Revolutionizing L-Alanyl-L-Glutamine Production: A Strategic Technical Analysis for Global Supply Chains
Introduction to Advanced Dipeptide Manufacturing Strategies
The pharmaceutical landscape for parenteral nutrition is constantly evolving, driven by the need for more efficient and cost-effective synthesis of critical dipeptides like L-alanyl-L-glutamine (Ala-Gln). Patent CN103387600A introduces a transformative preparation method that fundamentally shifts the raw material paradigm from expensive L-glutamine to readily available L-glutamic acid derivatives. This technical breakthrough addresses the longstanding economic bottlenecks in dipeptide manufacturing by leveraging a phthalyl-protection strategy that ensures high stereochemical purity while minimizing operational complexity. For global supply chain leaders, this represents a pivotal opportunity to secure a more stable and economical source of this vital nutritional intermediate, moving away from volatile pricing structures associated with traditional glutamine-dependent routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of L-alanyl-L-glutamine has been plagued by reliance on high-cost starting materials and cumbersome protection-deprotection sequences. Prior art, such as European Patent EP311057, typically employs benzyloxycarbonyl-protected L-alanine activated as an ester, which is then condensed with free L-glutamine. This approach is inherently flawed because L-glutamine itself is a premium-priced amino acid, susceptible to degradation and difficult to source in bulk quantities compared to its acid counterpart. Furthermore, these legacy processes often necessitate catalytic hydrogenation for the removal of protecting groups, introducing significant safety hazards related to high-pressure hydrogen handling and requiring specialized, capital-intensive reactor infrastructure that limits flexibility in multipurpose facilities.
The Novel Approach
In stark contrast, the methodology disclosed in the subject patent circumvents these issues by utilizing L-glutamic acid monoesters as the nucleophilic partner, coupled with phthalyl-L-alanyl halides as the electrophile. This strategic substitution replaces the costly glutamine moiety with a glutamic acid derivative that is subsequently converted to the amide functionality in the final step. The innovation lies in the elegance of the phthalyl group, which serves as a robust amine protector that can be cleaved simultaneously with the ester-to-amide conversion using ammonium hydroxide. This convergence of steps not only streamlines the synthetic pathway but also eliminates the need for precious metal catalysts, resulting in a process that is both economically superior and operationally simpler for large-scale chemical manufacturing.
Mechanistic Insights into Phthalyl-Mediated Peptide Coupling
The core of this synthetic innovation relies on the precise activation of the L-alanine carboxyl group via conversion to an acid halide, specifically the phthalyl-L-alanyl chloride or bromide. This activation creates a highly reactive electrophilic center that facilitates rapid acylation of the amino group on the L-glutamic acid monoester. The reaction is meticulously controlled within a biphasic organic-aqueous system, where the pH is maintained between 8.0 and 10.0 using bases such as sodium hydroxide or sodium carbonate. This specific pH window is critical; it ensures the amino group of the glutamic acid derivative remains sufficiently unprotonated to act as a nucleophile, while preventing the hydrolysis of the sensitive acid halide intermediate, thereby maximizing the coupling efficiency and minimizing the formation of inactive phthalyl-alanine byproducts.
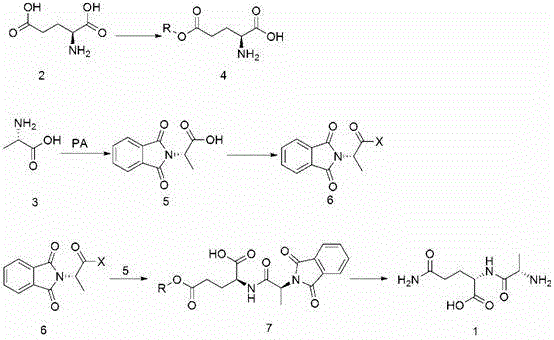
Following the condensation, the resulting protected dipeptide intermediate undergoes a sophisticated ammonolysis reaction that serves a dual mechanistic purpose. Upon treatment with concentrated ammonium hydroxide at mild temperatures ranging from 0 to 25°C, the phthalimide protecting group is cleaved via nucleophilic attack by ammonia, regenerating the free alpha-amino group of the alanine residue. Simultaneously, the ester functionality on the glutamic acid side chain undergoes aminolysis to form the primary amide, effectively constructing the glutamine moiety in situ. This tandem transformation is chemically elegant as it achieves two distinct structural modifications in a single operational step, significantly reducing solvent consumption and processing time while ensuring that the final crystallization yields a product with exceptional purity levels exceeding 99 percent as verified by HPLC analysis.
How to Synthesize L-Alanyl-L-Glutamine Efficiently
The implementation of this synthesis route requires careful attention to the stoichiometry and thermal conditions during the condensation phase to ensure optimal yield and impurity profiles. The process begins with the preparation of the key intermediates, specifically the phthalyl-L-alanyl halide and the glutamic acid monoester, which must be synthesized with high fidelity to prevent downstream contamination. Operators must strictly adhere to the specified pH control protocols during the coupling reaction to maintain the balance between reaction rate and intermediate stability. For a comprehensive understanding of the specific reagent ratios, solvent systems, and workup procedures required to replicate this high-efficiency pathway, please refer to the standardized synthesis guide provided below.
- Prepare L-glutamic acid monoester via acid-catalyzed esterification of L-glutamic acid with alcohol.
- Synthesize phthalyl-L-alanyl halide by reacting L-alanine with phthalic anhydride followed by halogenation.
- Condense the monoester and halide under alkaline conditions (pH 8.0-10.0) to form the protected dipeptide intermediate.
- Perform ammonolysis and deprotection using ammonium hydroxide to yield the final L-alanyl-L-glutamine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this manufacturing route offers profound advantages by decoupling production costs from the volatile market pricing of L-glutamine. By substituting the primary raw material with L-glutamic acid, a commodity chemical produced via large-scale fermentation with consistent availability, manufacturers can achieve substantial cost savings in dipeptide manufacturing. This shift not only stabilizes the bill of materials but also mitigates supply chain risks associated with sourcing specialized amino acids, ensuring a more reliable flow of goods for downstream pharmaceutical formulators who depend on consistent inventory levels for parenteral nutrition products.
- Cost Reduction in Manufacturing: The elimination of expensive L-glutamine as a starting material fundamentally alters the cost structure of the final API intermediate. Since L-glutamic acid is significantly cheaper and more abundant than L-glutamine, the raw material cost burden is drastically reduced without compromising the quality of the final dipeptide. Additionally, the removal of catalytic hydrogenation steps eliminates the need for expensive palladium or platinum catalysts and the associated recovery processes, further contributing to a leaner and more cost-effective production model that enhances overall profit margins for suppliers.
- Enhanced Supply Chain Reliability: Utilizing bulk commodity chemicals like phthalic anhydride and L-glutamic acid ensures a robust supply chain that is less susceptible to disruptions compared to routes relying on niche protected amino acids. The process utilizes common industrial solvents such as ethyl acetate and toluene, which are readily available globally, reducing the risk of logistical bottlenecks. This accessibility allows for flexible sourcing strategies and enables manufacturers to maintain continuous production schedules even during periods of regional supply constraints, guaranteeing on-time delivery for critical medical nutrition clients.
- Scalability and Environmental Compliance: The reaction conditions described operate at mild temperatures and atmospheric pressure, removing the safety barriers often associated with high-pressure hydrogenation reactors. This inherent safety profile simplifies the engineering requirements for scale-up, allowing for seamless transition from pilot batches to multi-ton commercial production without extensive equipment retrofitting. Furthermore, the avoidance of heavy metal catalysts reduces the environmental footprint regarding waste disposal and residual metal testing, aligning with increasingly stringent global regulatory standards for green chemistry and sustainable pharmaceutical manufacturing practices.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and validation of this novel synthesis pathway. These answers are derived directly from the experimental data and process parameters detailed in the patent literature, providing clarity on yield expectations, purity controls, and operational safety. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for commercial supply.
Q: Why is the glutamic acid-based route superior to traditional glutamine-based synthesis?
A: Traditional methods rely on expensive L-glutamine as a starting material, which drives up production costs significantly. The novel route described in patent CN103387600A utilizes L-glutamic acid, a widely available and low-cost fermentation product, thereby drastically reducing raw material expenses while maintaining high stereochemical integrity.
Q: How does the phthalyl protection strategy improve process efficiency?
A: The use of the phthalyl group offers a robust protection strategy that withstands the condensation conditions but can be cleanly removed during the final ammonolysis step. This dual-functionality simplifies the workflow by combining deprotection and side-chain amidation into a single operational unit, reducing solvent usage and processing time.
Q: What represent the key scalability advantages of this manufacturing process?
A: The process operates under mild temperature conditions (0-50°C) and utilizes common industrial solvents like ethyl acetate and toluene. The avoidance of sensitive catalytic hydrogenation steps, which require high-pressure equipment, makes this route inherently safer and easier to scale from pilot plant to multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Alanyl-L-Glutamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient dipeptide synthesis in the modern parenteral nutrition market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of L-alanyl-L-glutamine meets the highest international pharmacopeial standards, providing our partners with the confidence needed to navigate complex regulatory filings and market entry strategies.
We invite forward-thinking procurement leaders to engage with our technical procurement team to discuss how this optimized route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic potential of switching to this glutamic acid-based methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume needs, securing a competitive advantage in the global pharmaceutical intermediates market.
