Revolutionizing Terbutaline Production: A Safer, Scalable Suzuki Coupling Strategy for Global Supply Chains
Introduction to Next-Generation Terbutaline Manufacturing
The pharmaceutical industry is constantly seeking more efficient pathways for producing critical bronchodilators like Terbutaline. A significant technological breakthrough has been documented in patent CN113461555A, which details a novel preparation method that fundamentally restructures the synthetic logic of this vital API intermediate. Unlike legacy processes that rely on cumbersome protection-deprotection sequences, this innovation leverages the power of transition metal catalysis to achieve direct carbon-carbon bond formation. By integrating a Suzuki-Miyaura cross-coupling reaction as the core transformation, the process drastically reduces the step count while simultaneously enhancing the safety profile of the manufacturing operation. This report analyzes the technical merits of this approach, offering strategic insights for R&D directors and procurement leaders aiming to optimize their supply chains for respiratory therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Terbutaline has been plagued by inefficiencies inherent in multi-step functional group manipulations. Traditional routes, such as those starting from 3,5-dihydroxybenzoic acid, necessitate a lengthy sequence involving esterification, benzyl protection of phenolic hydroxyls, acylation, and oxidation before the final reduction steps. These conventional pathways are not only operationally complex but also introduce significant safety hazards, particularly during the debenzylation stage which typically requires catalytic hydrogenation with nickel and hydrogen gas under pressure. Furthermore, the reliance on bromination reactions in some prior art methods introduces challenges in controlling regioselectivity and managing hazardous halogenated byproducts. The cumulative effect of these factors is a low overall yield, often reported around 21%, and a high burden on waste treatment systems due to the extensive use of protecting groups.
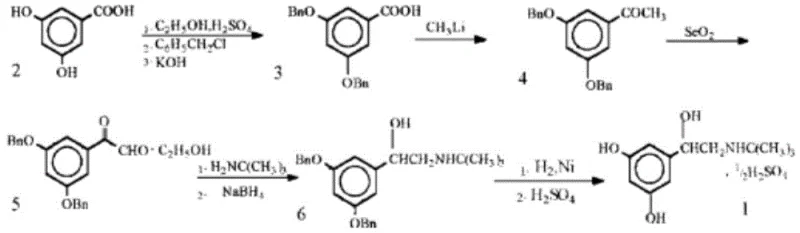
The Novel Approach
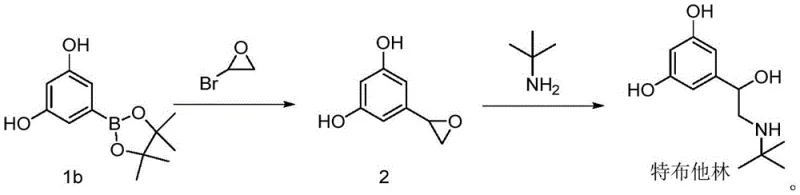
In stark contrast to the convoluted legacy pathways, the methodology disclosed in CN113461555A offers a streamlined alternative that bypasses the need for hydroxyl protection entirely. The core innovation lies in the direct coupling of a boronic acid derivative with a halogenated ethylene oxide, effectively constructing the carbon skeleton in a single catalytic step. This approach eliminates the dangerous catalytic hydrogenation step and the associated high-pressure equipment requirements, thereby markedly improving the safety factor of the production facility. The reaction conditions are notably mild, typically proceeding at temperatures between 80°C and 120°C, which are easily achievable in standard glass-lined or stainless steel reactors. By removing the protection-deprotection cycle, the process not only shortens the timeline but also significantly reduces the consumption of reagents and solvents, leading to a cleaner reaction profile and easier downstream purification.
Mechanistic Insights into Suzuki-Miyaura Cross-Coupling
The heart of this improved synthesis is the palladium-catalyzed Suzuki coupling between the 3,5-dihydroxyphenylboronic acid derivative and the halogenated epoxide. Mechanistically, this involves the oxidative addition of the palladium catalyst to the carbon-halogen bond of the epoxide, followed by transmetallation with the organoboron species activated by a base such as cesium carbonate or potassium phosphate. The subsequent reductive elimination releases the coupled product and regenerates the active Pd(0) catalyst. This cycle is highly robust and tolerant of the free phenolic hydroxyl groups present on the aromatic ring, which is a critical advantage over other cross-coupling methods that might require masking these reactive sites. The choice of ligand and palladium source, such as Pd(PPh3)4 or Pd(dppf)Cl2, plays a pivotal role in maintaining catalyst activity throughout the reaction, ensuring high conversion rates even at moderate temperatures.
From an impurity control perspective, this mechanism offers distinct advantages over electrophilic aromatic substitution routes. Traditional bromination methods often suffer from over-bromination or isomer formation, creating impurities that are structurally similar to the target and difficult to separate. The Suzuki coupling, being a directed cross-coupling event, exhibits superior regioselectivity, primarily generating the desired meta-substituted product. Additionally, the absence of strong reducing agents like lithium aluminum hydride or high-pressure hydrogen minimizes the risk of over-reduction side reactions. The resulting intermediate possesses a high HPLC purity, often exceeding 99%, which simplifies the final crystallization of the sulfate salt and ensures the final API meets stringent pharmacopeial standards for related substances.
How to Synthesize Terbutaline Efficiently
Implementing this advanced synthetic route requires precise control over reaction parameters to maximize the efficiency of the palladium cycle. The process begins with the preparation of the boronic ester precursor, followed by the critical coupling step with the epoxide, and concludes with the ring-opening amination. Each stage demands specific solvent systems and base selections to drive the equilibrium towards the product while suppressing potential side reactions like epoxide polymerization. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for process chemists to replicate these high-yielding results in a pilot or production setting.
- Preparation of 3,5-dihydroxyphenylboronic acid pinacol ester via palladium-catalyzed reaction of 5-haloresorcinol with pinacol diboron.
- Execution of Suzuki coupling between the boronic ester and halogenated ethylene oxide using Pd catalysts and base to form the key intermediate.
- Ring-opening amination of the epoxide intermediate with tert-butylamine followed by salification to yield Terbutaline Sulfate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Suzuki-based route translates into tangible strategic benefits beyond mere technical elegance. The elimination of hazardous unit operations, such as high-pressure hydrogenation, reduces the capital expenditure required for specialized reactor infrastructure and lowers the ongoing costs associated with safety compliance and insurance. Furthermore, the simplification of the synthetic sequence means fewer unit operations, less solvent turnover, and reduced labor hours per kilogram of output. This efficiency gain directly impacts the cost of goods sold (COGS), making the supply of Terbutaline more resilient against market fluctuations in raw material pricing. The use of commercially available and stable starting materials also mitigates the risk of supply disruptions that can occur with exotic or custom-synthesized intermediates.
- Cost Reduction in Manufacturing: The most significant economic driver of this new process is the complete removal of benzyl protecting groups. In traditional synthesis, the reagents for protection and the catalysts for deprotection represent a substantial portion of the material cost. By designing a route that tolerates free hydroxyl groups, the process eliminates these expensive reagent classes entirely. Additionally, the avoidance of nickel-catalyzed hydrogenation removes the need for costly metal recovery processes and the disposal of spent catalyst waste. The overall reduction in step count inherently lowers utility consumption and reactor occupancy time, driving down the manufacturing overhead significantly without compromising on quality.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the complexity of sourcing specialized intermediates. This novel route relies on fundamental building blocks like resorcinol derivatives and pinacol diboron, which are produced at a massive global scale for various industries. This commoditization of raw materials ensures a stable supply base with multiple qualified vendors, reducing the risk of single-source bottlenecks. Moreover, the robustness of the Suzuki reaction conditions allows for flexible manufacturing scheduling, as the process is less sensitive to minor variations in temperature or mixing compared to sensitive hydrogenation or cryogenic reactions, ensuring consistent on-time delivery performance.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden environmental liabilities, particularly regarding solvent waste and heavy metal residues. This methodology utilizes common organic solvents like toluene and dioxane, which have well-established recovery and recycling protocols in modern facilities. The palladium catalyst, while precious, can be effectively scavenged from the final product using standard filtration or adsorption techniques, ensuring the API meets strict heavy metal limits. The reduction in total process mass intensity (PMI) due to fewer steps means less waste generation per kilogram of product, aligning perfectly with green chemistry initiatives and reducing the environmental footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Suzuki coupling technology for Terbutaline production. These answers are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on how this method compares to incumbent technologies in terms of safety, purity, and operational feasibility.
Q: How does the Suzuki coupling route improve safety compared to traditional Terbutaline synthesis?
A: Traditional routes often require catalytic hydrogenation using Nickel and Hydrogen gas, which poses significant safety risks and requires high-pressure equipment. The novel Suzuki route operates under mild atmospheric conditions without hazardous hydrogenation steps.
Q: What are the purity advantages of avoiding benzyl protection groups?
A: By eliminating the need for benzyl protection and subsequent deprotection steps, the process avoids the formation of difficult-to-remove benzyl-related impurities, resulting in significantly higher HPLC purity profiles suitable for API applications.
Q: Is this synthetic route scalable for industrial manufacturing?
A: Yes, the route utilizes robust palladium catalysts and common solvents like toluene and dioxane. The reaction conditions (80-120°C) are easily manageable in standard stainless steel reactors, facilitating seamless commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Terbutaline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven execution capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the Suzuki coupling route are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped to handle palladium-catalyzed reactions safely and efficiently, supported by rigorous QC labs that enforce stringent purity specifications for every batch. We are committed to delivering high-quality pharmaceutical intermediates that empower your drug development timelines and commercial success.
We invite you to engage with our technical team to explore how this optimized synthesis can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic impact of switching to this streamlined process. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to support your decision-making. Contact our technical procurement team today to discuss your supply needs and secure a reliable, cost-effective source of high-purity Terbutaline for your global operations.
