Revolutionizing Biotin Intermediate Production: High-Purity (3aS,6aR)-Lactone via Continuous Flow Technology
The pharmaceutical industry constantly seeks more efficient pathways for synthesizing critical vitamin intermediates, and the production of (3aS,6aR)-lactone stands as a pivotal challenge in the manufacture of (+)-Biotin (Vitamin H). Patent CN114560865A introduces a groundbreaking continuous flow preparation method that fundamentally transforms this synthetic landscape by leveraging advanced micro-reaction systems. This innovative approach replaces traditional batch processing with a seamless, multi-step continuous workflow that integrates desymmetrization, reduction, and cyclization into a single automated sequence. The result is a robust manufacturing protocol capable of delivering total product yields exceeding 90% with exceptional optical purity, where the enantiomeric excess (ee) consistently surpasses 99%. For R&D directors and procurement specialists, this represents a significant leap forward in process intensification, offering a reliable pharmaceutical intermediate supplier pathway that minimizes waste and maximizes throughput.
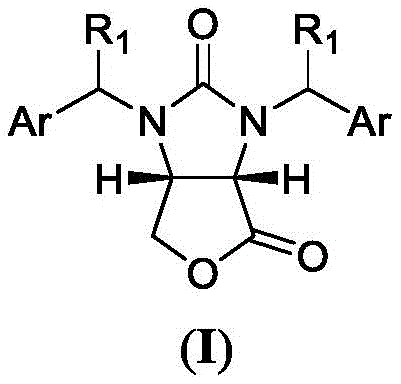
Historically, the synthesis of (3aS,6aR)-lactone has been plagued by inefficiencies inherent to conventional batch methodologies. Prior art, including methods described by Gerecke et al. and various patents utilizing chiral resolutions with pseudoephedrine or dehydroabietine, suffers from cumbersome operational procedures and low single-batch yields. These traditional routes often require multiple isolation and purification steps, such as diastereomeric crystallization, which not only extend the production timeline to over 18 hours but also introduce significant material losses. Furthermore, the reliance on expensive chiral auxiliaries that are difficult to recover creates a substantial economic burden, making cost reduction in pharmaceutical intermediate manufacturing a critical priority. The batch processes also struggle with heat and mass transfer limitations, leading to inconsistent reaction profiles and potential safety hazards when scaling up exothermic steps.
In stark contrast, the novel approach detailed in the patent utilizes a sophisticated micro-reaction system comprising sequentially connected micro-mixers and micro-channel reactors to overcome these legacy limitations. By pumping the cyclic acid anhydride substrate and the chiral auxiliary propylene glycol solution simultaneously into a multi-level structure micro-channel mixer, the system achieves instantaneous mixing and precise temperature control. This continuous desymmetry reaction flows directly into a reduction stage with borohydride and finally into an acid-catalyzed cyclization, all without intermediate workup. This integration reduces the total reaction time to merely 15 minutes while suppressing side reactions that typically degrade yield in batch tanks. The transition from batch to continuous flow effectively eliminates the need for metathesis reactions and filtration steps associated with isolating half-ester intermediates, thereby streamlining the entire value chain.
Mechanistic Insights into Micro-Reactor Catalyzed Desymmetrization and Cyclization
The core of this technological breakthrough lies in the precise control of stereochemistry during the initial desymmetrization step. The process employs a cyclic acid anhydride substrate, structurally represented as formula (II), which reacts with a chiral auxiliary, specifically (S)-1,2-propylene glycol derivatives shown as formula (III). 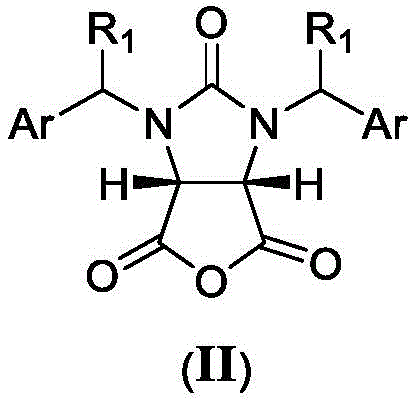 . In the first micro-channel reactor, maintained at temperatures between -20°C and 80°C, the chiral auxiliary induces high diastereoselectivity, yielding the (4S,5R)-half ester intermediate with greater than 98% diastereomeric excess (de). The superior mixing efficiency of the micro-reactor ensures that the chiral environment is uniform throughout the reaction volume, preventing the formation of racemic byproducts that often occur in poorly mixed batch vessels. This step is critical for establishing the stereocenters required for the final biotin structure.
. In the first micro-channel reactor, maintained at temperatures between -20°C and 80°C, the chiral auxiliary induces high diastereoselectivity, yielding the (4S,5R)-half ester intermediate with greater than 98% diastereomeric excess (de). The superior mixing efficiency of the micro-reactor ensures that the chiral environment is uniform throughout the reaction volume, preventing the formation of racemic byproducts that often occur in poorly mixed batch vessels. This step is critical for establishing the stereocenters required for the final biotin structure.
Following the desymmetrization, the reaction stream undergoes a continuous reduction and subsequent ring closure. The effluent from the first reactor is immediately mixed with a borohydride solution, preferably lithium borohydride, in a second micro-mixer. This reduction step converts the ester functionality into the corresponding alcohol, which is then subjected to acid-catalyzed cyclization in the third reactor using inorganic mineral acids like hydrochloric acid. 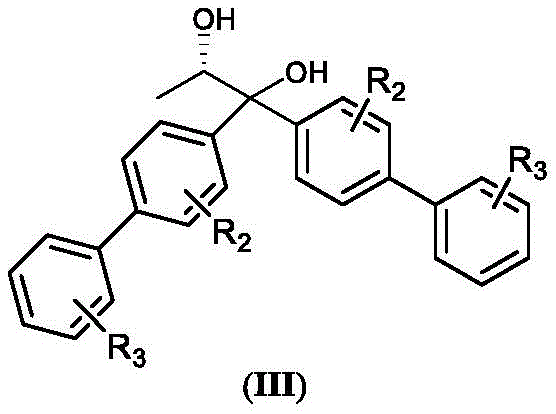 . The rapid heat dissipation capabilities of the micro-channels allow the cyclization to proceed at elevated temperatures (up to 150°C) safely, driving the equilibrium towards the desired lactone product. This seamless transition between chemical transformations prevents the degradation of sensitive intermediates and ensures that the final product retains an enantiomeric excess of 100%, as demonstrated in specific embodiments where yields reached 96.8%.
. The rapid heat dissipation capabilities of the micro-channels allow the cyclization to proceed at elevated temperatures (up to 150°C) safely, driving the equilibrium towards the desired lactone product. This seamless transition between chemical transformations prevents the degradation of sensitive intermediates and ensures that the final product retains an enantiomeric excess of 100%, as demonstrated in specific embodiments where yields reached 96.8%.
How to Synthesize (3aS,6aR)-Lactone Efficiently
Implementing this continuous flow synthesis requires a specialized setup involving precise fluid dynamics and thermal management to replicate the patent's success. The process dictates the use of specific multi-level structure micro-channel mixers rather than standard T-mixers or static mixers, as the latter have been shown to result in incomplete substrate conversion and lower yields (dropping to ~85-89% in comparative examples). Operators must carefully control the molar ratios of the cyclic anhydride, chiral auxiliary, and organic base, ideally maintaining a ratio of 1:(1.05~1.3):(0.11~1.5) to optimize material usage while ensuring complete reaction. The detailed standardized synthesis steps, including specific flow rates, residence times, and temperature gradients for each reactor stage, are outlined below to guide technical teams in replicating this high-efficiency protocol.
- Simultaneously pump cyclic acid anhydride substrate and chiral auxiliary propylene glycol solution into the first micro-mixer for continuous desymmetry reaction.
- Mix the effluent with borohydride solution in a second micro-mixer and proceed to the second micro-channel reactor for continuous reduction.
- Combine the reduced mixture with inorganic mineral acid in a third micro-mixer and enter the third micro-channel reactor for final continuous ring closure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this continuous flow methodology offers profound strategic benefits beyond mere technical superiority. The elimination of expensive and hard-to-recover chiral auxiliaries, replaced by readily available (S)-1,2-propylene glycol derivatives, directly translates to substantial cost savings in raw material procurement. Additionally, the drastic reduction in reaction time from over 18 hours to just 15 minutes significantly increases the asset turnover rate of manufacturing equipment, allowing facilities to produce higher volumes without expanding their physical footprint. This intensification of production capacity means that suppliers can respond more agilely to market demand fluctuations, reducing the risk of stockouts for critical vitamin intermediates.
- Cost Reduction in Manufacturing: The continuous nature of the process removes the need for intermediate isolation, filtration, and drying steps that are labor-intensive and solvent-heavy in batch processing. By integrating the desymmetrization, reduction, and cyclization into a single flow, the consumption of organic solvents is minimized, and the energy required for heating and cooling cycles is drastically lowered. Furthermore, the ability to recover the chiral auxiliary easily enhances the overall atom economy of the process, leading to a leaner cost structure that improves margin potential for high-purity pharmaceutical intermediate buyers.
- Enhanced Supply Chain Reliability: Traditional batch synthesis is prone to variability between batches due to mixing inconsistencies and human intervention, which can lead to supply disruptions if a batch fails quality control. The automated, continuous flow system described in the patent operates with high reproducibility, ensuring consistent product quality and reducing the lead time for high-purity pharmaceutical intermediates. The modular nature of the micro-reactor system also means that maintenance can be performed on individual units without shutting down the entire production line, thereby guaranteeing a more stable and continuous supply for downstream biotin manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces safety risks, particularly with exothermic reactions involving borohydrides and strong acids. This micro-reactor approach mitigates these risks by maintaining a very small hold-up volume at any given time, making the reaction intrinsically safer and easier to permit under strict environmental regulations. The commercial scale-up of complex pharmaceutical intermediates is achieved through numbering-up parallel channels rather than increasing vessel size, which preserves the reaction efficiency and safety profile. This facilitates easier compliance with green chemistry principles by reducing waste generation and energy consumption per kilogram of product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this continuous flow technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on how this method compares to existing industry standards. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this new synthesis route for their own production lines.
Q: What are the primary advantages of using continuous flow for (3aS,6aR)-lactone synthesis compared to batch processing?
A: Continuous flow synthesis drastically reduces reaction time from over 18 hours to approximately 15 minutes while increasing total yield from roughly 88-90% to over 95%. It also eliminates complex isolation steps between reactions, enhancing overall process efficiency and safety.
Q: Which chiral auxiliary is utilized to ensure high enantiomeric excess in this method?
A: The process employs (S)-1,2-propylene glycol derivatives as the chiral auxiliary. These auxiliaries offer high diastereoselectivity (>98% de), are cost-effective, widely available, and can be easily recovered, significantly lowering production costs compared to expensive alternatives like ephedrine derivatives.
Q: Is this micro-reactor technology scalable for industrial manufacturing?
A: Yes, the technology is highly scalable through a strategy of numbering-up parallel micro-channels. This approach maintains the precise heat and mass transfer characteristics of the laboratory scale while allowing for large-scale industrial production without the safety risks associated with large batch reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (3aS,6aR)-Lactone Supplier
As the global demand for vitamins and fine chemicals continues to rise, securing a partner with the technical capability to execute complex continuous flow syntheses is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this innovation, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that ensure every batch of (3aS,6aR)-lactone meets the highest international standards. We understand that consistency is key in the pharmaceutical supply chain, and our advanced manufacturing infrastructure is designed to deliver the reliability and volume that multinational corporations require.
We invite you to collaborate with us to leverage these cutting-edge process improvements for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this continuous flow technology can optimize your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for next-generation chemical manufacturing solutions.
