Revolutionizing Adiponitrile Production with Aqueous Biphasic Hydrocyanation Technology
Revolutionizing Adiponitrile Production with Aqueous Biphasic Hydrocyanation Technology
The chemical industry's relentless pursuit of efficient, environmentally benign processes for synthesizing critical polymer precursors has found a significant breakthrough in the technology disclosed in patent CN1198151A. This seminal work details an advanced method for the hydrocyanation of ethylenically unsaturated organic compounds, specifically targeting the production of nitriles such as adiponitrile, a vital monomer for Nylon 6,6. Unlike conventional approaches that struggle with catalyst recovery and metal contamination, this invention leverages a sophisticated aqueous biphasic catalytic system. By utilizing specific water-soluble phosphine ligands in conjunction with transition metal compounds, typically nickel, the process achieves a remarkable balance between high catalytic activity and ease of separation. For R&D directors and process engineers, this represents a paradigm shift from homogeneous organic systems to heterogeneous-like aqueous systems that retain the kinetic benefits of homogeneous catalysis while offering the separability of heterogeneous processes. The implications for scaling up the production of high-purity fine chemical intermediates are profound, offering a pathway to reduce operational complexity and environmental footprint simultaneously.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the hydrocyanation of olefins, such as the conversion of butadiene to pentenenitriles and subsequently to adiponitrile, has relied heavily on homogeneous catalysis in organic media. As noted in prior art like French Patent 1,599,761, these traditional methods employ nickel catalysts complexed with triaryl phosphines in solvents like benzene or acetonitrile. While effective in terms of reaction kinetics, these systems suffer from severe downstream processing drawbacks. The primary limitation lies in the difficulty of separating the catalyst solution from the reaction products at the end of the cycle. Since both the catalyst and the products are dissolved in the same organic phase, separation often requires energy-intensive distillation or complex extraction procedures. This not only increases operational costs but also leads to significant losses of the expensive transition metal catalyst, typically nickel, into the product stream or waste residues. Furthermore, the presence of residual heavy metals in the final nitrile product poses quality control challenges for downstream polymerization processes, necessitating additional purification steps that erode profit margins and complicate the supply chain for high-purity agrochemical intermediates and polymer additives.
The Novel Approach
The methodology presented in CN1198151A fundamentally addresses these separation bottlenecks by introducing a biphasic reaction medium consisting of water and a water-immiscible organic solvent. The core innovation lies in the use of specially designed water-soluble phosphines, defined by general formulas (I) and (II), which solubilize the transition metal catalyst exclusively in the aqueous phase. During the reaction, the olefinic substrate and hydrogen cyanide react at the interface or within the aqueous phase, but the resulting nitrile products, being organic and water-insoluble, partition into the organic phase or form a separate layer. This physical property difference allows for a straightforward separation of the product from the catalyst simply by decantation or phase separation once the reaction is complete. The aqueous catalyst phase, retaining the valuable nickel complex and phosphine ligands, can then be directly recycled into the next batch or continuous flow reactor without extensive regeneration. This approach not only drastically simplifies the工艺流程 (process flow) but also minimizes the discharge of heavy metal-containing waste, aligning with increasingly stringent global environmental regulations regarding industrial effluent and metal disposal.
Mechanistic Insights into Ni-Catalyzed Aqueous Hydrocyanation
The efficacy of this hydrocyanation process is deeply rooted in the coordination chemistry between the transition metal center and the tailored water-soluble ligands. The catalyst system typically employs a zero-valent nickel source, such as bis(cyclooctadiene)nickel(0), which is stabilized by the water-soluble phosphine ligands in the aqueous medium. These ligands, often containing hydrophilic substituents like sulfonate (-SO3M), carboxylate (-COOM), or phosphonate groups, ensure that the active catalytic species remains confined to the water phase. The reaction mechanism involves the oxidative addition of hydrogen cyanide to the nickel center, followed by the coordination and insertion of the olefinic double bond. A critical aspect of this mechanism is the role of Lewis acid promoters, such as zinc chloride or tin chloride, which are added to the aqueous phase. These promoters interact with the catalyst complex to enhance the linearity of the addition reaction, favoring the formation of linear nitriles (like adiponitrile) over branched isomers, which is crucial for the quality of the final polymer precursor. The specific electronic and steric properties of the phosphine ligands, particularly the bidentate structures shown in the patent, play a pivotal role in stabilizing the active nickel species against decomposition, thereby extending the catalyst's operational lifetime.
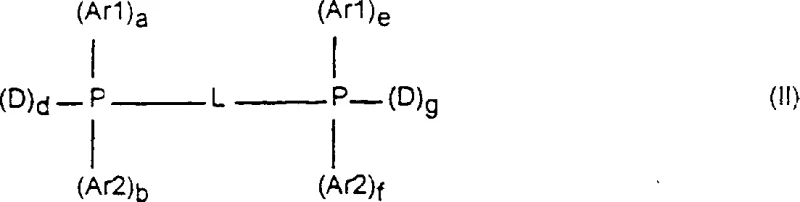
As illustrated in the structural representation of Formula (II), the bidentate phosphine ligands feature two phosphorus atoms linked by a divalent radical L, which can be an alkylene or arylene group. This chelating effect creates a more rigid and stable coordination environment around the nickel atom compared to monodentate ligands. The stability provided by these chelating ligands is essential for maintaining catalytic activity over multiple cycles, addressing the historical issue of catalyst deactivation seen in earlier aqueous systems like those using simple sulfonated triphenylphosphine. Furthermore, the presence of hydrophilic groups on the aryl rings (Ar1, Ar2) ensures high water solubility, preventing the ligand from leaching into the organic product phase. This precise molecular design ensures that the catalyst remains active and contained, facilitating the high turnover numbers and selectivity required for commercial viability. The ability to tune the substituents on the phosphine backbone allows chemists to optimize the electron density at the metal center, fine-tuning the reaction rate and selectivity for specific substrates like pentenenitriles or butadiene.
How to Synthesize Adiponitrile Efficiently
The practical implementation of this hydrocyanation technology involves a streamlined sequence of operations designed for both batch and continuous manufacturing environments. The process begins with the in-situ generation or pre-formation of the active nickel-phosphine catalyst complex in an aqueous medium, often requiring a reducing agent if higher oxidation state nickel salts are used as precursors. Once the catalyst solution is prepared, the olefinic substrate, such as 3-pentenenitrile, is introduced along with a Lewis acid promoter to enhance linear selectivity. Hydrogen cyanide is then fed into the reactor under controlled conditions, typically at temperatures ranging from 30°C to 120°C, to drive the addition reaction. The beauty of this system lies in its workup; upon completion, the mixture separates into distinct aqueous and organic layers, allowing the product-rich organic phase to be drawn off while the catalyst remains behind for reuse.
- Prepare the aqueous catalyst solution by mixing a transition metal compound (such as nickel cyclooctadiene) with a specific water-soluble phosphine ligand (Formula I or II) and a reducing agent if necessary, under inert atmosphere.
- Introduce the olefinic substrate (e.g., pentenenitrile) and hydrogen cyanide into the reactor containing the catalyst solution, optionally adding a Lewis acid promoter like zinc chloride to enhance linearity.
- Maintain reaction temperature between 30°C and 120°C, then separate the organic product phase from the aqueous catalyst phase by simple decantation for catalyst recycling.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN1198151A offers compelling economic and logistical advantages that extend far beyond simple yield improvements. The most significant benefit is the drastic reduction in operating costs associated with catalyst consumption and waste management. In traditional organic solvent processes, the loss of nickel catalyst into the product or waste streams represents a direct financial drain, not only due to the cost of the metal itself but also due to the expensive procedures required to remove trace metals from the final product to meet purity specifications. By confining the catalyst to the aqueous phase, this new method virtually eliminates metal loss, allowing for extensive recycling of the catalytic solution. This translates to a substantial decrease in the cost of goods sold (COGS) over the lifecycle of the plant. Additionally, the elimination of complex distillation steps for solvent recovery reduces energy consumption and maintenance requirements, further enhancing the overall cost-efficiency of the manufacturing process.
- Cost Reduction in Manufacturing: The shift to an aqueous biphasic system removes the need for large volumes of volatile organic solvents and the associated recovery infrastructure. Traditional methods often require energy-intensive distillation columns to separate products from solvents and catalysts, which incurs high utility costs and capital expenditure. In contrast, the phase separation in this novel process is driven by gravity or simple centrifugation, requiring significantly less energy. Furthermore, the extended lifetime of the catalyst due to the stabilizing effect of the water-soluble phosphines means that the frequency of catalyst replenishment is drastically reduced. This qualitative improvement in catalyst stability directly correlates to lower raw material costs and reduced downtime for catalyst changeovers, providing a robust buffer against fluctuations in nickel prices and ensuring more predictable budgeting for long-term production contracts.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the complexity of chemical synthesis and the dependency on specialized reagents. The simplified workflow of this hydrocyanation method enhances reliability by reducing the number of unit operations and potential failure points in the production line. The ability to operate in a continuous mode, where the aqueous catalyst is retained in the reactor while product is continuously withdrawn, allows for steady-state production that is easier to schedule and manage. This consistency is crucial for meeting the just-in-time delivery requirements of downstream polymer manufacturers. Moreover, the use of water as the primary solvent mitigates risks associated with the storage and handling of flammable organic solvents, improving plant safety and reducing insurance premiums. The robustness of the catalyst system also means that production is less susceptible to interruptions caused by catalyst deactivation, ensuring a steady flow of high-purity intermediates to customers.
- Scalability and Environmental Compliance: As regulatory pressures mount regarding industrial emissions and heavy metal discharge, the environmental profile of a chemical process becomes a key factor in supply chain qualification. This aqueous hydrocyanation technology inherently addresses these concerns by minimizing the generation of hazardous waste. The containment of the nickel catalyst within the aqueous phase prevents heavy metal contamination of the organic product and reduces the load on wastewater treatment facilities. This compliance advantage facilitates faster regulatory approvals for new production lines and reduces the risk of fines or shutdowns due to environmental non-compliance. From a scalability perspective, the biphasic nature of the reaction is well-suited for large-scale reactors, as heat transfer and mixing in aqueous systems are generally more manageable than in viscous organic media. This ease of scale-up ensures that supply can be rapidly expanded to meet market demand without the need for proportional increases in waste treatment capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced hydrocyanation technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into existing production facilities or for procurement specialists assessing the quality and consistency of the supply.
Q: How does the aqueous biphasic system improve catalyst recovery compared to traditional organic solvent methods?
A: Traditional methods often require complex distillation or extraction to separate the catalyst from the product, leading to metal loss. The aqueous biphasic system described in CN1198151A utilizes water-soluble phosphines that keep the catalyst in the aqueous phase, allowing for simple phase separation (decantation) of the organic product, thereby minimizing metal waste and simplifying recycling.
Q: What specific ligands are preferred for enhancing catalyst life in this hydrocyanation process?
A: The patent highlights the use of specific monodentate or bidentate water-soluble phosphines, such as sulfonated triaryl phosphines or those containing hydrophilic groups like carboxylates or phosphonates. Examples include CBDTS (1,2-bis[bis(sulfonyl-phenyl)-phosphinomethyl]tetramethylene sodium salt) which demonstrate improved stability and activity compared to standard sulfonated phosphines.
Q: Can this process be adapted for the continuous production of adiponitrile?
A: Yes, the method is explicitly designed for both batch and continuous operation. The ability to retain the aqueous catalyst phase in the reactor while continuously removing the organic product phase makes it highly suitable for industrial scale-up and continuous manufacturing of key intermediates like adiponitrile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Adiponitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the aqueous biphasic hydrocyanation technology detailed in CN1198151A for the production of high-value nitrile intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate these laboratory-scale innovations into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot studies to full-scale output is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, leveraging our rigorous QC labs to monitor every batch for trace metal content and isomeric purity, guaranteeing that our adiponitrile and pentenenitrile supplies are perfectly suited for the demanding requirements of nylon polymerization and fine chemical synthesis.
We invite global partners to collaborate with us to leverage this cost-effective and environmentally sustainable technology for their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a reliable, high-quality supply of critical chemical intermediates while optimizing your production costs and environmental footprint through the adoption of next-generation catalytic technologies.
