Advanced Manufacturing of Terbutaline Sulfate: A Scalable, Non-Toxic Route for Global Respiratory Therapeutics
The global demand for effective respiratory therapeutics continues to drive innovation in the synthesis of beta-2 adrenergic agonists, with Terbutaline Sulfate remaining a cornerstone treatment for asthma and chronic obstructive pulmonary disease (COPD). A significant technological breakthrough in this domain is detailed in patent CN110981739A, which introduces a highly efficient, four-step preparation method that fundamentally reimagines the synthetic landscape for this critical active pharmaceutical ingredient (API). Unlike legacy processes that rely on cumbersome protection-deprotection sequences and hazardous reagents, this novel approach utilizes 3,5-dihydroxyacetophenone as a direct starting material, leveraging a unique copper-mediated bromination strategy to streamline the entire value chain. For pharmaceutical manufacturers and procurement leaders, this patent represents not just a chemical optimization, but a strategic opportunity to secure a more robust, cost-effective, and environmentally compliant supply of high-purity respiratory intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Terbutaline Sulfate has been plagued by significant operational inefficiencies and safety hazards inherent to traditional organic synthesis pathways. Prominent prior art, such as the route described in US3937838 by the original research manufacturer, necessitates a lengthy sequence involving esterification, benzyl protection, hydrolysis, acylation, and bromination, ultimately requiring the use of virulent diazomethane. Diazomethane is notoriously unstable and explosive, posing severe safety risks that complicate facility licensing and increase insurance premiums for manufacturing sites. Furthermore, other established methods, including those developed by research institutes in Shanghai, rely on dangerous metal reagents like methyllithium and toxic selenium oxide. These reagents not only introduce high costs due to their specialized handling requirements but also generate complex waste streams that are difficult and expensive to treat, thereby inflating the total cost of ownership for the final API.
The Novel Approach
In stark contrast to these convoluted legacy routes, the methodology disclosed in CN110981739A offers a streamlined, direct functionalization strategy that dramatically simplifies the molecular construction of Terbutaline Sulfate. By selecting 3,5-dihydroxyacetophenone as the foundational building block, the inventors have successfully eliminated the need for hydroxyl protection groups, which traditionally add at least two synthetic steps (protection and deprotection) to the process. The core innovation lies in the direct alpha-bromination of the ketone using Copper(II) Bromide (CuBr2), a reagent that is both inexpensive and significantly safer to handle than elemental bromine or N-bromosuccinimide in this specific context. This approach collapses the synthetic timeline, reduces solvent consumption, and avoids the use of high-pressure hydrogenation equipment often required for debenzylation in older routes, presenting a clear path toward leaner, more agile manufacturing operations.
Mechanistic Insights into CuBr2-Mediated Direct Bromination and Epoxide Formation
From a process chemistry perspective, the most compelling aspect of this patent is the chemoselective bromination achieved in the first step without protecting the phenolic hydroxyl groups. Typically, phenols are highly reactive towards electrophilic aromatic substitution, which would lead to ring bromination rather than the desired alpha-bromination of the acetyl side chain. However, the use of Copper(II) Bromide in a mixed solvent system of chloroform and ethyl acetate at controlled temperatures (40-80°C) facilitates a specific radical or Lewis-acid mediated mechanism that targets the alpha-carbon of the ketone. This selectivity is crucial for maintaining high purity profiles, as it minimizes the formation of ring-brominated impurities that are difficult to separate downstream. The subsequent transformation involves a sophisticated tandem reaction where Sodium Borohydride reduces the ketone to an alcohol, which immediately undergoes intramolecular nucleophilic substitution to form the epoxide ring under basic conditions (NaOH), creating the critical 2-(3,5-dihydroxyphenyl)oxirane intermediate with high stereochemical integrity.
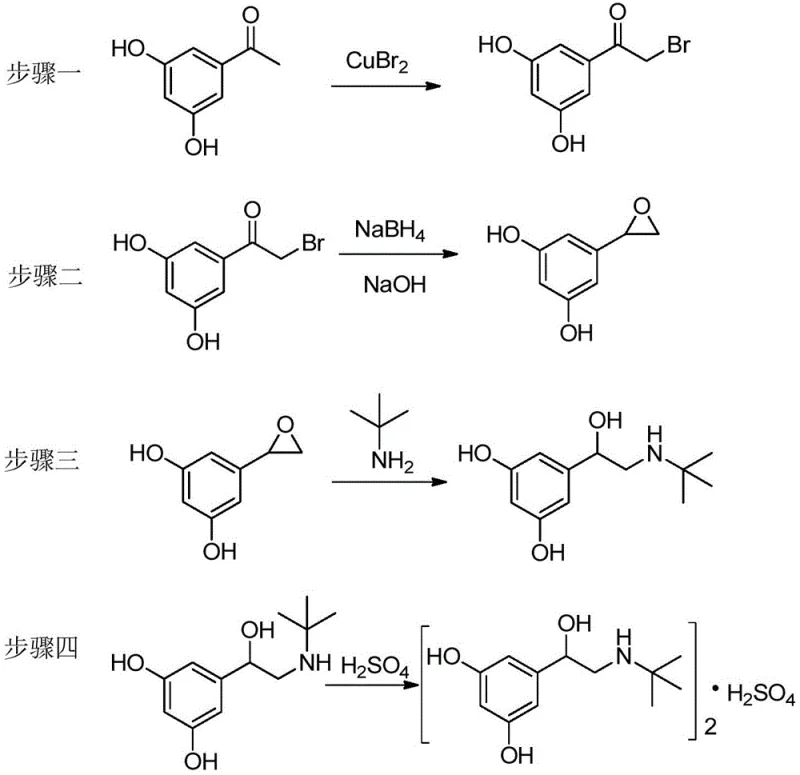
Following the formation of the epoxide, the synthesis proceeds via a regioselective ring-opening reaction with tert-butylamine. This step is pivotal as it installs the essential amine functionality required for beta-adrenergic activity. The reaction is conducted in acetonitrile at moderate temperatures (25-80°C), ensuring that the nucleophilic attack occurs primarily at the less hindered carbon of the epoxide ring, thereby preserving the chiral center adjacent to the aromatic ring. This mechanistic precision is vital for R&D directors focused on impurity control, as it limits the generation of regioisomers that could compromise the biological efficacy of the final drug product. The final salification with sulfuric acid is a straightforward crystallization process that yields the stable sulfate salt, ready for formulation, demonstrating a complete process design that prioritizes both chemical efficiency and product quality.
How to Synthesize Terbutaline Sulfate Efficiently
The implementation of this synthesis route requires precise control over reaction parameters to maximize yield and minimize impurity formation, particularly during the exothermic reduction and cyclization phases. The patent outlines a robust protocol that begins with the dissolution of Copper(II) Bromide in ethyl acetate, followed by the controlled addition of the acetophenone derivative to manage heat evolution. Detailed standard operating procedures for temperature ramping, quenching strategies, and solvent swaps are critical for translating this laboratory-scale success into a GMP-compliant manufacturing process. For technical teams looking to adopt this methodology, understanding the specific stoichiometry—such as the 1:1 to 1:4 molar ratio of substrate to copper bromide—is essential for optimizing raw material usage and ensuring complete conversion before proceeding to the reduction step.
- Perform direct alpha-bromination of 3,5-dihydroxyacetophenone using Copper(II) Bromide (CuBr2) in a chloroform/ethyl acetate solvent system at 40-80°C.
- Execute a tandem reduction and cyclization using Sodium Borohydride (NaBH4) and Sodium Hydroxide (NaOH) in ethanol to form the key epoxide intermediate.
- Conduct ring-opening of the epoxide with tert-butylamine in acetonitrile, followed by salt formation with sulfuric acid to yield the final API intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers transformative potential in terms of cost structure and supply reliability. By fundamentally redesigning the synthetic pathway to eliminate high-risk reagents and reduce the total number of unit operations, manufacturers can achieve significant operational expenditure (OpEx) reductions. The removal of protection and deprotection steps alone translates to massive savings in solvent procurement, waste disposal fees, and reactor occupancy time, allowing for higher throughput without capital expansion. Furthermore, the reliance on commodity chemicals like Copper(II) Bromide and Sodium Borohydride, rather than specialized organometallics, insulates the supply chain from the volatility often seen in the market for exotic reagents, ensuring consistent production scheduling and delivery performance.
- Cost Reduction in Manufacturing: The elimination of benzyl protection groups removes the need for expensive benzyl halides and the subsequent hydrogenation catalysts (like Palladium on Carbon) required for their removal. This simplification drastically reduces the bill of materials (BOM) and lowers the energy intensity of the process by avoiding high-pressure hydrogenation reactors. Additionally, the avoidance of toxic reagents like diazomethane eliminates the need for specialized scrubbing systems and hazardous waste incineration, leading to substantial indirect cost savings in environmental compliance and facility maintenance.
- Enhanced Supply Chain Reliability: Starting from 3,5-dihydroxyacetophenone leverages a widely available bulk chemical feedstock, reducing dependency on custom-synthesized intermediates that may have single-source supply risks. The robustness of the reaction conditions, which do not require cryogenic temperatures or inert atmosphere gloveboxes for every step, makes the process highly transferable across different manufacturing sites globally. This flexibility ensures that supply continuity can be maintained even if one production facility faces disruptions, providing a strategic buffer for pharmaceutical companies managing global inventory levels.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to traditional routes, aligning with modern green chemistry principles and increasingly stringent environmental regulations. The use of ethanol and ethyl acetate as primary solvents facilitates easier solvent recovery and recycling, further enhancing the sustainability profile of the manufacturing operation. This environmental advantage not only reduces regulatory friction but also appeals to end-markets and investors who prioritize sustainable sourcing and eco-friendly production methodologies in their supply chain audits.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and validation of this advanced synthesis route for Terbutaline Sulfate. These answers are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on safety, scalability, and quality control aspects. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: How does this new route improve safety compared to traditional Terbutaline synthesis?
A: This method eliminates the use of highly toxic and explosive reagents such as diazomethane and dangerous organometallics like methyllithium or selenium oxide found in older patents. By utilizing Copper(II) Bromide and Sodium Borohydride, the process operates under mild conditions without high-pressure hydrogenation, significantly reducing operational risk and hazardous waste generation.
Q: What are the cost advantages of starting from 3,5-dihydroxyacetophenone?
A: Starting from 3,5-dihydroxyacetophenone allows for a direct functionalization strategy that bypasses the need for hydroxyl protection and deprotection steps (such as benzylation) required in conventional routes. This reduction in synthetic steps directly lowers solvent consumption, reagent costs, and processing time, resulting in substantial overall manufacturing cost reductions.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for industrial scalability. It avoids complex purification steps associated with removing heavy metal catalysts or protecting groups. The reaction conditions are mild (temperatures between 0°C and 80°C) and utilize common organic solvents like ethanol and ethyl acetate, making it highly adaptable for multi-ton production facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Terbutaline Sulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a more efficient synthetic route requires a partner with deep technical expertise and proven scale-up capabilities. As a leading CDMO and fine chemical manufacturer, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN110981739A are fully realized in a commercial setting. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required for this copper-mediated chemistry, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest pharmacopeial standards for respiratory drug intermediates.
We invite global pharmaceutical partners to collaborate with us to leverage this innovative technology for their Terbutaline Sulfate supply needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this non-toxic route for your specific volume requirements. We encourage you to contact us today to discuss specific COA data, route feasibility assessments, and how we can jointly optimize the supply chain for this critical life-saving medication.
