Scalable Asymmetric Synthesis of Pregabalin: A Three-Step Route for High-Purity API Manufacturing
The pharmaceutical landscape for neurological therapeutics continues to evolve, driven by the demand for more efficient and sustainable manufacturing processes for high-volume drugs like Pregabalin. Patent CN103833562A introduces a transformative methodology for the asymmetric synthesis of this critical GABA analogue, addressing long-standing inefficiencies in legacy production routes. This technical disclosure outlines a novel three-step sequence that leverages a Knoevenagel condensation followed by a chiral catalytic nitro-Michael addition, culminating in a sophisticated one-pot hydrolysis and reduction cascade. For industry stakeholders, this represents a significant paradigm shift away from resolution-dependent strategies, offering a pathway to higher theoretical yields and reduced environmental burden. The robustness of this approach lies in its ability to generate the chiral center early in the synthesis with exceptional stereocontrol, utilizing accessible starting materials such as diethyl malonate and 3-methylbutyraldehyde. By integrating these chemical innovations, manufacturers can achieve a final product purity ranging from 98.1% to 99.7%, with enantiomeric excess values reaching as high as 99.6% under optimized conditions. This level of precision not only meets stringent regulatory standards for active pharmaceutical ingredients but also streamlines the supply chain by minimizing the number of isolation and purification stages required.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Pregabalin has been plagued by inherent inefficiencies associated with racemic synthesis followed by chiral resolution. Traditional routes, such as those depicted in earlier literature, often involve the preparation of a racemic intermediate which must then be separated, inherently discarding up to 50% of the produced material as the unwanted enantiomer. Furthermore, alternative strategies employing enzymatic hydrolysis, while theoretically elegant, frequently suffer from practical drawbacks including low conversion rates ranging from 17.5% to 38.6% and the necessity for iterative racemization cycles to improve yield. 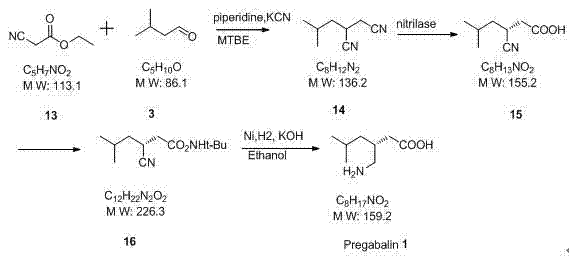 Additionally, some established pathways rely on the use of hazardous reagents such as potassium cyanide during the formation of nitrile intermediates, posing significant safety and environmental compliance challenges for large-scale facilities. Routes utilizing chiral auxiliaries, as illustrated in other comparative schemes, introduce further complexity through extended reaction sequences and the requirement for expensive, stoichiometric chiral reagents that must be recovered and recycled.
Additionally, some established pathways rely on the use of hazardous reagents such as potassium cyanide during the formation of nitrile intermediates, posing significant safety and environmental compliance challenges for large-scale facilities. Routes utilizing chiral auxiliaries, as illustrated in other comparative schemes, introduce further complexity through extended reaction sequences and the requirement for expensive, stoichiometric chiral reagents that must be recovered and recycled. 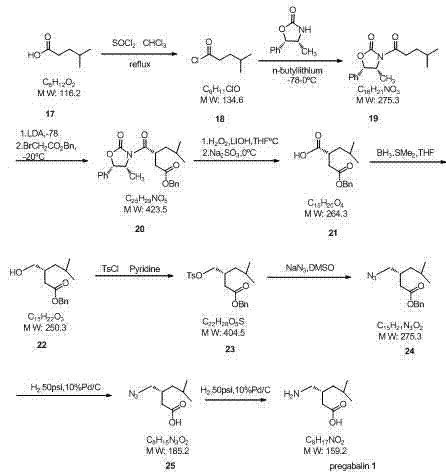 These conventional methods collectively contribute to elevated production costs, increased waste generation, and prolonged lead times, creating substantial bottlenecks for procurement and supply chain teams aiming to secure reliable pharmaceutical intermediates supplier partnerships.
These conventional methods collectively contribute to elevated production costs, increased waste generation, and prolonged lead times, creating substantial bottlenecks for procurement and supply chain teams aiming to secure reliable pharmaceutical intermediates supplier partnerships.
The Novel Approach
In stark contrast to these legacy methodologies, the novel approach detailed in the patent utilizes a direct asymmetric construction of the carbon skeleton, effectively bypassing the need for resolution entirely. The core of this innovation is the strategic application of a chiral catalyst during the addition of nitromethane to an unsaturated diester intermediate, a reaction that establishes the critical stereocenter with high fidelity. 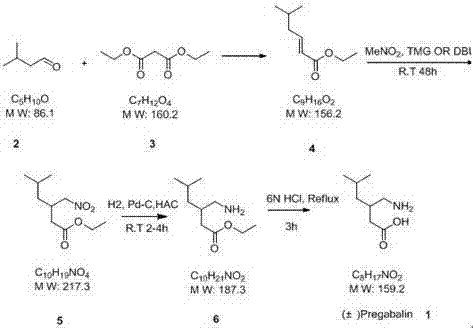 This route begins with a straightforward Knoevenagel condensation to form the alkene precursor, followed by the key asymmetric step which can achieve ee values exceeding 99% when optimized with specific quinine-derived catalysts. The subsequent transformation involves a remarkable tandem process where hydrolysis, decarboxylation, and nitro-group reduction occur simultaneously under acidic reflux conditions. This consolidation of three distinct chemical transformations into a single operational step drastically reduces solvent consumption, energy usage, and processing time. By eliminating the wasteful resolution step and avoiding toxic cyanide reagents, this method offers a cleaner, safer, and more economically viable pathway for the cost reduction in API manufacturing, aligning perfectly with modern green chemistry principles and industrial scalability requirements.
This route begins with a straightforward Knoevenagel condensation to form the alkene precursor, followed by the key asymmetric step which can achieve ee values exceeding 99% when optimized with specific quinine-derived catalysts. The subsequent transformation involves a remarkable tandem process where hydrolysis, decarboxylation, and nitro-group reduction occur simultaneously under acidic reflux conditions. This consolidation of three distinct chemical transformations into a single operational step drastically reduces solvent consumption, energy usage, and processing time. By eliminating the wasteful resolution step and avoiding toxic cyanide reagents, this method offers a cleaner, safer, and more economically viable pathway for the cost reduction in API manufacturing, aligning perfectly with modern green chemistry principles and industrial scalability requirements.
Mechanistic Insights into Chiral Nitro-Michael Addition and Tandem Reduction
The mechanistic elegance of this synthesis centers on the asymmetric nitro-Michael addition, where the choice of chiral catalyst dictates the stereochemical outcome of the reaction. The patent highlights the efficacy of chiral guanidine, thiourea, and particularly quinine alkaloid derivatives in facilitating this transformation. When a catalyst such as phenylbenzene hydroquinine pyrimidine ester is employed, it creates a highly organized transition state that favors the attack of the nitromethane anion on one specific face of the electrophilic double bond. This precise spatial control ensures that the resulting nitro-diester intermediate possesses the desired (S)-configuration with minimal formation of the (R)-isomer. The reaction proceeds in nitromethane solvent, which acts both as the reactant and the medium, simplifying the workup procedure. Following the establishment of chirality, the process moves to a critical tandem sequence involving acid-catalyzed hydrolysis of the ester groups, thermal decarboxylation of the resulting beta-keto acid, and concomitant reduction of the nitro group to the primary amine using zinc powder in hydrochloric acid. This multi-component cascade is kinetically balanced to prevent the accumulation of unstable intermediates, ensuring a smooth conversion to the final amino acid structure.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing the formation of side products typically associated with stepwise syntheses. The direct nature of the asymmetric addition reduces the likelihood of racemization, a common issue in base-catalyzed enolate chemistries. Furthermore, the one-pot final step limits the exposure of reactive intermediates to external contaminants and reduces the number of physical handling operations where cross-contamination could occur. The use of zinc and hydrochloric acid for reduction is a classic, robust method that effectively converts the nitro group without affecting the newly formed carboxylic acid or the alkyl chain. The resulting crude product can be purified through simple acid-base extraction and crystallization, leveraging the zwitterionic nature of Pregabalin to separate it from non-amphoteric organic impurities. This rigorous control over the reaction pathway ensures that the final API meets the stringent purity specifications required for global regulatory submission, with reported chemical purity consistently above 98%.
How to Synthesize Pregabalin Efficiently
The execution of this synthesis requires careful attention to catalyst loading and temperature control during the asymmetric addition step to maximize enantioselectivity. The process is designed to be operationally simple, utilizing standard reactor equipment capable of handling reflux conditions and exothermic additions. Detailed standard operating procedures for each stage, including specific molar ratios, solvent choices, and workup protocols, are essential for reproducing the high yields and purity reported in the patent documentation. For a comprehensive guide on the exact experimental parameters and troubleshooting tips for scaling this reaction, please refer to the standardized synthesis steps provided below.
- Perform Knoevenagel condensation between diethyl malonate and 3-methylbutyraldehyde using an organic weak base to form the unsaturated diester intermediate.
- Execute asymmetric nitro-Michael addition with nitromethane in the presence of a chiral catalyst (e.g., quinine derivative) to establish the stereocenter.
- Conduct simultaneous hydrolysis, decarboxylation, and reduction under acidic conditions with zinc powder to yield crude Pregabalin, followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this asymmetric synthesis route presents a compelling value proposition centered on cost efficiency and supply security. By fundamentally altering the molecular construction strategy, the process eliminates the intrinsic 50% yield loss associated with chiral resolution, effectively doubling the theoretical output from the same quantity of raw materials. This dramatic improvement in atom economy translates directly into significant cost savings on starting materials and waste disposal, addressing the critical need for cost reduction in pharmaceutical intermediates manufacturing. Moreover, the avoidance of expensive chiral resolving agents and stoichiometric chiral auxiliaries further lowers the bill of materials, making the final product more price-competitive in the generic market. The simplified workflow, characterized by fewer isolation steps and a consolidated final transformation, reduces the overall cycle time, thereby enhancing the responsiveness of the supply chain to market fluctuations.
- Cost Reduction in Manufacturing: The elimination of the resolution step is the primary driver for cost optimization, as it removes the need to process and discard half of the synthesized material. Additionally, the use of readily available commodity chemicals like diethyl malonate and nitromethane, rather than specialized chiral pool starting materials, stabilizes raw material costs and mitigates supply risk. The consolidation of hydrolysis, decarboxylation, and reduction into a single pot significantly lowers utility costs by reducing heating and cooling cycles, as well as minimizing solvent usage and recovery loads. These factors combine to create a leaner manufacturing process that offers substantial margin improvements without compromising on quality.
- Enhanced Supply Chain Reliability: Reliance on easily sourced, non-proprietary starting materials ensures a robust and resilient supply chain that is less susceptible to disruptions from single-source vendors. The process does not depend on complex biocatalysts or sensitive enzymes that require cold-chain logistics or have limited shelf lives, simplifying inventory management. The robustness of the chemical steps, which tolerate standard industrial conditions, allows for flexible production scheduling and easier technology transfer between manufacturing sites. This reliability is crucial for reducing lead time for high-purity APIs, ensuring that downstream formulation partners receive their materials on schedule.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, utilizing reaction types that are well-understood and easily managed in large-scale reactors, facilitating the commercial scale-up of complex pharmaceutical intermediates. The absence of highly toxic reagents like potassium cyanide simplifies environmental health and safety (EHS) compliance, reducing the regulatory burden and the cost of specialized waste treatment. The improved atom efficiency results in a lower E-factor (mass of waste per mass of product), aligning with corporate sustainability goals and reducing the environmental footprint of the manufacturing operation. This green chemistry profile enhances the brand reputation of the manufacturer and facilitates smoother regulatory approvals in environmentally conscious markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this specific asymmetric synthesis technology. The answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these details is essential for R&D teams assessing the technical fit and for commercial teams negotiating supply agreements.
Q: What is the enantiomeric excess (ee) achievable with this synthetic route?
A: The patent data indicates that by optimizing the chiral catalyst, specifically using phenylbenzene hydroquinine pyrimidine ester, the ee value of the key intermediate can reach up to 99.7%, resulting in a final product ee value of 99.6%.
Q: Does this process require chiral resolution steps?
A: No, this method utilizes asymmetric catalysis to directly generate the target chiral intermediate, completely eliminating the need for traditional chiral resolution steps that typically result in 50% material loss.
Q: What are the primary advantages regarding reaction conditions?
A: The process operates under relatively mild conditions, avoiding the use of highly toxic reagents like potassium cyanide found in enzymatic routes, and consolidates hydrolysis, decarboxylation, and reduction into a single operational unit.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pregabalin Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting advanced synthetic routes like the one described in CN103833562A to maintain competitiveness in the global pharmaceutical market. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial plant is seamless and efficient. We are committed to delivering high-purity Pregabalin that adheres to the most rigorous international quality standards, supported by our state-of-the-art rigorous QC labs and comprehensive analytical capabilities. Our facility is equipped to handle the specific requirements of asymmetric catalysis and tandem reactions, guaranteeing consistent batch-to-batch quality and stringent purity specifications for our clients.
We invite you to collaborate with us to optimize your supply chain and leverage the economic benefits of this innovative manufacturing process. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this route can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to make informed decisions about your API sourcing strategy. By partnering with us, you gain access to a reliable partner dedicated to driving value through chemical innovation and operational excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
