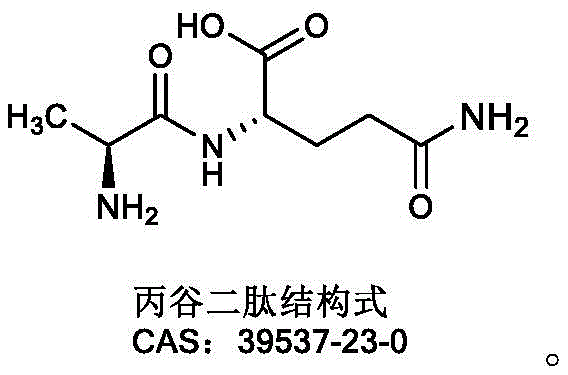
Revolutionizing L-Alanyl-L-Glutamine Production via High-Activity Alpha-Amino Acid Ester Acyltransferase
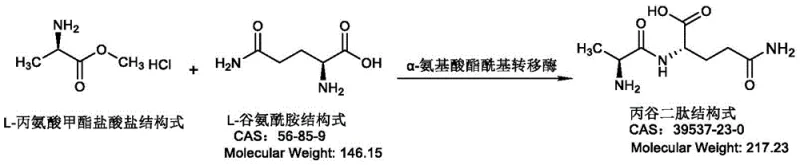
The pharmaceutical industry continuously seeks robust and efficient pathways for synthesizing critical amino acid derivatives, particularly those used in parenteral nutrition. Patent CN115521961A, published in late 2022, introduces a significant breakthrough in the biocatalytic production of L-Alanyl-L-Glutamine, a stable dipeptide widely recognized as the superior carrier for glutamine supplementation. This technology leverages specific variants of alpha-amino acid ester acyltransferase (AET) derived from distinct bacterial sources such as Arcticibacter tournemirensis and Sphingobacterium spiritivorum. Unlike traditional chemical synthesis which often suffers from racemization risks and harsh reaction conditions, this enzymatic approach offers a stereoselective route that maintains the integrity of the chiral centers essential for biological activity. The patent details how these engineered enzymes facilitate the coupling of L-alanine methyl ester and L-glutamine with remarkable efficiency, addressing the long-standing market demand for high-purity glutamine dipeptides suitable for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of glutamine dipeptides has faced substantial hurdles regarding yield optimization and impurity control. Conventional chemical methods typically require protection and deprotection steps for the amino and carboxyl groups, leading to complex multi-step processes that generate significant chemical waste. Furthermore, maintaining optical purity during chemical coupling is challenging, often resulting in the formation of diastereomers that are difficult to separate and potentially toxic. Even earlier enzymatic attempts, such as those utilizing acyltransferases from Empedobacter brevis, often reported conversion rates hovering around 67% to 70% at moderate substrate concentrations. These limitations translate directly into higher production costs and supply chain vulnerabilities for manufacturers relying on outdated technologies, as the purification burden increases exponentially with lower conversion efficiencies.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by utilizing highly active recombinant enzymes that operate under mild physiological conditions. By employing alpha-amino acid ester acyltransferases with amino acid sequences corresponding to SEQ ID NO: 2, 4, or 6, the process achieves conversion rates exceeding 87% and reaching up to 98.2% in optimized examples. This novel approach eliminates the need for protecting groups, as the enzyme naturally discriminates between functional groups, catalyzing the formation of the peptide bond specifically between the alpha-amino group of glutamine and the carbonyl of the alanine ester. The reaction proceeds efficiently in aqueous buffers at temperatures between 20°C and 40°C, drastically reducing energy consumption compared to thermal chemical processes. This streamlined workflow not only enhances the overall yield but also simplifies the downstream purification strategy, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Alpha-Amino Acid Ester Acyltransferase Catalysis
The core of this technological advancement lies in the specific catalytic mechanism of the alpha-amino acid ester acyltransferase (AET). This enzyme functions by recognizing L-alanine methyl ester as an acyl donor and transferring the alanyl moiety to the alpha-amino group of L-glutamine, which acts as the nucleophile. The patent specifies that the enzyme possesses a broad substrate spectrum but exhibits exceptional specificity for this particular dipeptide synthesis, minimizing the formation of poly-peptide byproducts which are common in non-specific protease reactions. The catalytic cycle involves the formation of an acyl-enzyme intermediate, followed by nucleophilic attack by the amine group of glutamine. Crucially, the engineered variants described in the patent demonstrate a specific enzyme activity above 40 U/mg, indicating a highly optimized active site architecture that facilitates rapid turnover. This high activity allows for the use of lower enzyme loading or shorter reaction times, which is a critical parameter for industrial scalability.
Impurity control is inherently managed through the stereospecificity of the biocatalyst. Since the enzyme is derived from biological systems and expressed in E. coli, it strictly recognizes the L-configuration of the substrates, thereby preventing the formation of D-isomer impurities that could compromise the safety profile of the final API. The reaction conditions, maintained at a pH of 8.0 to 9.0, ensure that the substrates remain in their optimal ionization states for binding while minimizing non-enzymatic hydrolysis of the methyl ester. Furthermore, the patent highlights the ability to use crude enzyme solutions or even whole cells, suggesting that the enzyme is robust enough to function effectively without extensive purification. This reduces the introduction of extraneous proteins or contaminants into the reaction matrix, simplifying the subsequent filtration and chromatography steps required to meet stringent pharmaceutical purity specifications.
How to Synthesize L-Alanyl-L-Glutamine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for replicating these high-efficiency results in a laboratory or pilot plant setting. The process begins with the construction of expression vectors, such as pET-21a, containing the specific gene sequences for the acyltransferase, followed by transformation into competent E. coli BL21(DE3) cells. Induction is carefully controlled using IPTG at low temperatures (22-26°C) to ensure proper protein folding and maximize soluble expression. Once the biomass is harvested, it can be used directly as a whole-cell catalyst or processed into a crude enzyme solution. The actual coupling reaction involves mixing the substrates in a phosphate buffer system, where the molar ratio of L-alanine methyl ester hydrochloride to L-glutamine is typically maintained at 1:1. Detailed standardized synthesis steps see the guide below.
- Construct expression vectors containing the gene sequences for alpha-amino acid ester acyltransferase (e.g., SEQ ID NO: 1, 3, or 5) and transform them into E. coli BL21(DE3) host cells.
- Culture the transformed strains in LB medium at 37°C until OD600 reaches 0.8, then induce enzyme expression with 0.5mM IPTG at 25°C for 16 hours.
- Catalyze the reaction by mixing L-alanine methyl ester hydrochloride and L-glutamine with the crude enzyme solution at pH 8.0-9.0 and 25-30°C to achieve high conversion rates.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic technology offers compelling strategic benefits beyond mere technical feasibility. The shift from chemical synthesis to biocatalysis fundamentally alters the cost structure of producing L-Alanyl-L-Glutamine. By eliminating the need for expensive protecting group reagents and hazardous organic solvents, the raw material costs are significantly reduced. Moreover, the high conversion rates reported in the patent mean that less starting material is wasted, directly improving the atom economy of the process. This efficiency gain translates into substantial cost savings in raw material procurement, allowing manufacturers to offer more competitive pricing in the global market for parenteral nutrition ingredients.
- Cost Reduction in Manufacturing: The enzymatic process operates under mild conditions (25-30°C) and atmospheric pressure, which drastically lowers energy consumption compared to high-temperature chemical reflux methods. Additionally, the ability to use crude enzyme preparations or whole cells removes the costly and time-consuming steps associated with enzyme purification. This simplification of the upstream process reduces capital expenditure on specialized equipment and lowers operational expenses related to waste disposal, as the aqueous waste stream is far easier to treat than solvent-heavy chemical waste.
- Enhanced Supply Chain Reliability: The reliance on fermentation-derived enzymes ensures a consistent and renewable supply of the catalyst, unlike chemical catalysts which may depend on scarce precious metals. The robustness of the described strains, capable of high-level expression in standard E. coli hosts, means that production can be scaled rapidly to meet surges in demand without long lead times for catalyst sourcing. This reliability is crucial for maintaining continuous supply lines for critical care nutrition products, mitigating the risk of shortages that can occur with complex multi-step chemical syntheses.
- Scalability and Environmental Compliance: The process is inherently green, aligning with increasingly strict environmental regulations governing pharmaceutical manufacturing. The absence of heavy metal catalysts and volatile organic compounds (VOCs) simplifies regulatory compliance and reduces the environmental footprint of the facility. The high solubility of the substrates and products in water facilitates easy handling and pumping in large-scale reactors, ensuring that the transition from bench scale to multi-ton commercial production is smooth and predictable, thereby securing long-term supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented enzymatic technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of the alpha-amino acid ester acyltransferase disclosed in CN115521961A?
A: The enzyme exhibits high specific activity (above 40 U/mg) and achieves conversion rates exceeding 87%, significantly improving process efficiency compared to prior art methods which often struggle with lower yields or stability issues.
Q: Which substrates are required for the enzymatic synthesis of L-Alanyl-L-Glutamine?
A: The process utilizes L-alanine methyl ester (or its hydrochloride salt) as the acyl donor and L-glutamine as the nucleophilic acceptor, reacting under mild aqueous conditions without the need for hazardous organic solvents.
Q: Is this enzymatic process suitable for large-scale industrial production?
A: Yes, the patent highlights the use of whole-cell catalysis and crude enzyme solutions, which simplifies downstream processing and reduces energy consumption, making it highly scalable for commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Alanyl-L-Glutamine Supplier
As the global demand for high-quality parenteral nutrition ingredients continues to rise, partnering with an experienced CDMO is essential for navigating the complexities of biocatalytic manufacturing. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of L-Alanyl-L-Glutamine meets the highest international pharmacopoeia standards. We understand the critical nature of this intermediate in clinical settings and prioritize quality assurance at every step of the production lifecycle.
We invite you to collaborate with us to leverage this advanced enzymatic technology for your product pipeline. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this green synthesis route can optimize your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of this vital pharmaceutical intermediate.
