Scalable Aqueous Phase Synthesis of Quinoxaline Intermediates Using Biodegradable Ionic Liquids
Scalable Aqueous Phase Synthesis of Quinoxaline Intermediates Using Biodegradable Ionic Liquids
The pharmaceutical and fine chemical industries are constantly seeking sustainable alternatives to traditional synthetic routes that rely heavily on volatile organic compounds and harsh reaction conditions. Patent CN102010376B introduces a groundbreaking methodology for the synthesis of quinoxaline compounds, a class of nitrogen-containing heterocycles vital for drug development and advanced material science. This innovation leverages a biodegradable acidic functional ionic liquid as a catalyst within an aqueous reaction medium, effectively replacing toxic organic solvents. The technical breakthrough lies in the unique design of the catalyst, which combines high catalytic activity with environmental compatibility, allowing for reactions to proceed at ambient temperatures with exceptional efficiency. For R&D directors and process engineers, this represents a paradigm shift towards greener manufacturing protocols that do not compromise on yield or purity. By utilizing water as the primary solvent, the process eliminates the need for complex solvent recovery systems and reduces the overall carbon footprint of the synthesis operation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinoxaline derivatives has relied on condensation reactions between substituted o-phenylenediamines and 1,2-dicarbonyl compounds conducted in organic solvents such as glacial acetic acid, methanol, or dimethyl sulfoxide. These traditional protocols often require extended reaction times ranging from 2 to 12 hours and frequently necessitate elevated temperatures or microwave irradiation to drive the reaction to completion. Furthermore, the reliance on organic media introduces significant safety hazards, including flammability and toxicity, while generating substantial volumes of hazardous waste that require costly disposal procedures. The catalysts employed in these legacy methods are often non-degradable, leading to potential environmental accumulation and regulatory compliance issues. Additionally, equipment corrosion is a common challenge when using acidic organic solvents, resulting in increased maintenance costs and potential contamination of the final product with metal ions. The reported yields for these conventional methods typically fluctuate between 34% and 85%, indicating a lack of robustness and consistency that is unacceptable for large-scale commercial production.
The Novel Approach
In stark contrast to the limitations of solvent-dependent synthesis, the novel approach detailed in the patent utilizes a specifically designed biodegradable ionic liquid with a quaternary ammonium cationic structure to catalyze the reaction in pure water. This method operates under remarkably mild conditions, with reaction temperatures maintained between 20°C and 40°C, thereby eliminating the energy costs associated with heating and cooling cycles. The reaction kinetics are significantly accelerated, with completion times reduced to a mere 10 to 60 minutes, demonstrating a substantial improvement in throughput capacity. The use of water as the reaction medium not only enhances safety by removing flammable solvents but also simplifies the work-up procedure, as the product often precipitates out as a solid upon completion. This solid-liquid two-phase system allows for easy separation via filtration, avoiding the need for energy-intensive distillation or extraction processes. The result is a streamlined, high-yield process that delivers product purity levels suitable for pharmaceutical applications while adhering to strict environmental standards.
Mechanistic Insights into Acidic Ionic Liquid Catalysis
The core of this technological advancement is the specialized ionic liquid catalyst, which functions as a Brønsted acid to activate the carbonyl groups of the 1,2-dicarbonyl substrate. The catalyst features a long-chain alkyl quaternary ammonium cation coupled with a sulfonic acid functional group, creating an amphiphilic structure that is highly soluble in water yet effective at the organic-aqueous interface. 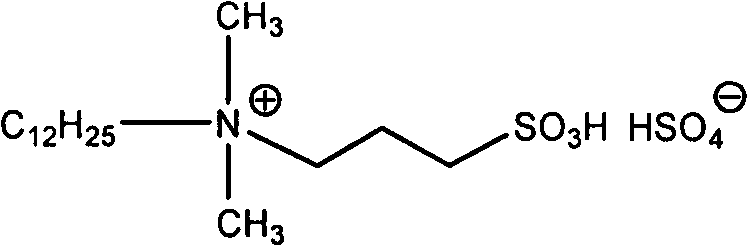 This structural design ensures that the catalyst remains stable and active throughout the reaction cycle without leaching or deactivation. The acidic protons provided by the sulfonic acid moiety facilitate the nucleophilic attack of the amine on the carbonyl carbon, lowering the activation energy barrier for the condensation step. Unlike homogeneous mineral acids which can cause severe corrosion and are difficult to separate, this ionic liquid remains in the aqueous phase after the product precipitates, allowing for seamless recycling. The mechanism avoids the formation of complex by-products often seen in radical-based or metal-catalyzed pathways, resulting in a cleaner impurity profile that simplifies downstream purification.
This structural design ensures that the catalyst remains stable and active throughout the reaction cycle without leaching or deactivation. The acidic protons provided by the sulfonic acid moiety facilitate the nucleophilic attack of the amine on the carbonyl carbon, lowering the activation energy barrier for the condensation step. Unlike homogeneous mineral acids which can cause severe corrosion and are difficult to separate, this ionic liquid remains in the aqueous phase after the product precipitates, allowing for seamless recycling. The mechanism avoids the formation of complex by-products often seen in radical-based or metal-catalyzed pathways, resulting in a cleaner impurity profile that simplifies downstream purification.
Impurity control is inherently managed by the specificity of the ionic liquid catalysis and the aqueous environment. In traditional organic solvents, side reactions such as polymerization or oxidation of the sensitive diamine starting materials can occur, leading to colored impurities that are difficult to remove. The aqueous phase acts as a heat sink, preventing localized hot spots that could trigger thermal degradation of the reactants. Furthermore, the biodegradable nature of the catalyst means that any trace amounts remaining in the wastewater can be broken down by biological treatment processes, preventing long-term ecological toxicity. The recrystallization step using ethanol further refines the product, removing any residual starting materials or minor by-products to achieve high-purity specifications. This dual approach of selective catalysis and physical separation ensures that the final quinoxaline compounds meet the rigorous quality standards required for API intermediates and electronic materials.
How to Synthesize Quinoxaline Compounds Efficiently
The implementation of this synthesis route is straightforward and designed for immediate scalability in a GMP-compliant environment. The process begins with the precise weighing of substituted o-phenylenediamine and the corresponding 1,2-dicarbonyl compound, maintaining a strict 1:1 molar ratio to maximize atom economy. The biodegradable ionic liquid catalyst is then added at a loading of 5 to 20 mol% relative to the diamine, followed by the addition of water which constitutes 50 to 80% of the total reaction mass. The mixture is stirred vigorously at room temperature to ensure homogeneity and efficient mass transfer between the phases. Detailed standardized synthesis steps follow below to guide the technical team in replicating these results.
- Prepare the reaction mixture by combining substituted o-phenylenediamine and 1,2-dicarbonyl compound in a 1: 1 molar ratio with 5-20 mol% of the biodegradable ionic liquid catalyst.
- Add water as the reaction medium, constituting 50-80% of the total mass of the materials, and stir the mixture at ambient temperature (20-40°C).
- After 10-60 minutes, filter the resulting solid-liquid two-phase system, wash the crude product with water, and recrystallize from ethanol to obtain pure quinoxaline.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this aqueous ionic liquid technology offers profound strategic benefits beyond simple chemical efficiency. The elimination of expensive and regulated organic solvents drastically reduces the raw material costs associated with solvent purchase, storage, and disposal. Since water is the primary medium, the risks associated with flammable inventory are removed, potentially lowering insurance premiums and facility safety requirements. The ability to recycle the catalyst directly in the filtrate means that the effective consumption of this high-value additive is minimized over multiple batches, leading to substantial long-term cost savings. Furthermore, the shortened reaction time increases the utilization rate of existing reactor vessels, allowing for higher production volumes without the need for capital expenditure on new equipment. This efficiency gain translates directly into a more competitive pricing structure for the final quinoxaline intermediates.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the removal of volatile organic compounds (VOCs) from the supply chain. Traditional methods require significant investment in solvent recovery units and emission control systems to comply with environmental regulations, all of which add overhead to the cost of goods sold. By switching to water, these capital and operational expenses are virtually eliminated. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, further driving down utility costs. The high yield of 86-95% ensures that raw material wastage is kept to an absolute minimum, maximizing the return on investment for every kilogram of starting material purchased. Qualitative analysis suggests that the total cost of production could be significantly lower than conventional solvent-based routes due to these cumulative efficiencies.
- Enhanced Supply Chain Reliability: Sourcing organic solvents can be subject to market volatility and logistical disruptions, whereas water is universally available and immune to supply shocks. The robustness of the ionic liquid catalyst, which is stable and easy to prepare from widely available raw materials, ensures a consistent supply of the key reagent. The simplified work-up procedure, which relies on filtration rather than complex distillation, reduces the dependency on specialized maintenance and spare parts for processing equipment. This reliability minimizes the risk of production downtime, ensuring that delivery schedules to downstream pharmaceutical customers are met consistently. The biodegradable nature of the catalyst also simplifies waste management logistics, as the aqueous waste stream can be treated in standard facilities without requiring specialized hazardous waste contractors.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental challenges, but this aqueous method is inherently safer for large-scale operations. The absence of exothermic runaway risks associated with organic solvents makes the scale-up from laboratory to commercial tonnage much more predictable and controllable. Regulatory compliance is significantly easier to achieve since the process avoids the use of listed hazardous air pollutants and generates biodegradable waste. This alignment with green chemistry principles enhances the corporate sustainability profile, which is increasingly important for securing contracts with major multinational corporations that have strict ESG mandates. The process is designed to be environmentally friendly from start to finish, ensuring long-term viability in a tightening regulatory landscape.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid catalyzed synthesis. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing manufacturing lines.
Q: What are the environmental advantages of this ionic liquid catalyst compared to traditional solvents?
A: Unlike traditional organic solvents such as glacial acetic acid or dimethyl sulfoxide which pose disposal challenges, this method uses water as the medium and a biodegradable quaternary ammonium-based ionic liquid. Activated sludge tests confirm the catalyst wastewater undergoes biochemical reactions easily, ensuring minimal environmental impact.
Q: Can the ionic liquid catalyst be recycled for subsequent batches?
A: Yes, the catalyst demonstrates excellent stability in water and does not deactivate during the reaction. The filtered aqueous phase containing the ionic liquid and unreacted raw materials can be reused directly for the next synthesis cycle without additional treatment, maintaining the 1:1 reactant ratio.
Q: What yields can be expected from this aqueous phase synthesis method?
A: The patent data indicates significantly improved efficiency compared to conventional methods, with product yields ranging from 86% to 95%. This high efficiency is achieved under mild conditions (20-40°C) within a short reaction time of 10 to 60 minutes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoxaline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green chemistry technologies like the biodegradable ionic liquid catalysis described in CN102010376B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for pharmaceutical intermediates and fine chemicals. We are committed to delivering high-purity quinoxaline compounds that adhere to the highest quality standards while maintaining the environmental benefits of this aqueous synthesis route.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments for your target quinoxaline derivatives. By partnering with us, you gain access to a reliable supply of eco-friendly intermediates that can enhance your product portfolio and support your sustainability goals.
