Advanced Catalytic Hydrogenation Strategy for Commercial Scale-up of High-Purity Terbutaline Sulfate
Advanced Catalytic Hydrogenation Strategy for Commercial Scale-up of High-Purity Terbutaline Sulfate
The pharmaceutical landscape for respiratory therapeutics continues to demand robust, scalable, and environmentally sustainable manufacturing processes for critical active pharmaceutical ingredients. A significant technological breakthrough in this domain is detailed in patent CN107513023B, which outlines a novel preparation method for terbutaline sulfate, a vital short-acting β2-receptor agonist used extensively in the management of Chronic Obstructive Pulmonary Disease (COPD) and asthma. This patent introduces a paradigm shift from traditional multi-step purification protocols to a streamlined, acidic catalytic hydrogenation route that inherently stabilizes the sensitive polyphenol structure of the molecule. By leveraging specific reaction conditions involving organic acid solvents and quantitative salification, this method addresses long-standing industry challenges related to oxidative degradation, inorganic salt contamination, and solvent consumption. For R&D directors and procurement specialists seeking a reliable terbutaline sulfate supplier, understanding the mechanistic advantages of this route is crucial for securing a supply chain that delivers both high purity and cost efficiency.
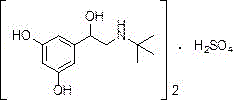
The core innovation lies in the direct transformation of benzyl-protected precursors into the final sulfate salt without isolating the unstable free base in an aqueous or alkaline environment. Conventional synthesis routes often struggle with the susceptibility of the 1,3-resorcinol moiety to oxidation, particularly during the high-temperature distillation or pH adjustment steps required to isolate the free amine. The methodology described in CN107513023B circumvents these pitfalls by maintaining an acidic milieu throughout the critical deprotection phase. This approach not only enhances the chemical stability of the intermediate but also facilitates a telescoped process where debenzylation and salt formation occur in a highly controlled sequence. The result is a manufacturing protocol that yields terbutaline sulfate with exceptional purity levels, often exceeding 99%, while drastically simplifying the downstream processing requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of terbutaline sulfate has relied on routes that introduce significant operational complexities and quality risks. Prior art, such as the methods disclosed in SE335359 and US3937838, typically involves the hydrogenation of dibenzyloxy precursors followed by a cumbersome workup procedure. In these traditional processes, the reaction mixture is often subjected to alkaline conditions to adjust the pH for salt formation, which inevitably introduces large quantities of inorganic salts, such as sodium sulfate, into the system. The presence of these inorganic impurities complicates the purification process, necessitating extensive recrystallization steps using large volumes of methanol to meet pharmacopeial standards for residue on ignition. Furthermore, the isolation of the free base terbutaline often requires evaporation of solvents under reduced pressure, sometimes at elevated temperatures, which creates a high risk of oxidative degradation for the electron-rich resorcinol ring. This oxidative instability not only lowers the overall yield but also generates difficult-to-remove colored impurities that compromise the aesthetic and chemical quality of the final API.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a strategic combination of organic acid solvents and precise stoichiometric control to overcome these historical limitations. Instead of relying on aqueous workups and pH adjustments with alkalis, the new method employs solvents like glacial acetic acid, formic acid, or propionic acid as the medium for catalytic hydrogenation. This choice of solvent serves a dual purpose: it acts as an effective medium for the metal-catalyzed removal of benzyl protecting groups and simultaneously provides an acidic environment that protects the phenolic hydroxyl groups from oxidation. Following the hydrogenation step, the process bypasses the isolation of the free base entirely. Instead, a quantitative amount of concentrated sulfuric acid is added directly to the filtrate. This direct salification ensures that the terbutaline is immediately converted into its stable sulfate salt form, preventing any exposure to conditions that might trigger degradation. The subsequent recovery of the organic acid solvent and crystallization from alcohol yields a product of superior quality with minimal impurity profiles, representing a substantial advancement in process chemistry.
Mechanistic Insights into Acidic Catalytic Hydrogenation and Salification
The success of this synthetic route hinges on the intricate interplay between the solvent system, the catalyst, and the reaction kinetics. The use of organic acids like acetic acid is not merely a solvent choice but a critical mechanistic feature. In the presence of a metal catalyst such as palladium on carbon (Pd/C), nickel (Ni), or tin chloride (SnCl4), the benzyl ether bonds in the precursor compounds are cleaved via hydrogenolysis. The acidic nature of the solvent protonates the amino group of the intermediate as it forms, which helps to solubilize the species and prevents it from adsorbing too strongly onto the catalyst surface in a way that might promote side reactions. More importantly, the low pH environment suppresses the formation of phenoxide ions, which are highly susceptible to single-electron transfer oxidation processes that lead to quinone-like impurities. By keeping the phenolic groups protonated, the process effectively 'locks' the molecule in a stable state during the most vulnerable phase of the synthesis.
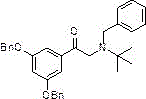
Furthermore, the mechanism of salt formation in this process is engineered for precision. Traditional methods often rely on titrating the pH of an aqueous solution to a specific range (e.g., pH 5.5), which is prone to local over-acidification or under-acidification, leading to inconsistent salt stoichiometry. The patented method dictates the addition of concentrated sulfuric acid in a molar ratio of approximately 0.495 to 0.505 equivalents relative to the starting amine precursor. This near-perfect stoichiometric match ensures the formation of the 2:1 terbutaline-to-sulfate salt structure without excess free acid or free base. The absence of excess reagents means there are no competing ions to interfere with the crystal lattice formation during the final crystallization step. Consequently, the product precipitates as a white crystalline substance with high structural integrity and minimal inclusion of mother liquor impurities. This mechanistic control over the salification step is the key driver behind the observed improvements in both yield and purity, offering a robust solution for the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Terbutaline Sulfate Efficiently
The implementation of this advanced synthesis route requires careful attention to reaction parameters to maximize the benefits of the acidic hydrogenation strategy. The process begins with the dissolution of the benzyl-protected precursor, either the ketone (Compound I) or the alcohol (Compound II), in a suitable organic acid solvent. A heterogeneous metal catalyst is then introduced, and the system is purged with hydrogen to initiate the debenzylation reaction under mild thermal conditions. Once the reduction is complete, the catalyst is removed via filtration, and the critical salification step is performed by adding a pre-calculated amount of sulfuric acid. The solvent is then recovered via distillation, and the residue is treated with an alcohol anti-solvent to induce crystallization. This sequence minimizes unit operations and reduces the time the product spends in solution, thereby limiting opportunities for degradation. For a detailed breakdown of the specific operational parameters, including temperature ranges, catalyst loading ratios, and stirring times, please refer to the standardized synthesis guide below.
- Dissolve the benzyl-protected precursor (Compound I or II) in an organic acid solvent such as glacial acetic acid and add a metal catalyst like Pd/C or Raney Nickel.
- Perform catalytic hydrogenation under normal pressure at 40-60°C to achieve simultaneous debenzylation and reduction, yielding the free base intermediate in situ.
- Filter the catalyst, add a stoichiometric amount of concentrated sulfuric acid to the filtrate, recover the solvent, and crystallize the product using alcohol solvents.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented manufacturing process offers profound advantages for procurement managers and supply chain leaders focused on cost reduction in pharmaceutical intermediates manufacturing. The primary economic benefit stems from the drastic simplification of the post-reaction workup. By eliminating the need for aqueous extraction, pH adjustment with alkalis, and extensive recrystallization cycles, the process significantly reduces the consumption of auxiliary chemicals and solvents. The ability to recover and recycle the organic acid solvent, such as acetic acid, further drives down raw material costs and minimizes waste disposal fees. Moreover, the high yield and purity achieved in a single pass reduce the need for reprocessing batches, which directly translates to higher throughput and better asset utilization for manufacturing facilities. These efficiencies collectively contribute to a more competitive cost structure without compromising on the stringent quality standards required for respiratory APIs.
- Cost Reduction in Manufacturing: The elimination of inorganic salt byproducts is a major cost-saving factor. Traditional routes that generate sodium sulfate or similar salts require expensive wastewater treatment and disposal processes to meet environmental regulations. By avoiding the introduction of these salts through the quantitative acid addition method, the facility avoids these downstream treatment costs entirely. Additionally, the high reaction efficiency means that less starting material is wasted, optimizing the cost of goods sold (COGS). The process also operates at relatively mild temperatures and normal hydrogen pressure, which reduces energy consumption compared to high-pressure or high-temperature alternatives. These cumulative factors create a leaner, more cost-effective production model that enhances margin potential for the final API.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by complex processes that have multiple failure points. The robustness of this acidic hydrogenation route enhances reliability by reducing the number of critical control points. Since the process does not rely on sensitive pH titrations or difficult separations of oil-water mixtures, the risk of batch failure due to operational error is minimized. The use of commercially available and stable catalysts like Pd/C ensures that raw material sourcing is straightforward and not subject to the volatility of exotic reagent markets. Furthermore, the shortened production cycle time allows for faster turnaround on orders, enabling suppliers to respond more agilely to fluctuations in market demand for COPD medications. This operational resilience is a key attribute for any reliable terbutaline sulfate supplier aiming to serve global pharmaceutical markets.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies environmental challenges, but this method is inherently greener. The reduction in solvent usage and the ability to recycle the reaction medium align with modern green chemistry principles, reducing the facility's overall environmental footprint. The absence of heavy metal contaminants or difficult-to-treat saline waste streams simplifies regulatory compliance and permitting for manufacturing sites. As global regulations on pharmaceutical manufacturing emissions become stricter, having a process that naturally generates less waste provides a significant strategic advantage. The scalability is further supported by the use of standard hydrogenation equipment found in most fine chemical plants, meaning that capacity can be expanded from pilot scale to multi-ton production without requiring specialized or custom-built reactor infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of terbutaline sulfate using this advanced methodology. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, offering clarity on how this process differentiates itself from legacy manufacturing techniques. Understanding these nuances is essential for technical teams evaluating potential partners for API production.
Q: How does the acidic hydrogenation method improve product stability compared to traditional routes?
A: Traditional methods often involve alkaline conditions or aqueous workups that expose the polyphenol structure of terbutaline to oxidative degradation. The patented acidic hydrogenation method maintains the reaction mixture in an acidic environment throughout the process, which significantly stabilizes the resorcinol moiety against oxidation, resulting in higher purity and yield without the need for extensive refining.
Q: What are the advantages of using quantitative sulfuric acid addition in this process?
A: By adding a precise stoichiometric amount of concentrated sulfuric acid (approximately 0.5 equivalents relative to the amine precursor) directly to the reaction filtrate, the process avoids the introduction of inorganic salts typically associated with pH adjustment using alkalis. This eliminates the formation of byproduct salts like sodium sulfate, simplifying the purification process and ensuring the final product meets stringent residue on ignition specifications.
Q: Can the solvents and catalysts be recycled in this manufacturing process?
A: Yes, the process is designed with sustainability in mind. The organic acid solvent, such as acetic acid, can be recovered via distillation after the reaction and salification steps. Additionally, the heterogeneous metal catalysts used, such as palladium on carbon, can be filtered and potentially regenerated or recycled, reducing both raw material costs and environmental waste disposal burdens.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Terbutaline Sulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated acidic hydrogenation technique described in CN107513023B can be seamlessly implemented at an industrial level. We understand that high-purity terbutaline sulfate is not just a chemical commodity but a critical component in life-saving respiratory therapies. Therefore, our manufacturing facilities are equipped with rigorous QC labs and advanced analytical instrumentation to verify that every batch meets stringent purity specifications and complies with international pharmacopeial standards. Our dedication to process optimization ensures that we can deliver this complex intermediate with the consistency and reliability that global pharmaceutical supply chains demand.
We invite procurement directors and R&D leaders to engage with us to explore how this advanced manufacturing route can benefit your specific supply chain needs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential efficiencies of adopting this greener, more efficient synthesis method. We encourage you to contact our technical procurement team today to request specific COA data, discuss route feasibility assessments, and discover how we can support your long-term goals for sustainable and cost-effective API sourcing. Let us collaborate to bring high-quality respiratory medicines to patients worldwide through superior chemical engineering.
