Advanced Stereoselective Synthesis of (3aS,6aR)-Lactone for Commercial Biotin Production
Introduction to Advanced Lactone Synthesis Technology
The pharmaceutical industry constantly seeks robust pathways for producing high-value vitamin intermediates, and the stereoselective synthesis of (3aS,6aR)-lactone stands as a cornerstone for D-Biotin (Vitamin H) manufacturing. As detailed in patent CN114634515A, a groundbreaking methodology has been established that transcends the limitations of historical resolution techniques. This innovation leverages a sophisticated desymmetrization strategy coupled with efficient reduction and cyclization steps to deliver the target lactone with exceptional optical purity. By integrating chiral propylene glycol derivatives as auxiliary agents, the process achieves total yields exceeding 88% and enantiomeric excess (ee) values greater than 99%. For R&D directors and procurement specialists, this represents a paradigm shift towards more economical and scalable production of critical pharmaceutical intermediates, ensuring a stable supply chain for essential nutraceuticals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of (3aS,6aR)-lactone relied heavily on resolution strategies that were inherently inefficient and cost-prohibitive. Pioneering work by Gerecke et al. and subsequent patents utilized expensive chiral resolving agents such as pseudoephedrine, dehydroabietine amine, or cholesterol to separate racemic mixtures. These traditional approaches suffered from significant drawbacks, including low single-batch yields due to the theoretical 50% maximum yield limit of resolution processes. Furthermore, the operational complexity was high, often requiring multiple recrystallization steps to achieve acceptable optical purity, which drastically increased solvent consumption and processing time. The reliance on scarce or synthetically challenging chiral amines also introduced volatility into the supply chain, making cost prediction difficult for large-scale API manufacturing.
The Novel Approach
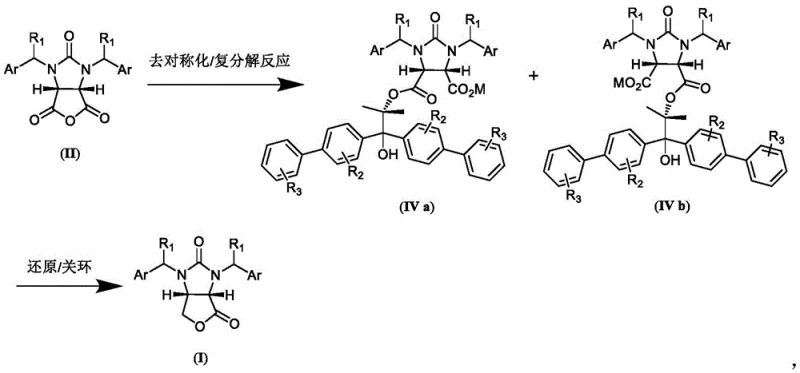
In stark contrast, the novel approach outlined in the patent data introduces a catalytic desymmetrization pathway that bypasses the inefficiencies of classical resolution. By reacting a meso-cyclic acid anhydride with a chiral (S)-1,2-propylene glycol derivative in the presence of an organic base, the process selectively opens one carbonyl group to form a single diastereomer of the dicarboxylic acid monoester salt. This method not only theoretically allows for 100% conversion of the starting anhydride but also utilizes auxiliaries that are significantly cheaper and easier to synthesize than traditional alkaloids. The subsequent transformation involves a straightforward reduction followed by acid-catalyzed cyclization, streamlining the entire workflow. This shift from resolution to asymmetric synthesis fundamentally alters the economic landscape of biotin intermediate production, offering a reliable pharmaceutical intermediate supplier a distinct competitive advantage.
Mechanistic Insights into Desymmetrization and Cyclization
The core of this technological breakthrough lies in the precise stereochemical control exerted during the desymmetrization of the cyclic acid anhydride. The reaction initiates with the nucleophilic attack of the chiral propylene glycol auxiliary on one of the carbonyl carbons of the meso-anhydride. The bulky substituents on the auxiliary, such as the biphenyl groups found in preferred embodiments, create a steric environment that favors attack from a specific face, thereby establishing the desired (4S,5R) configuration in the intermediate half-ester. Following this, a metathesis reaction with a metal hydroxide, preferably lithium hydroxide, converts the acidic proton into a stable salt form, facilitating purification through simple filtration. This step is crucial for removing unreacted starting materials and ensuring the high diastereomeric excess required for the final product's optical purity.
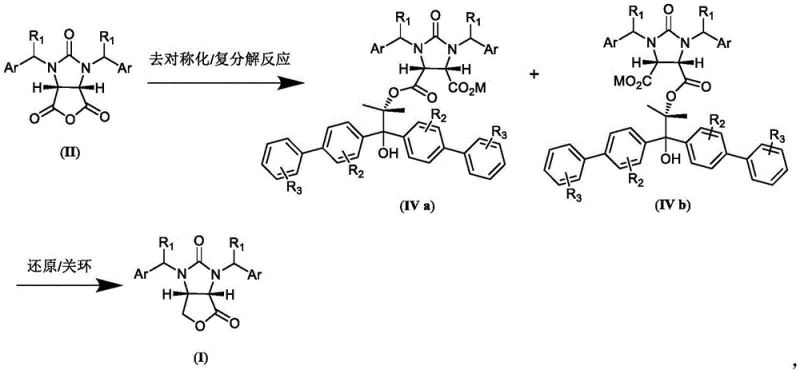
Following the formation of the dicarboxylic acid monoester salt, the synthesis proceeds through a reduction and cyclization sequence that preserves the established stereocenters. The salt is treated with a borohydride reducing agent, such as lithium borohydride, in a solvent like tetrahydrofuran. This step selectively reduces the carboxylic acid moiety (or the ester depending on specific conditions described) to a hydroxymethyl group without affecting the urea linkage or the aromatic systems. The final ring closure is triggered by the addition of an inorganic mineral acid, typically hydrochloric acid, under heated conditions. This acid-catalyzed lactonization forms the fused furano-imidazole ring system characteristic of the (3aS,6aR)-lactone. The robustness of this mechanism ensures that impurities are minimized, as the reaction conditions are mild enough to prevent racemization yet vigorous enough to drive the cyclization to completion.
How to Synthesize (3aS,6aR)-Lactone Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and temperature control to maximize the benefits of the desymmetrization strategy. The process begins by mixing the cyclic anhydride and the chiral auxiliary in a dry organic solvent like toluene, followed by the slow addition of an organic base such as tri-n-butylamine at controlled low temperatures to manage exotherms. Once the monoester salt is formed and isolated, it undergoes reduction in a second solvent system, followed by the critical acid workup. The detailed standardized synthesis steps see the guide below for specific molar ratios and thermal profiles optimized for industrial reactors.
- Perform desymmetrization of cyclic acid anhydride with chiral propylene glycol auxiliary in organic solvent with organic base, followed by metathesis to form dicarboxylic acid monoester salt.
- Reduce the dicarboxylic acid monoester salt using borohydride in a second organic solvent.
- Execute ring closure reaction under inorganic mineral acid catalysis to obtain the final (3aS,6aR)-lactone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis method offers profound strategic benefits beyond mere technical elegance. The primary advantage stems from the drastic simplification of the raw material portfolio. By replacing scarce, high-cost chiral resolving agents with commercially abundant propylene glycol derivatives, manufacturers can significantly reduce raw material expenditure and mitigate supply risks associated with niche chemical suppliers. Furthermore, the elimination of multiple recrystallization steps inherent in resolution processes leads to a substantial reduction in solvent usage and waste generation, directly lowering operational costs and environmental compliance burdens. This efficiency translates into a more resilient supply chain capable of meeting fluctuating market demands for biotin without the bottlenecks typical of older technologies.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the replacement of expensive chiral auxiliaries with cost-effective alternatives that do not compromise on stereoselectivity. Traditional methods often incurred high costs due to the loss of 50% of the material during resolution and the expense of recovering chiral bases. In this new pathway, the high yield and the ability to potentially recover and recycle the chiral auxiliary contribute to a leaner cost structure. Additionally, the use of common solvents like toluene and THF, rather than specialized mixtures, further optimizes the cost reduction in API manufacturing by leveraging existing infrastructure and bulk purchasing power.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of widely available starting materials. The cyclic anhydrides and propylene glycol derivatives used in this synthesis are produced on a large industrial scale, ensuring that production schedules are not disrupted by the scarcity of specialized reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and do not require ultra-low temperature cryogenics for extended periods, also enhances equipment utilization rates. This reliability is critical for reducing lead time for high-purity intermediates, allowing manufacturers to respond swiftly to downstream formulation needs.
- Scalability and Environmental Compliance: The commercial scale-up of complex lactones is often hindered by difficult purification steps, but this method simplifies isolation through precipitation and filtration of the intermediate salt. This solid-phase isolation is inherently easier to scale than liquid-liquid extractions or chromatographic separations. Moreover, the high atom economy of the desymmetrization reaction means less chemical waste is generated per kilogram of product. This aligns with modern green chemistry principles, facilitating easier regulatory approval and reducing the costs associated with waste treatment and disposal in large-scale facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this stereoselective synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy processes in terms of yield, purity, and operational simplicity. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their own production lines.
Q: What are the advantages of this new synthesis method over traditional resolution methods?
A: Traditional methods often rely on expensive chiral auxiliaries like ephedrine derivatives or cholesterol, requiring complex diastereomeric crystallization with low single-batch yields. This novel approach utilizes cost-effective (S)-1,2-propylene glycol derivatives, achieving total yields exceeding 88% and ee values greater than 99% through a simpler desymmetrization pathway that avoids cumbersome resolution steps.
Q: How does this process ensure high optical purity for pharmaceutical applications?
A: The process employs a highly stereoselective desymmetrization reaction using bulky chiral auxiliaries such as (S)-1,1-bis([1,1'-biphenyl]-4-yl)-1,2-propanediol. The steric hindrance provided by the biphenyl groups ensures excellent diastereoselectivity during the ring-opening of the cyclic anhydride, consistently delivering products with enantiomeric excess (ee) values surpassing 99%, which is critical for downstream Vitamin H synthesis.
Q: Is this synthesis method suitable for large-scale industrial manufacturing?
A: Yes, the method is designed for industrial scalability. It utilizes readily available raw materials like tri-n-butylamine and toluene, operates under mild reaction temperatures ranging from -20°C to 80°C, and simplifies workup procedures through filtration and standard extraction. The elimination of complex enzymatic steps or difficult-to-recover chiral reagents significantly enhances its feasibility for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (3aS,6aR)-Lactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of (3aS,6aR)-lactone meets the exacting standards required for Vitamin H synthesis. Our capability to implement advanced stereoselective chemistries allows us to offer products with superior optical purity and consistent quality.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your specific manufacturing requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits tailored to your volume needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on the foundation of the most advanced and reliable chemical technologies available in the market.
