Revolutionizing A82846B Production: Advanced Fermentation Recycling for Commercial Scale-up
Revolutionizing A82846B Production: Advanced Fermentation Recycling for Commercial Scale-up
The pharmaceutical industry's relentless pursuit of efficient antibiotic synthesis has found a significant breakthrough in the realm of glycopeptide intermediates, specifically through the innovations detailed in patent CN109811024B. This pivotal intellectual property discloses a sophisticated method for preparing A82846B, a critical intermediate for the synthesis of the potent antibiotic oritavancin, via an optimized microbial fermentation process. Traditionally, the fermentation of Amycolatopsis orientalis yields a complex mixture of structurally similar compounds, creating a bottleneck in downstream purification and driving up manufacturing costs. However, this novel approach fundamentally alters the economic landscape by introducing a closed-loop recycling system where impurity waste liquids are not discarded but rather chemically transformed and reintroduced into the bioreactor. By leveraging alkaline degradation of extraction by-products, the process achieves a dramatic enhancement in fermentation units and selectively boosts the ratio of the target A82846B component. For global procurement and R&D leaders, this represents a paradigm shift from linear consumption to circular efficiency, offering a pathway to substantially reduce the cost of goods sold (COGS) while ensuring a robust supply chain for high-value anti-infective agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
In the traditional manufacturing landscape of glycopeptide antibiotics, the fermentation of Amycolatopsis orientalis has long been plagued by inherent inefficiencies related to product selectivity and waste management. When cultured using standard media, the microorganism naturally produces a triad of closely related substances: A82846A, A82846B, and A82846C. The structural homology among these three compounds is so profound that separating the desired A82846B from its analogs requires extensive and costly chromatographic interventions. Historically, the fermentation broth contains a suboptimal ratio of A82846B, often overshadowed by the presence of A82846A and A82846C, which are considered impurities in the context of oritavancin synthesis. Consequently, a significant portion of the fermentation output ends up in the extraction waste stream. In conventional workflows, this waste liquid, rich in unutilized biological potential, is typically treated as hazardous effluent or discarded, representing a direct loss of raw materials and a missed opportunity for yield optimization. Furthermore, the low initial titer of the target compound necessitates larger fermentation volumes to meet production quotas, thereby inflating utility costs, increasing the footprint of manufacturing facilities, and complicating the environmental compliance profile of the operation.
The Novel Approach
The methodology outlined in patent CN109811024B introduces a transformative strategy that turns the traditional waste problem into a feedstock solution. Instead of viewing the extraction residue as trash, this innovative process identifies the impurity waste liquid—specifically the fraction containing high concentrations of A82846A and A82846C—as a valuable resource. The core of this novelty lies in the alkaline degradation step, where the waste liquid is subjected to high pH conditions (pH ≥ 11.5) to break down the complex glycopeptide structures into simpler, bioavailable precursors. These degradation products are then strategically fed back into the fermentation medium during subsequent culture cycles. This recycling mechanism effectively tricks the microbial metabolism, providing the Amycolatopsis orientalis strain with a enriched nutrient profile that favors the biosynthesis of A82846B over its analogs. The result is a dual benefit: a drastic reduction in the volume of chemical waste requiring treatment and a simultaneous, significant surge in the fermentation unit of the target molecule. This approach not only streamlines the downstream purification burden by improving the initial component ratio but also aligns perfectly with modern green chemistry principles, making it highly attractive for sustainable manufacturing initiatives.
Mechanistic Insights into Metabolic Recycling and Structural Selectivity
To fully appreciate the technical sophistication of this fermentation optimization, one must examine the structural nuances of the glycopeptide family involved. The challenge in A82846B production stems from the minute chemical differences between the co-produced analogs. As illustrated in the structural comparison below, A82846A, A82846B, and A82846C share an identical heptapeptide backbone and sugar moieties, differing primarily in the halogenation pattern of the aromatic rings. A82846B possesses two chlorine atoms, whereas A82846A has one, and A82846C has none. This subtle variation makes them notoriously difficult to separate via standard crystallization or extraction techniques, often requiring expensive preparative HPLC. The patented process cleverly bypasses this separation bottleneck at the source. By subjecting the mixture of A, B, and C to alkaline hydrolysis, the process likely cleaves specific amide or ester bonds, or perhaps modifies the sugar attachments, generating fragments that the microorganism can re-assimilate. 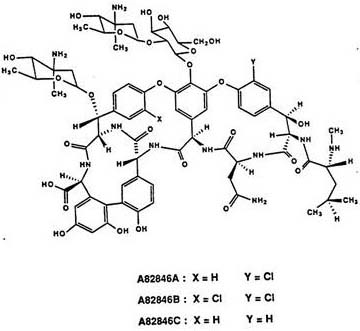
The metabolic re-engineering achieved through this recycling loop suggests that the degradation products serve as superior precursors for the chlorination enzymes present in the Amycolatopsis orientalis strain. When the alkaline degradation liquid is added to the fermentation medium, it appears to shift the metabolic flux towards the fully chlorinated A82846B form. Experimental data from the patent indicates that adding varying concentrations of this recycled liquid can nearly triple the fermentation unit compared to control groups, while simultaneously pushing the component ratio of A82846B from roughly 37% to nearly 60%. This implies that the recycled fragments are not just generic nutrients but are specifically channelled into the biosynthetic pathway of the target antibiotic. For R&D directors, this mechanistic insight offers a powerful lever for process control; by fine-tuning the concentration of the recycled degradation liquid, manufacturers can dynamically adjust the output profile of the bioreactor to maximize the yield of the most valuable component, thereby minimizing the formation of hard-to-remove impurities right from the start.
How to Synthesize A82846B Efficiently
The implementation of this high-yield fermentation strategy requires precise adherence to a sequence of biological and chemical operations designed to maximize the recycling efficiency. The process begins with the preparation of a robust seed culture using optimized media containing maltodextrin and soy flour, followed by the main fermentation cycle where the magic of metabolic recycling occurs. The critical innovation lies in the post-fermentation handling, where the broth is extracted, and the 'waste' fraction is chemically activated before being returned to the start of the cycle. This creates a continuous improvement loop in the production line. For technical teams looking to adopt this methodology, understanding the specific parameters of the alkaline degradation and the timing of the recycle addition is paramount to success. The detailed standardized synthesis steps for implementing this recycling protocol are provided in the guide below.
- Inoculate Amycolatopsis orientalis into a seed culture medium containing maltodextrin, glucose, and soy flour to obtain a mature seed solution.
- Ferment the seed liquid in a production medium, then extract the broth to separate high-purity A82846B product from impurity waste liquid containing A82846A and A82846C.
- Perform alkaline degradation on the impurity waste liquid at pH ≥ 11.5, then recycle the resulting degradation solution back into the fermentation medium to enhance biosynthesis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of the technology described in CN109811024B translates directly into tangible bottom-line improvements and risk mitigation. The primary commercial driver here is the substantial reduction in raw material costs per kilogram of finished A82846B. By recycling the impurity stream, the effective yield of the fermentation process is drastically increased without a proportional increase in the input of expensive fermentation substrates like soy flour or yeast extract. This efficiency gain means that less raw material is required to produce the same amount of active pharmaceutical ingredient (API) intermediate, leading to significant cost savings in the overall manufacturing budget. Furthermore, the reduction in waste volume lowers the operational expenditures associated with environmental compliance and waste disposal, which are increasingly stringent and costly in the global chemical sector. The ability to generate higher titers in the same fermentation vessel volume also implies a reduction in the number of batches required to meet demand, thereby optimizing facility utilization rates and reducing energy consumption per unit of product.
- Cost Reduction in Manufacturing: The elimination of waste and the conversion of by-products into valuable precursors fundamentally alters the cost structure of A82846B production. By recovering materials that were previously lost, the process effectively lowers the variable cost of production. This is particularly critical in the competitive market for glycopeptide antibiotics, where margin pressure is high. The qualitative improvement in yield means that the fixed costs of the facility—such as depreciation, labor, and maintenance—are spread over a larger output of saleable product, driving down the unit cost significantly. Additionally, the improved component ratio reduces the load on downstream purification columns, extending their lifespan and reducing the frequency of expensive resin replacement, which is a major cost center in antibiotic manufacturing.
- Enhanced Supply Chain Reliability: Supply continuity is a top priority for pharmaceutical buyers, and this fermentation optimization enhances reliability by decoupling production capacity from raw material volatility. Since the process generates more product per batch, manufacturers can maintain inventory levels with fewer production runs, reducing the risk of supply disruptions caused by equipment downtime or raw material shortages. The robustness of the Amycolatopsis orientalis strain in the presence of recycled degradation products also suggests a stable and reproducible process, which is essential for regulatory compliance and consistent quality. This stability allows suppliers to offer more reliable lead times and commit to larger volume contracts with confidence, knowing that the process is resilient to minor fluctuations in feedstock quality.
- Scalability and Environmental Compliance: Scaling up fermentation processes is often fraught with challenges, but this recycling method is inherently scalable as it relies on standard unit operations like pH adjustment and filtration. The environmental benefits are equally compelling; by minimizing the discharge of organic-rich waste liquids, the process helps manufacturers meet increasingly rigorous environmental, social, and governance (ESG) targets. This 'green' credential is becoming a key differentiator in supplier selection for multinational corporations committed to sustainability. The reduced environmental footprint not only mitigates regulatory risk but also enhances the brand reputation of the supply chain partners, making the A82846B produced via this method a preferred choice for eco-conscious pharmaceutical developers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced fermentation recycling technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear picture of what potential partners can expect when integrating this methodology into their supply chains. Understanding these details is crucial for evaluating the feasibility and impact of adopting this novel production route for oritavancin intermediates.
Q: How does the alkaline degradation recycling method improve A82846B yield?
A: The process converts impurity components A82846A and A82846C found in waste liquid into precursors that Amycolatopsis orientalis can re-utilize, significantly shifting the metabolic balance towards the desired A82846B product.
Q: What are the critical pH conditions for the impurity degradation step?
A: The alkaline degradation must be performed in an environment with a pH not less than 11.5, preferably between 11.5 and 12.5, to effectively break down the impurities without destroying usable precursors.
Q: Why is separating A82846B from A82846A and C traditionally difficult?
A: These three glycopeptide substances possess extremely similar chemical structures, differing only slightly in chlorination and hydrogen substitution, which makes chromatographic separation energy-intensive and costly without prior fermentation optimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable A82846B Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the complex fermentation and recycling protocols described in patent CN109811024B can be executed with precision at an industrial scale. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced fermentation suites capable of maintaining the stringent purity specifications required for pharmaceutical intermediates. We understand that consistency is key in API manufacturing, and our quality management systems are designed to monitor every critical parameter of the fermentation process, from seed culture viability to the efficiency of the alkaline degradation recycle loop.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cutting-edge technology for their oritavancin projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this recycling method can optimize your budget. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, cost-effective, and high-quality supply of A82846B that meets the demanding standards of the global healthcare market.
