Scalable Cabazitaxel Production via Ambient Temperature Methylation and Optimized Side-Chain Coupling
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for high-value oncology therapeutics, and the preparation of Cabazitaxel (Jevtana) remains a critical focus area for generic API manufacturers. The patent CN113698368A, published on November 26, 2021, introduces a transformative methodology that addresses the longstanding safety and efficiency bottlenecks inherent in traditional taxane synthesis. This technical disclosure outlines a streamlined five-step sequence starting from 10-deacetylbaccatin III (10-DAB), utilizing a novel methylation strategy that operates effectively at ambient temperatures. By replacing hazardous reagents like methyl iodide and avoiding extreme cryogenic conditions, this process offers a compelling value proposition for reliable cabazitaxel intermediate suppliers aiming to optimize their production pipelines. The core innovation lies in the simultaneous methylation of the 7-position and 10-position hydroxyl groups in a single step, drastically shortening the reaction timeline while maintaining high stereochemical integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Cabazitaxel has been plagued by significant operational hazards and economic inefficiencies that hinder cost reduction in API manufacturing. Traditional routes often rely on methyl iodide, a volatile and carcinogenic alkylating agent that poses severe inhalation risks to production personnel and requires extensive containment infrastructure. Furthermore, alternative pathways utilizing methyl trifluoromethanesulfonate necessitate harsh reaction conditions, specifically ultra-low temperatures ranging from -70°C to -30°C, which demand energy-intensive cryogenic cooling systems and specialized solvents like benzophenone-distilled tetrahydrofuran. Another prevalent method involves Raney nickel hydrogenation, which introduces substantial safety liabilities due to the pyrophoric nature of the catalyst and the risks associated with high-pressure hydrogen gas handling. These legacy processes not only inflate capital expenditure through the need for specialized equipment but also complicate regulatory compliance regarding worker safety and environmental emissions, making them less attractive for modern green chemistry initiatives.
The Novel Approach
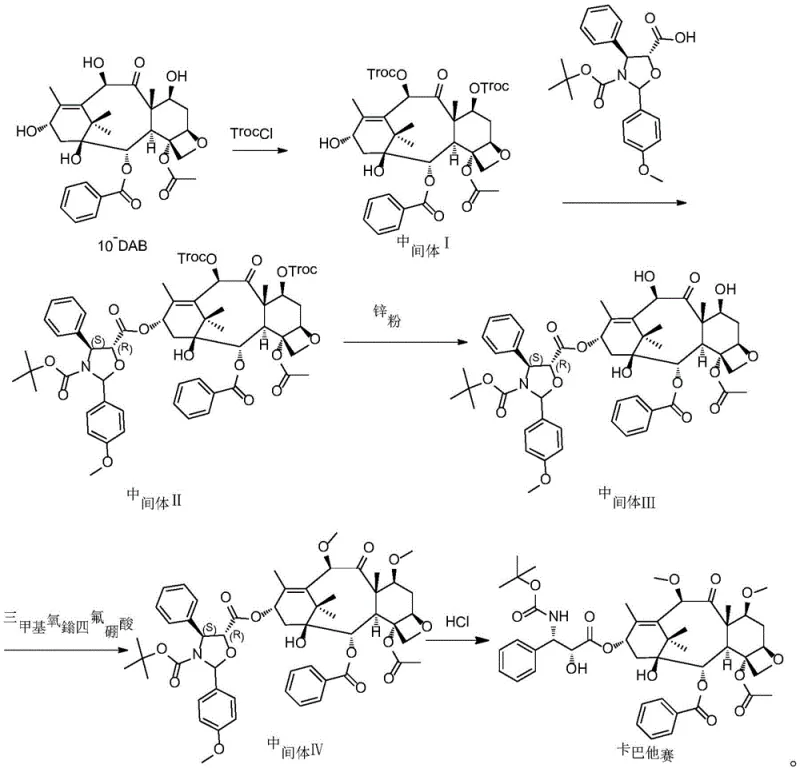
In stark contrast, the methodology disclosed in CN113698368A leverages a sophisticated protection-deprotection-methylation sequence that circumvents these historical pitfalls. The process initiates with the selective protection of hydroxyl groups using 2,2,2-trichloroethyl chloroformate (TrocCl), followed by a precise esterification with the docetaxel side chain acid. 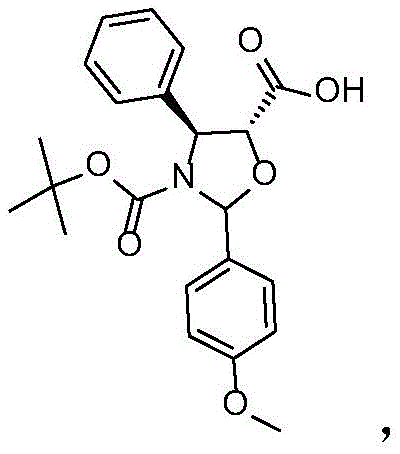 . This coupling is facilitated by N,N'-dicyclohexylcarbodiimide (DCC) and 4-dimethylaminopyridine (DMAP) in a toluene system, ensuring high conversion rates without the need for exotic catalysts. The subsequent removal of the Troc protecting groups via zinc powder reduction is conducted under mild acidic conditions, preserving the delicate taxane core. Most critically, the methylation step employs trimethyloxonium tetrafluoroborate, a stable and manageable solid reagent that allows for the simultaneous introduction of methyl groups at the C-7 and C-10 positions at temperatures between 10°C and 20°C. This elimination of cryogenic requirements and toxic gases represents a paradigm shift towards safer, more sustainable commercial scale-up of complex pharmaceutical intermediates.
. This coupling is facilitated by N,N'-dicyclohexylcarbodiimide (DCC) and 4-dimethylaminopyridine (DMAP) in a toluene system, ensuring high conversion rates without the need for exotic catalysts. The subsequent removal of the Troc protecting groups via zinc powder reduction is conducted under mild acidic conditions, preserving the delicate taxane core. Most critically, the methylation step employs trimethyloxonium tetrafluoroborate, a stable and manageable solid reagent that allows for the simultaneous introduction of methyl groups at the C-7 and C-10 positions at temperatures between 10°C and 20°C. This elimination of cryogenic requirements and toxic gases represents a paradigm shift towards safer, more sustainable commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Trimethyloxonium-Mediated Methylation
The mechanistic elegance of this synthesis is best understood through the specific interaction between the taxane substrate and the methylating agent in the presence of a proton sponge. In Step S4 of the protocol, Intermediate III is dissolved in dichloromethane and treated with 1,8-bis(dimethylamino)naphthalene (Proton Sponge) alongside trimethyloxonium tetrafluoroborate. The Proton Sponge acts as a non-nucleophilic base, effectively scavenging protons generated during the methylation event without interfering with the electrophilic methyl transfer. The trimethyloxonium cation serves as a potent methyl donor, attacking the oxygen atoms at the 7 and 10 positions of the baccatin III core. Unlike methyl iodide, which often requires strong bases that can induce elimination side reactions or epimerization, this reagent operates under neutral to slightly basic conditions that preserve the stereochemistry of the adjacent chiral centers. The reaction proceeds over a duration of 8 to 12 hours, allowing for complete conversion while minimizing the formation of demethylated impurities or over-alkylated byproducts.
Furthermore, the impurity profile is tightly controlled through the strategic use of molecular sieves and precise stoichiometric ratios. The inclusion of molecular sieves in the reaction mixture ensures the removal of trace moisture, which could otherwise hydrolyze the sensitive trimethyloxonium reagent or lead to the degradation of the intermediate. Following the methylation, the workup involves a careful precipitation strategy using methanol and water, which selectively isolates Intermediate IV while leaving soluble impurities in the mother liquor. This purification logic is carried through to the final deprotection step, where dilute hydrochloric acid in methanol cleaves the remaining ester protections to yield the final API. The entire sequence is designed to maximize the recovery of the high-value taxane skeleton, resulting in a total yield of approximately 30.7%, a figure that significantly outperforms the single-digit yields typical of older generation processes.
How to Synthesize Cabazitaxel Efficiently
The synthesis of Cabazitaxel via this patented route requires strict adherence to the specified reaction parameters to ensure optimal purity and yield. The process begins with the dissolution of 10-deacetylbaccatin III in a dichloromethane and pyridine mixture, cooled to 0-5°C before the dropwise addition of TrocCl. Following the isolation of Intermediate I, the coupling reaction with the side chain acid must be monitored closely to prevent racemization, utilizing a toluene solvent system with DCC activation. The subsequent zinc-mediated deprotection is exothermic and requires controlled addition to maintain the temperature below 10°C. For the critical methylation step, the ratio of trimethyloxonium tetrafluoroborate to the substrate is maintained between 1.4 and 1.6 (W/W) to drive the reaction to completion without excessive reagent waste. Detailed standardized synthesis steps, including specific workup procedures and crystallization protocols, are provided in the guide below.
- Protect 10-DAB with TrocCl in DCM/Pyridine at 0-10°C to form Intermediate I.
- Couple Intermediate I with docetaxel side chain acid using DCC/DMAP in toluene to form Intermediate II.
- Remove Troc groups from Intermediate II using zinc powder in ethyl acetate/acetic acid to yield Intermediate III.
- Methylate Intermediate III at positions 7 and 10 using trimethyloxonium tetrafluoroborate to obtain Intermediate IV.
- Hydrolyze Intermediate IV with HCl in methanol to finalize Cabazitaxel production.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route translates directly into enhanced operational resilience and reduced total cost of ownership. By eliminating the need for cryogenic reactors capable of sustaining -70°C, manufacturers can utilize standard glass-lined or stainless steel vessels equipped with conventional chillers, thereby drastically lowering capital investment requirements. The substitution of gaseous or highly volatile methylating agents with a stable solid reagent simplifies logistics, storage, and handling, removing the need for specialized gas containment systems and reducing the risk of supply chain disruptions caused by hazardous material transport regulations. Moreover, the ambient temperature operation significantly reduces energy consumption associated with cooling and heating cycles, contributing to lower utility costs and a smaller carbon footprint for the manufacturing facility.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the reaction infrastructure and the improvement in overall yield. By consolidating the methylation of the 7 and 10 positions into a single step, the process reduces the number of unit operations, solvent usage, and labor hours required per kilogram of output. The avoidance of expensive and dangerous reagents like Raney nickel and methyl triflate further lowers the raw material costs and waste disposal fees. Additionally, the higher yield of roughly 30% compared to historical benchmarks means that less starting material (10-DAB) is required to produce the same amount of final API, directly improving the margin profile for high-purity cabazitaxel production.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as 10-deacetylbaccatin III, which is a natural extraction product, ensures a stable supply base compared to routes requiring semi-synthetic precursors that may have limited availability. The robustness of the reaction conditions, which tolerate minor fluctuations in temperature better than cryogenic processes, reduces the risk of batch failures and production delays. This reliability is crucial for maintaining consistent inventory levels and meeting the rigorous delivery schedules demanded by global pharmaceutical clients, effectively reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the process offers distinct advantages by minimizing the generation of hazardous waste streams. The absence of heavy metal catalysts like nickel eliminates the need for complex metal scavenging steps and reduces the burden on wastewater treatment facilities. The use of common organic solvents like dichloromethane, toluene, and ethyl acetate facilitates efficient solvent recovery and recycling, aligning with green chemistry principles. This environmental compatibility simplifies the permitting process for new manufacturing lines and ensures long-term compliance with increasingly stringent international environmental regulations, supporting the commercial scale-up of complex oncology drugs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Cabazitaxel synthesis method. These answers are derived directly from the experimental data and beneficial effects described in patent CN113698368A, providing clarity on the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term viability of the supply source.
Q: How does this patent improve safety compared to traditional Cabazitaxel synthesis?
A: This method eliminates the use of highly toxic methyl iodide and dangerous Raney nickel hydrogenation. Instead, it utilizes trimethyloxonium tetrafluoroborate for methylation at ambient temperatures, significantly reducing occupational health risks and explosion hazards associated with high-pressure hydrogenation.
Q: What yield improvements can be expected with this novel route?
A: According to the patent data, this optimized route achieves a total yield of approximately 30.7%, which represents a substantial increase compared to the roughly 4% yield observed in prior art methods like WO 96/30355. This efficiency gain is driven by the one-step methylation strategy.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is highly scalable because it operates at mild temperatures (10-20°C) rather than requiring extreme cryogenic conditions (-70°C) or complex high-pressure equipment. The use of readily available starting materials like 10-deacetylbaccatin III further supports continuous commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cabazitaxel Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the growing global demand for oncology therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify that every batch of Cabazitaxel adheres to the highest international standards. Our facility is equipped to handle the specific solvent systems and reagent requirements of this patented process, guaranteeing a consistent supply of high-quality API intermediates.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this safer and more efficient production method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a successful and profitable collaboration in the competitive pharmaceutical market.
