Advanced Zero-Valent Nickel Catalysis for Efficient Adiponitrile Precursor Manufacturing and Commercial Scale-Up
The chemical industry's relentless pursuit of efficient nylon precursors has found a significant breakthrough in the technology disclosed in patent CN1169143A, which outlines an improved liquid-phase process for the hydrocyanation of diolefin compounds. This seminal work addresses the critical challenge of converting abundant feedstocks like 1,3-butadiene into non-conjugated acyclic nitriles, specifically targeting the production of adiponitrile, a vital monomer for Nylon 6,6 manufacturing. The core innovation lies in the utilization of a catalyst precursor composition comprising zero-valent nickel coordinated with specific multidentate phosphite ligands, a departure from traditional systems that often relied on less efficient monodentate ligands or required harsh promoters. By leveraging this advanced catalytic architecture, manufacturers can achieve superior conversion rates and selectivity without the operational complexities associated with earlier generations of hydrocyanation technology. The process not only streamlines the synthesis of 3- and 4-linear monoolefin nitriles but also facilitates their subsequent isomerization, creating a cohesive and economically attractive pathway for high-volume chemical production. 
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the hydrocyanation of alkenes, particularly non-activated olefins and diolefins, has been plagued by the necessity for Lewis acid promoters to achieve acceptable reaction rates and selectivity. Traditional methods, such as those disclosed in earlier patents like US 3,496,215, often utilized monodentate phosphite nickel catalysts which suffered from limited stability and required the addition of corrosive Lewis acids to activate the substrate. These promoters introduced significant downstream processing burdens, including the need for extensive neutralization steps, complex waste treatment protocols, and potential equipment corrosion issues that increased maintenance costs and downtime. Furthermore, conventional catalyst systems frequently exhibited poor regioselectivity, leading to high proportions of branched nitrile isomers like 2-methyl-3-butenenitrile, which are less desirable for adiponitrile synthesis and require energy-intensive isomerization or separation processes. The reliance on these older technologies often resulted in lower overall yields and higher operational expenditures, creating a bottleneck for manufacturers aiming to scale production efficiently in a competitive market environment.
The Novel Approach
The novel approach presented in this patent fundamentally shifts the paradigm by employing zero-valent nickel complexes stabilized by multidentate phosphite ligands, effectively eliminating the need for external Lewis acid promoters. This strategic modification allows the reaction to proceed under milder conditions while maintaining high catalytic activity, thereby reducing energy consumption and enhancing process safety. The specific geometry and electronic properties of the multidentate ligands, such as those represented by Formula I, create a steric environment around the nickel center that favors the formation of linear nitrile products over branched isomers. This intrinsic selectivity simplifies the product profile, reducing the load on downstream purification units and increasing the overall atom economy of the process. Additionally, the robustness of these catalyst systems supports both batch and continuous flow operations, offering flexibility for manufacturers to optimize their production lines for maximum throughput and minimal waste generation. 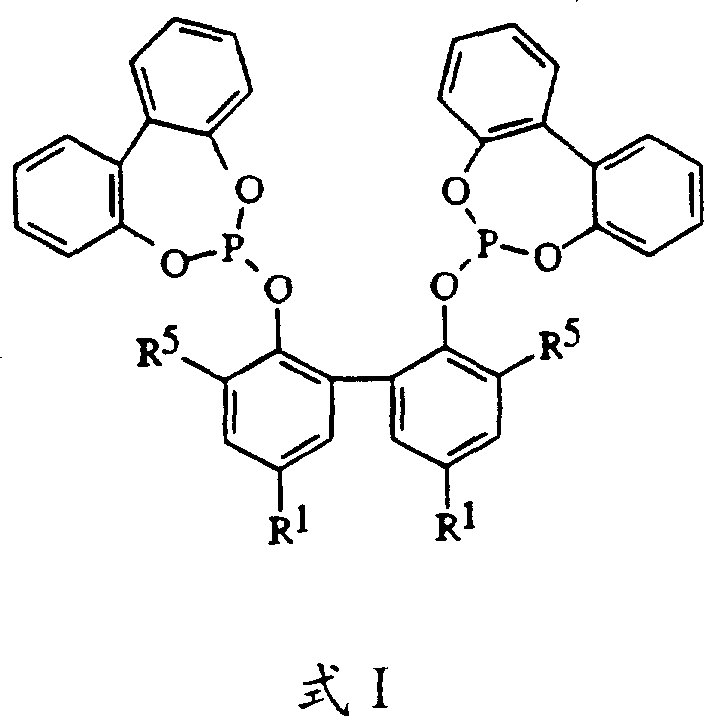
Mechanistic Insights into Zero-Valent Nickel Catalyzed Hydrocyanation
The mechanistic efficacy of this process is rooted in the unique coordination chemistry between the zero-valent nickel center and the multidentate phosphite ligands, which modulate the electronic density and steric bulk of the active catalytic species. Upon mixing the nickel source, such as Ni(COD)2, with the ligand, a stable complex forms that is capable of oxidative addition with hydrogen cyanide, followed by the insertion of the diolefin substrate into the nickel-hydride bond. The multidentate nature of the ligand ensures that the nickel center remains coordinatively saturated enough to prevent decomposition yet open enough to facilitate substrate binding and migratory insertion. Crucially, the substituents on the ligand framework, particularly the bulky tertiary alkyl groups at the ortho-positions of the phenolic rings, exert a profound influence on the transition state geometry, steering the reaction pathway towards the formation of linear 3- and 4-pentenenitriles rather than the thermodynamically favored branched isomers. This steric control is essential for maximizing the yield of precursors that can be directly converted into adiponitrile, thereby enhancing the overall efficiency of the nylon value chain.
Furthermore, the catalyst system demonstrates remarkable versatility in facilitating the isomerization of non-conjugated acyclic nitriles, a critical step for valorizing branched byproducts generated during the initial hydrocyanation. The same zero-valent nickel-phosphite complex that drives the addition of HCN across the double bond is also capable of catalyzing the migration of the double bond and the cyano group to form the desired linear configuration. This dual functionality implies that a single catalyst system can manage both the primary synthesis and the corrective isomerization steps, simplifying reactor design and operational protocols. The mechanism likely involves the formation of pi-allyl nickel intermediates, where the ligand environment stabilizes specific conformers that favor the release of the linear product. Understanding these subtle electronic and steric interactions allows process chemists to fine-tune ligand structures for even greater performance, ensuring that the technology remains at the forefront of industrial catalysis for fine chemical intermediates.
How to Synthesize Linear Pentenenitriles Efficiently
The synthesis of high-purity linear pentenenitriles using this patented technology involves a carefully controlled sequence of catalyst preparation, reaction execution, and product isolation that leverages the unique properties of the zero-valent nickel-multidentate ligand system. Operators must first ensure the exclusion of oxygen and moisture to maintain the integrity of the zero-valent nickel species, typically conducting the process under an inert nitrogen atmosphere within specialized reactors constructed from compatible materials like stainless steel or Hastelloy. The detailed standardized synthesis steps involve precise stoichiometric ratios of the nickel precursor to the phosphite ligand, optimized temperature profiles ranging from ambient to moderately elevated levels, and specific solvent choices that balance solubility with ease of product recovery. This rigorous approach ensures that the catalytic cycle operates at peak efficiency, delivering consistent quality and yield across multiple production batches while minimizing the formation of hazardous byproducts.
- Preparation of the catalyst precursor by mixing zero-valent nickel sources like Ni(COD)2 with specific multidentate phosphite ligands (e.g., Formula I) in an inert solvent such as propionitrile.
- Execution of the hydrocyanation reaction by introducing 1,3-butadiene and hydrogen cyanide into the reactor containing the catalyst solution at temperatures ranging from 0°C to 150°C under inert atmosphere.
- Performance of the isomerization step on the resulting non-conjugated acyclic nitriles using the same catalyst system to convert branched isomers into valuable 3- and 4-linear monoolefin nitriles.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this hydrocyanation technology translates into tangible strategic advantages that extend far beyond simple reaction chemistry, impacting the total cost of ownership and supply resilience. The elimination of Lewis acid promoters represents a direct reduction in raw material procurement costs, as these additives are often expensive and subject to volatile market pricing, while their removal also drastically simplifies waste management logistics by reducing the volume of corrosive effluent requiring neutralization. Moreover, the enhanced selectivity for linear nitriles means that less feedstock is wasted on producing off-spec branched isomers, effectively increasing the yield per ton of butadiene consumed and lowering the variable cost per unit of output. This efficiency gain is compounded by the ability to run the process in continuous mode, which smooths out production schedules and reduces the inventory buffers typically required for batch processing, thereby freeing up working capital. 
- Cost Reduction in Manufacturing: The removal of Lewis acid promoters from the process workflow eliminates a significant cost center associated with both the purchase of these reagents and the disposal of the resulting salt waste. By relying solely on the intrinsic activity of the nickel-phosphite complex, manufacturers can achieve substantial savings in operational expenditures related to chemical consumption and effluent treatment infrastructure. Additionally, the higher selectivity for linear products reduces the energy burden on downstream distillation columns, as there is less need to separate and recycle unwanted branched isomers, leading to lower utility costs for steam and cooling water. These cumulative savings contribute to a more competitive cost structure for adiponitrile and its derivatives, allowing companies to maintain healthy margins even in fluctuating commodity markets.
- Enhanced Supply Chain Reliability: The robustness of the zero-valent nickel catalyst system ensures consistent performance over extended periods, reducing the frequency of catalyst change-outs and the associated production stoppages that can disrupt supply continuity. The ability to utilize standard industrial solvents and operate under relatively mild pressure conditions means that the process can be implemented in existing facilities with minimal retrofitting, accelerating the time-to-market for new capacity. Furthermore, the reduced dependency on specialized promoters mitigates the risk of supply chain bottlenecks for critical reagents, ensuring that production schedules remain unaffected by external vendor delays. This reliability is crucial for meeting the just-in-time delivery requirements of major polymer manufacturers who depend on a steady flow of high-quality intermediates.
- Scalability and Environmental Compliance: The liquid-phase nature of this hydrocyanation process makes it inherently scalable, allowing for seamless transition from pilot plant trials to full commercial production without the engineering challenges often associated with gas-phase reactions. The absence of corrosive Lewis acids significantly lowers the environmental footprint of the facility, simplifying compliance with increasingly stringent global regulations regarding hazardous waste discharge and worker safety. This environmental advantage not only reduces regulatory risk but also enhances the corporate sustainability profile, which is becoming an increasingly important factor for customers selecting long-term suppliers in the specialty chemicals sector. The process design supports the implementation of closed-loop systems that maximize resource efficiency and minimize emissions, aligning with modern green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced hydrocyanation technology, providing clarity for stakeholders evaluating its potential integration into their manufacturing portfolios. These insights are derived directly from the experimental data and process descriptions contained within the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these nuances is essential for optimizing the deployment of this technology and realizing its full economic potential.
Q: What is the primary advantage of using multidentate phosphite ligands in this hydrocyanation process?
A: The use of multidentate phosphite ligands, particularly those defined in Formula I of the patent, eliminates the necessity for Lewis acid promoters which were traditionally required. This simplification significantly reduces raw material costs, minimizes corrosive waste streams, and enhances the selectivity towards linear pentenenitriles essential for adiponitrile production.
Q: Can this process be adapted for continuous flow manufacturing?
A: Yes, the patent explicitly describes embodiments suitable for continuous operation, including fixed-bed catalyst systems. The stability of the zero-valent nickel complex with these specific ligands allows for prolonged catalyst life and consistent throughput, making it highly viable for large-scale industrial continuous processing.
Q: How does this technology impact the purity profile of the final nitrile products?
A: By avoiding Lewis acid promoters and utilizing sterically bulky ligands, the process achieves superior regioselectivity, drastically reducing the formation of unwanted branched byproducts. This results in a cleaner product stream that requires less energy-intensive downstream purification to meet the stringent purity specifications required for polymer-grade adiponitrile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Adiponitrile Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the zero-valent nickel catalyzed hydrocyanation process described in patent CN1169143A and are fully equipped to support its commercial realization for our global partners. As a premier CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory concept to industrial reality is seamless and efficient. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that validate every batch against the highest industry standards, guaranteeing that the adiponitrile intermediates we supply meet the exacting requirements of downstream polymerization processes. We understand that consistency is key in the fine chemical sector, and our state-of-the-art facilities are designed to deliver that reliability at scale.
We invite forward-thinking organizations to collaborate with us to leverage this cutting-edge technology for their specific applications, whether in nylon production or other specialized chemical syntheses. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this promoter-free process for your unique operational context. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your supply chain. Together, we can redefine the standards of excellence in fine chemical manufacturing.
