalent manganese species under carbon monoxide pressure. Unlike palladium cycles which rely on oxidative addition and reductive elimination, the manganese-mediated pathway operates via a radical manifold. The trivalent manganese acts as a single-electron oxidant, generating alkyl radicals from the organoboron precursors. These radicals subsequently trap two molecules of carbon monoxide in a sequential insertion process before being intercepted by the amine nucleophile. This mechanistic divergence is crucial for R&D teams as it explains the observed tolerance for functional groups that typically poison palladium catalysts. The radical nature of the intermediate species allows for the activation of robust C-B bonds without requiring harsh conditions, ensuring that sensitive functional groups on the amine or boron components remain intact throughout the transformation.
Furthermore, the selectivity profile of this reaction is a direct consequence of the specific coordination environment and oxidation state of the manganese center. The process effectively avoids the over-reduction or incomplete carbonylation that often plagues other transition metal systems. By carefully controlling the stoichiometry of the manganese oxidant relative to the amine and boron inputs, the reaction drives the equilibrium towards the dicarbonylated product. This precise control minimizes the generation of impurities, resulting in a cleaner crude reaction profile. For process development scientists, this means fewer chromatographic separations and a more straightforward path to GMP-grade material. The ability to predictably manage the radical lifecycle ensures that the synthesis remains robust even when scaling from milligram to kilogram quantities, providing a reliable foundation for commercial manufacturing.
How to Synthesize Alpha-Ketoamide Derivatives Efficiently
Implementing this synthesis requires careful attention to pressure and temperature parameters to maximize the efficiency of the carbonylation steps. The protocol involves charging a high-pressure reactor with the amine substrate, the alkyl boron reagent, and the manganese oxidant in a solvent like 1,2-dichloroethane. The system is then pressurized with carbon monoxide to approximately 60 atmospheres and heated to 60°C for roughly 16 hours. This specific combination of mild thermal energy and high gas pressure is critical for driving the double insertion of CO. Detailed standardized synthetic steps and optimization data for specific substrates are provided in the guide below to assist technical teams in replicating these results.
- Combine amine, alkyl boron reagent, and trivalent manganese acetate dihydrate in a suitable solvent such as 1,2-dichloroethane within a high-pressure reactor.
- Purge the reactor with carbon monoxide and pressurize to approximately 60 atmospheres, then heat the mixture to 60°C for 16 hours.
- Upon completion, isolate the target alpha-ketoamide derivative through standard purification techniques like column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this manganese-based methodology offers profound strategic benefits that extend beyond simple yield metrics. The primary advantage lies in the drastic reduction of raw material costs associated with the catalyst system. By eliminating the dependency on palladium and specialized phosphine ligands, manufacturers can insulate their production costs from the volatile fluctuations of the precious metals market. This stability allows for more accurate long-term budgeting and pricing strategies for downstream clients. Moreover, the use of non-toxic manganese simplifies the regulatory landscape regarding heavy metal residues in the final API or intermediate, potentially reducing the burden on quality control laboratories and accelerating batch release times.
- Cost Reduction in Manufacturing: The substitution of expensive palladium catalysts with inexpensive manganese acetate results in significant direct material savings. Since manganese salts are commodity chemicals produced at massive scales globally, their supply is secure and pricing is stable compared to rare earth or precious metals. Additionally, the high selectivity of the reaction reduces the consumption of solvents and silica gel required for purification, as less time is spent separating the product from amide by-products. This cumulative effect leads to a substantially lower cost of goods sold (COGS) without compromising the quality or purity of the final alpha-ketoamide intermediate.
- Enhanced Supply Chain Reliability: The starting materials for this process, specifically aryl amines and potassium organotrifluoroborates, are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. Unlike specialized electrophiles required for Pd-catalysis which may have long lead times, these reagents are often stock items in fine chemical catalogs. This abundance ensures that production schedules can be maintained consistently, even during periods of global supply chain disruption. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in reagent quality, further enhancing the reliability of the supply output.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the use of manganese aligns better with modern sustainability goals than palladium. Manganese is not classified as a toxic heavy metal in the same regulatory category as palladium, simplifying waste disposal and effluent treatment processes. The reaction operates at moderate temperatures (60°C), which reduces energy consumption compared to high-temperature alternatives. Furthermore, the avoidance of toxic by-products and the use of standard solvents like 1,2-dichloroethane (which can be recycled) make the process easier to scale from pilot plant to multi-ton commercial production while maintaining strict environmental compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this manganese-catalyzed technology. These answers are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on substrate compatibility and operational parameters. Understanding these nuances is essential for project managers evaluating the feasibility of adopting this route for their specific pipeline candidates.
Q: Why is the manganese-catalyzed method superior to traditional palladium systems?
A: Traditional methods rely on expensive palladium-phosphine catalysts and often restrict substrate scope to electron-donating electrophiles. The manganese-promoted method utilizes cheap, non-toxic manganese acetate, accepts a broader range of amines including aryl amines, and avoids the formation of simple amide by-products through a selective radical process.
Q: What are the typical reaction conditions for this synthesis?
A: The reaction is conducted in a high-pressure reactor under a carbon monoxide atmosphere, typically at 60 atmospheres of pressure. The optimal temperature is around 60°C, using 1,2-dichloroethane as the preferred solvent, with a reaction time of approximately 16 hours to ensure high conversion and selectivity.
Q: Can this method be applied to synthesize carbene ligand precursors?
A: Yes, the resulting alpha-ketoamide products serve as versatile intermediates. As demonstrated in the patent data, specific derivatives can undergo further condensation reactions with anilines to efficiently produce carbene ligand precursors, which are critical components in organometallic catalysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Ketoamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, scalable synthetic routes in the fast-paced pharmaceutical landscape. Our technical team has thoroughly analyzed the potential of the manganese-promoted double carbonylation method described in CN115572239A and is fully prepared to leverage this technology for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless. Our facilities are equipped with high-pressure reactors capable of safely handling carbon monoxide chemistry, and our stringent purity specifications guarantee that every batch meets the rigorous demands of global regulatory bodies.
We invite you to collaborate with us to explore how this cost-effective methodology can enhance your supply chain resilience. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific target molecules, demonstrating exactly how switching to this manganese-based route can optimize your budget. Please contact our technical procurement team today to request specific COA data for similar alpha-ketoamide structures and to discuss route feasibility assessments for your upcoming campaigns. Together, we can drive innovation and efficiency in the synthesis of these vital chemical building blocks.
