Scalable Synthesis of Pregabalin Intermediates via S-(+)-Leucine Chiral Pool Strategy
Introduction to Advanced Lyrica Intermediate Manufacturing
The pharmaceutical landscape for central nervous system (CNS) therapeutics continues to evolve, with Pregabalin (Lyrica) remaining a cornerstone treatment for neuropathic pain and epilepsy. The manufacturing of this critical active pharmaceutical ingredient (API) relies heavily on the efficiency and safety of its precursor synthesis. Patent CN101585778A discloses a groundbreaking preparation method that fundamentally shifts the paradigm from hazardous azide chemistry and inefficient racemic resolution to a streamlined chiral pool strategy. By utilizing S-(+)-leucine as the foundational building block, this technology offers a robust pathway that aligns perfectly with modern green chemistry principles and industrial safety standards. For R&D directors and procurement specialists seeking a reliable lyrica intermediate supplier, understanding the nuances of this patent is essential for securing a sustainable supply chain. The disclosed method not only mitigates the risks associated with toxic reagents but also simplifies the downstream processing, thereby enhancing the overall economic viability of producing high-purity Pregabalin.
This technical insight report delves deep into the mechanistic advantages and commercial implications of the process described in CN101585778A. We will analyze how the strategic selection of starting materials impacts the impurity profile, a critical factor for regulatory approval. Furthermore, we will explore how this route facilitates cost reduction in API manufacturing by eliminating expensive resolution steps and hazardous waste streams. As the global demand for generic versions of blockbuster drugs increases, the ability to scale complex syntheses safely becomes a key differentiator. The following analysis provides a comprehensive overview of why this specific synthetic route represents a superior choice for commercial scale-up of complex pharmaceutical intermediates, ensuring both quality consistency and operational excellence.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Pregabalin has been plagued by significant safety and efficiency challenges inherent to traditional methodologies. One prevalent approach involves the asymmetric synthesis of a trinitride intermediate, often relying on sodium azide as a key reagent. As illustrated in the chemical structure below, this pathway introduces severe occupational health hazards due to the extreme toxicity and potential explosivity of azide compounds.
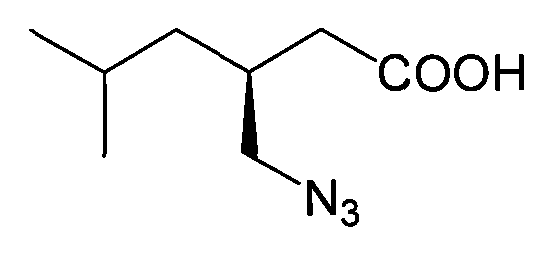
The handling of sodium azide requires specialized equipment, rigorous safety protocols, and complex waste treatment procedures, all of which drive up operational costs and limit production throughput. Another common conventional method involves the synthesis of a racemic mixture followed by chiral resolution. This approach, represented by the racemic compound structure shown below, suffers from a theoretical maximum yield of only 50% for the desired enantiomer unless dynamic kinetic resolution is employed, which adds further complexity.
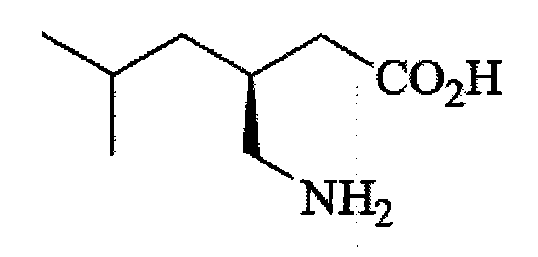
The necessity for resolution means that half of the synthesized material is essentially waste or requires energy-intensive recycling processes, drastically impacting the atom economy and environmental footprint of the manufacturing process. These limitations create bottlenecks in the supply chain, making it difficult to meet the surging global demand for Pregabalin without incurring prohibitive costs or safety risks.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in CN101585778A leverages the power of chiral pool synthesis to bypass these obstacles entirely. By initiating the synthesis with S-(+)-leucine, a naturally occurring and optically pure amino acid, the process inherently establishes the correct stereochemistry from the very first step. This eliminates the need for any chiral resolution later in the sequence, theoretically doubling the yield compared to racemic methods. The route is characterized by mild reaction conditions and the avoidance of hypertoxic reagents like sodium azide, making it significantly safer for industrial operators. The streamlined nature of this synthesis reduces the number of unit operations, which directly correlates to lower capital expenditure and reduced cycle times. For supply chain heads focused on reducing lead time for high-purity pharmaceutical intermediates, this approach offers a compelling solution that balances speed, safety, and quality.
Mechanistic Insights into Chiral Pool Alkylation Strategy
The core of this innovative synthesis lies in the transformation of the amino acid side chain while preserving the chiral center. The process begins with the reduction of S-(+)-leucine to the corresponding amino alcohol using lithium aluminum hydride. This step is critical as it converts the carboxylic acid functionality into a primary alcohol, setting the stage for subsequent functional group manipulations. The preservation of stereochemical integrity during this reduction is paramount, and the patent specifies controlled conditions to ensure no racemization occurs. Following this, the amino group is converted into a leaving group via diazotization and subsequent substitution with bromide, utilizing copper(I) bromide as a catalyst. This Sandmeyer-type transformation is highly efficient and allows for the introduction of a reactive handle for the crucial carbon-carbon bond-forming step.
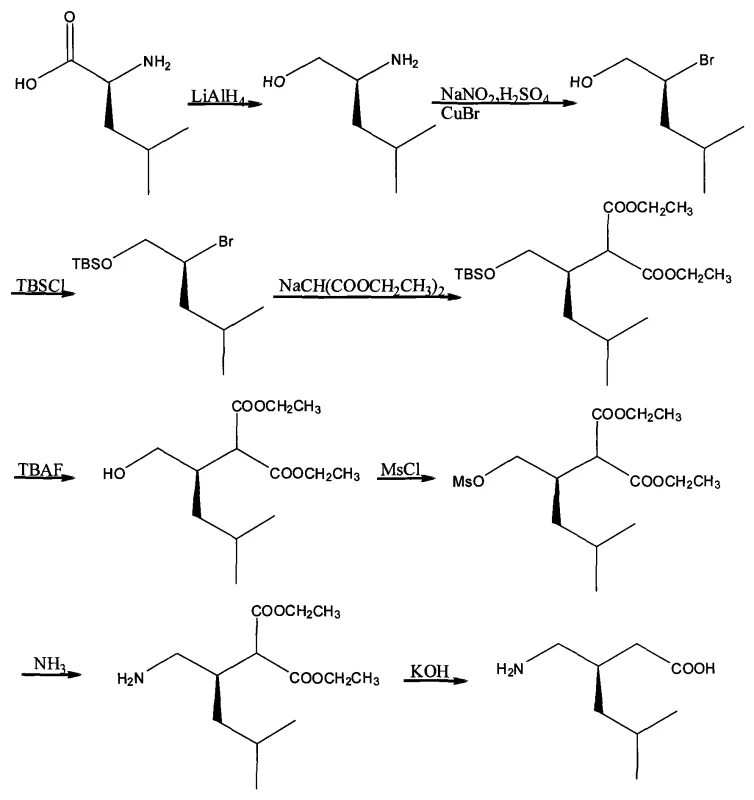
The subsequent alkylation of diethyl malonate with the chiral bromide intermediate is the pivotal step that constructs the carbon skeleton of Pregabalin. The use of a silyl protecting group (TBS) on the hydroxyl moiety ensures chemoselectivity during this alkylation, preventing side reactions at the oxygen atom. Once the malonate is attached, the protecting group is removed, and the resulting alcohol is activated as a mesylate. This activation facilitates a nucleophilic substitution with ammonia, introducing the primary amine group essential for the biological activity of the final drug. The final stages involve hydrolysis of the diester and decarboxylation, which unmask the carboxylic acid and complete the synthesis. Each step in this cascade is designed to maximize yield and minimize impurity formation, resulting in a product that meets stringent stringent purity specifications required for pharmaceutical applications.
How to Synthesize Lyrica Intermediates Efficiently
Executing this synthesis requires precise control over reaction parameters to maintain the high optical purity and yield promised by the patent. The process involves a sequence of eight distinct chemical transformations, each optimized for scalability and safety. From the initial reduction of the amino acid to the final decarboxylation, every step contributes to the overall efficiency of the route. The detailed standardized synthesis steps for implementing this chiral pool strategy are outlined in the guide below.
- Reduce S-(+)-leucine to S-2-amino-4-methylpentanol using LiAlH4 in THF.
- Convert the amino alcohol to S-2-bromo-4-methylpentanol via diazotization with NaNO2/H2SO4 and substitution with CuBr.
- Protect the hydroxyl group with TBSCl to form the silyl ether, followed by alkylation with diethyl malonate.
- Deprotect the silyl group using TBAF, mesylate the alcohol, and perform ammonolysis to introduce the amine.
- Hydrolyze the diester and decarboxylate under acidic or basic conditions to yield the final Lyrica acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of the synthesis method described in CN101585778A translates into tangible strategic benefits that extend beyond simple chemistry. The shift away from hazardous azide chemistry significantly lowers the barrier to entry for manufacturing facilities, as it reduces the need for specialized blast-proof infrastructure and complex effluent treatment systems. This simplification of the safety profile allows for more flexible production scheduling and reduces the risk of unplanned shutdowns due to safety incidents. Furthermore, the reliance on S-(+)-leucine, a commodity chemical produced via fermentation, ensures a stable and predictable raw material supply, insulating the supply chain from the volatility often associated with specialized chiral reagents.
- Cost Reduction in Manufacturing: The elimination of chiral resolution steps effectively doubles the theoretical yield of the process compared to racemic routes, leading to substantial savings in raw material consumption. Additionally, the removal of toxic sodium azide reduces waste disposal costs and the expense of safety monitoring equipment. The streamlined sequence of reactions minimizes solvent usage and energy consumption per kilogram of product, driving down the overall cost of goods sold (COGS) significantly.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials like S-(+)-leucine and diethyl malonate, the process mitigates the risk of supply disruptions caused by the scarcity of exotic catalysts. The robustness of the reaction conditions allows for consistent batch-to-batch quality, reducing the likelihood of failed batches that can delay shipments. This reliability is crucial for maintaining continuous production lines and meeting the just-in-time delivery requirements of major pharmaceutical clients.
- Scalability and Environmental Compliance: The absence of heavy metal catalysts and explosive reagents simplifies the regulatory approval process for new manufacturing sites. The process generates less hazardous waste, aligning with increasingly strict environmental regulations and corporate sustainability goals. This eco-friendly profile enhances the marketability of the final product and reduces the long-term liability associated with chemical manufacturing, making it an attractive option for sustainable pharmaceutical intermediates production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation, providing clarity on the operational advantages.
Q: Why is the S-(+)-leucine route preferred over azide-based synthesis for Lyrica?
A: The azide-based route involves highly toxic and potentially explosive sodium azide, posing significant safety risks and waste disposal challenges. The S-(+)-leucine route utilizes a safe, naturally occurring chiral pool material, eliminating these hazards while ensuring high optical purity without the need for resolution.
Q: How does this process improve supply chain reliability for Pregabalin intermediates?
A: By starting from S-(+)-leucine, a widely available and inexpensive commodity amino acid, the process reduces dependency on specialized chiral catalysts or difficult-to-source resolving agents. This ensures a stable raw material supply and simplifies procurement logistics for large-scale manufacturing.
Q: What are the key purification advantages of this synthetic method?
A: The method avoids the formation of racemic mixtures that require costly and yield-limiting resolution steps. Furthermore, the intermediates are designed to be easily purified via standard extraction and crystallization techniques, leading to a cleaner impurity profile and higher overall process efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lyrica Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has extensively evaluated the methodology presented in CN101585778A and possesses the technical capability to implement this chiral pool strategy at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of monitoring every critical quality attribute, guaranteeing that our Lyrica intermediates meet the stringent purity specifications demanded by top-tier drug manufacturers.
We invite you to collaborate with us to optimize your supply chain for Pregabalin production. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can improve your margins. Please contact our technical procurement team today to request specific COA data and discuss route feasibility assessments for your upcoming projects. Let us be your partner in delivering high-quality, cost-effective solutions for the next generation of CNS therapeutics.
