Advanced Synthesis of Rivaroxaban Intermediates: A Technical Breakthrough for Scalable API Manufacturing
Advanced Synthesis of Rivaroxaban Intermediates: A Technical Breakthrough for Scalable API Manufacturing
The global demand for effective anticoagulants has surged, driven by the increasing prevalence of venous thromboembolism (VTE) and the clinical shift towards oral direct Factor Xa inhibitors. Rivaroxaban, a cornerstone in this therapeutic class, requires robust and efficient synthetic routes to meet market needs. Patent CN102250076A introduces a transformative preparation method for a key Rivaroxaban intermediate, specifically the compound represented by formula (III). This innovation addresses critical bottlenecks in traditional synthesis, such as the reliance on toxic reagents and harsh reaction conditions. By leveraging a novel magnesium halide-catalyzed cyclization strategy, this technology offers a pathway to high-purity intermediates with superior atom economy. For pharmaceutical manufacturers, adopting this methodology represents a strategic move towards greener chemistry and enhanced production efficiency.
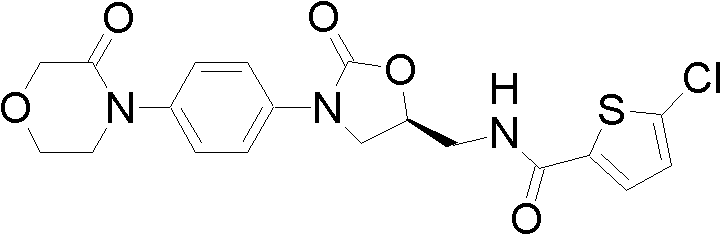
Rivaroxaban acts as a potent inhibitor of Factor Xa, preventing thrombus formation without the need for routine coagulation monitoring associated with warfarin. The structural complexity of this molecule, particularly the chiral oxazolidinone ring fused with a morpholinone phenyl system, poses significant synthetic challenges. The patent highlights a specific intermediate, 4-{4-[(5S)-5-(aminomethyl)-2-oxo-1,3-oxazolidine-3-yl]phenyl}morpholine-3-one, as a pivotal precursor. The ability to synthesize this intermediate efficiently determines the overall viability of the API production line. The disclosed method not only streamlines the construction of this core scaffold but also ensures the preservation of stereochemical integrity, which is paramount for the drug's efficacy and safety profile.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
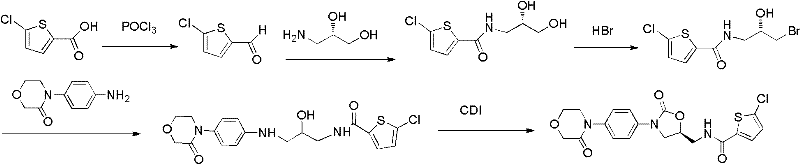
Historically, the synthesis of Rivaroxaban and its precursors has been plagued by significant technical and safety hurdles. Prior art, such as the routes disclosed in WO2005/068456 and WO 2009/023233, often relies on hazardous reagents like phosgene for carbonylation or organolithium compounds (butyllithium, tert-butyllithium) for lithiation steps. These reagents necessitate stringent safety protocols, specialized equipment for handling pyrophoric materials, and complex waste treatment systems to neutralize toxic byproducts. Furthermore, traditional pathways frequently involve multi-step sequences with low overall yields, requiring extensive purification between stages. The use of cryogenic conditions for certain lithiation reactions further escalates energy costs and limits the feasibility of scaling these processes to multi-ton production levels.
The Novel Approach
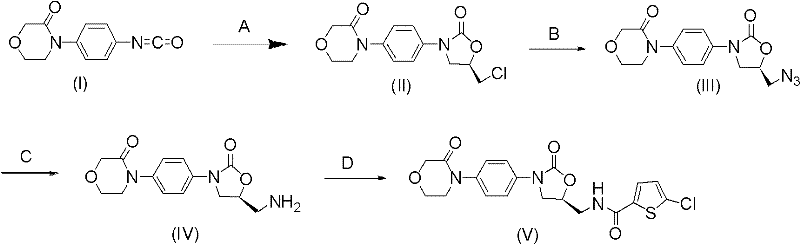
In stark contrast, the methodology outlined in Patent CN102250076A presents a streamlined, four-step sequence that circumvents these historical impediments. The core innovation lies in Step A, where 4-(4-isocyanatophenyl)-3-morpholinone reacts with (R)-epichlorohydrin under the catalytic influence of magnesium halides. This eliminates the need for dangerous acylating agents or strong bases typically required for ring closure. The subsequent conversion of the chloromethyl group to an azide (Step B) and its reduction to an amine (Step C) utilizes standard, scalable unit operations. This approach drastically simplifies the process flow, reduces the number of isolation steps, and operates under mild thermal conditions (50-90°C), making it inherently safer and more cost-effective for industrial application.
Mechanistic Insights into Magnesium Halide-Catalyzed Cyclization
The mechanistic elegance of this synthesis centers on the role of magnesium halides (MgX2, where X = I, Br, Cl) in facilitating the ring-opening and cyclization of the epoxide. In Step A, the magnesium cation acts as a Lewis acid, coordinating with the oxygen atom of the (R)-epichlorohydrin. This coordination activates the epoxide ring towards nucleophilic attack by the nitrogen of the isocyanate-derived intermediate. The specific choice of halide influences the reaction kinetics and stereoselectivity; for instance, magnesium iodide (MgI2) and magnesium bromide (MgBr2) have demonstrated superior performance compared to chloride, likely due to their higher Lewis acidity and better solubility in organic media. This catalytic activation allows the reaction to proceed at moderate temperatures (50°C) without the need for aggressive promoters, preserving the sensitive functional groups present in the morpholinone scaffold.
Following the cyclization, the introduction of the aminomethyl side chain via azide substitution (Step B) is a classic SN2 reaction, favored by the primary alkyl halide structure generated in Step A. The use of sodium azide in polar aprotic solvents like DMF or DMSO ensures high conversion rates. Crucially, the subsequent reduction of the azide to the amine (Step C) is performed via catalytic hydrogenation using Pd/C or Raney Nickel. This step is chemoselective, reducing the azide without affecting the oxazolidinone carbonyl or the thiophene ring (in later steps). The final acylation with 5-chlorothiophene-2-carbonyl chloride completes the assembly of the pharmacophore. This sequence minimizes the formation of regioisomers and impurities, resulting in a crude product that requires less intensive purification, thereby enhancing the overall process mass intensity (PMI).
How to Synthesize Rivaroxaban Intermediate Efficiently
The synthesis protocol described in the patent provides a robust framework for producing the key intermediate with high fidelity. The process begins with the dissolution of the isocyanate precursor in a solvent such as tetrahydrofuran (THF) or dichloromethane, followed by the addition of the magnesium catalyst and (R)-epichlorohydrin. After the cyclization is complete, the reaction mixture is worked up to isolate the chloromethyl-oxazolidinone. This intermediate is then subjected to azidation, followed by hydrogenation and acylation. The detailed operational parameters, including solvent ratios, temperature profiles, and catalyst loadings, are critical for reproducibility. For a comprehensive guide on executing this synthesis with optimal yield and purity, please refer to the standardized procedure below.
- Step A: React 4-(4-isocyanatophenyl)-3-morpholinone with (R)-epichlorohydrin using a magnesium halide catalyst (e.g., MgI2, MgBr2) in organic solvents like THF or DCM at 50°C to form the oxazolidinone ring.
- Step B: Perform nucleophilic substitution on the chloromethyl group using sodium azide in polar aprotic solvents (DMF, DMSO) at 60-90°C to introduce the azide functionality.
- Step C: Reduce the azide group to an amine via catalytic hydrogenation (Pd/C or Raney Ni) and immediately acylate with 5-chlorothiophene-2-carbonyl chloride to form the final amide bond.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits beyond mere technical novelty. The elimination of hazardous reagents like phosgene and organolithiums directly translates to reduced regulatory burden and lower insurance premiums associated with chemical storage and handling. Furthermore, the simplified operational workflow, characterized by fewer unit operations and milder reaction conditions, significantly lowers the barrier to entry for contract manufacturing organizations (CMOs). This accessibility fosters a more competitive supplier landscape, potentially driving down costs for the active pharmaceutical ingredient (API). The robustness of the magnesium-catalyzed step ensures consistent batch-to-batch quality, a critical factor for maintaining uninterrupted supply chains in the highly regulated pharmaceutical sector.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous reagents with inexpensive magnesium salts creates a direct material cost advantage. Additionally, the ability to run reactions at near-ambient or moderate temperatures (50-90°C) rather than cryogenic conditions drastically reduces energy consumption. The high atom economy of the cyclization step minimizes waste generation, lowering the costs associated with solvent recovery and waste disposal. These cumulative efficiencies contribute to a substantially lower cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: By utilizing commercially available and stable starting materials such as (R)-epichlorohydrin and common magnesium halides, the process mitigates the risk of raw material shortages often associated with specialty reagents. The operational simplicity allows for faster turnaround times between batches, enabling manufacturers to respond more agilely to fluctuations in market demand. This resilience is vital for ensuring the continuous availability of life-saving anticoagulant medications.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, avoiding the heat transfer limitations often encountered with exothermic lithiation reactions. The absence of heavy metal catalysts (other than standard hydrogenation catalysts which are easily removed) and toxic gases simplifies the environmental impact assessment. This alignment with green chemistry principles facilitates smoother regulatory approvals and supports corporate sustainability goals, making the supply chain more future-proof against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is essential for R&D teams evaluating process transfer and procurement teams assessing vendor capabilities. The answers are derived directly from the experimental data and beneficial effects reported in the patent documentation.
Q: What are the primary safety advantages of this new Rivaroxaban synthesis route?
A: Unlike conventional methods that utilize hazardous reagents such as phosgene, butyllithium, or tert-butyllithium, this patented process employs mild magnesium halide catalysts and avoids extreme low-temperature or anaerobic conditions, significantly reducing operational risks and environmental hazards.
Q: How does the magnesium halide catalyst improve stereoselectivity?
A: The use of specific magnesium halides (like MgI2 or MgBr2) facilitates a highly stereoselective cyclization reaction with (R)-epichlorohydrin, ensuring the formation of the desired (S)-configuration oxazolidinone core with high optical purity, which is critical for the biological activity of the final anticoagulant.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial suitability by utilizing common organic solvents, avoiding expensive transition metal catalysts, and operating under mild temperatures (50-90°C), which simplifies reactor requirements and lowers energy consumption compared to cryogenic traditional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rivaroxaban Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthetic routes in the production of high-value pharmaceutical intermediates. Our technical team has extensively analyzed the methodology presented in Patent CN102250076A and possesses the expertise to implement this magnesium-catalyzed process at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical materials. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Rivaroxaban intermediate adheres to the highest quality standards required for API synthesis.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced technology for your supply chain. By optimizing the synthesis of this key intermediate, we can help you achieve significant operational efficiencies and cost savings. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are ready to provide specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless integration of this innovative chemistry into your manufacturing portfolio.
