Advanced Manufacturing of Metoprolol Intermediates: Technical Breakthroughs and Commercial Scalability
The pharmaceutical industry continuously seeks robust synthetic pathways for beta-blockers, with metoprolol remaining a cornerstone therapy for cardiovascular conditions. Patent CN111116328A introduces a refined preparation method for the critical metoprolol intermediate, p-(2-methoxy) ethylphenol, addressing long-standing challenges in cost and scalability. This technical disclosure outlines a three-step sequence starting from p-chlorophenol, utilizing etherification, Grignard addition, and hydrolysis to achieve high purity without relying on precious metal catalysts. For R&D Directors and Procurement Managers, this route represents a significant shift towards more economical and sustainable manufacturing practices. The structural integrity of the final API depends heavily on the quality of this intermediate, making the optimization of its synthesis a priority for supply chain resilience. 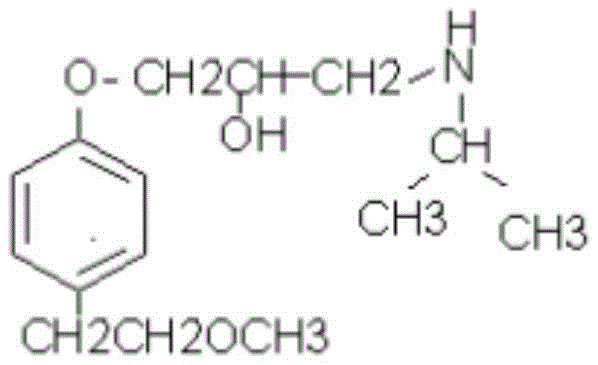
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key phenolic intermediates has often relied on transition metal catalysis, particularly palladium-based systems, which introduce substantial economic and operational burdens. Prior art, such as Patent CN 109553513A, describes routes involving p-bromophenol and methyl vinyl ether under palladium catalysis, which necessitates expensive ligands and rigorous metal scavenging protocols. These stoichiometric metal reagents, including aluminum trichloride in other variations, generate significant hazardous waste streams that complicate environmental compliance and increase disposal costs. Furthermore, the use of high-price initial raw materials like p-bromophenol creates volatility in the supply chain, exposing manufacturers to market fluctuations that can erode profit margins. The operational requirements for maintaining inert atmospheres and handling toxic reagents in these conventional methods also demand specialized equipment and highly trained personnel, limiting accessibility for many production facilities. Consequently, the industry has faced a persistent need for a method that balances chemical efficiency with economic practicality.
The Novel Approach
The patented methodology offers a transformative alternative by leveraging cheap and easily obtained raw materials, specifically p-chlorophenol, to construct the carbon framework efficiently. By replacing precious metal catalysts with magnesium and utilizing dimethyl sulfate for etherification, the process drastically simplifies the reaction conditions while maintaining high selectivity. This novel approach eliminates the need for complex phosphine ligands and the associated downstream purification steps required to remove trace metal contaminants from the final product. The reaction steps are designed for convenient operation, with specific temperature controls ranging from 40°C to 50°C that are easily manageable in standard industrial reactors. Moreover, the strategy incorporates solvent recovery systems for toluene and diethyl ether, ensuring that the process is not only chemically sound but also environmentally responsible. This shift towards base-metal chemistry and recyclable solvents positions the method as a superior choice for cost reduction in API manufacturing.
Mechanistic Insights into Mg-Catalyzed Grignard Addition and Etherification
The core of this synthetic route lies in the formation of the carbon-carbon bond via a Grignard reaction, which is meticulously controlled to ensure high yield and purity. In the second step, p-chloroanisole reacts with magnesium chips in diethyl ether to form the corresponding organomagnesium intermediate, a process that requires careful initiation often aided by iodine crystals to activate the metal surface. Once the Grignard reagent is formed, ethylene oxide is introduced under strict temperature control at 10°C to 20°C to prevent runaway exothermic reactions and ensure regioselective ring opening. This nucleophilic attack on the epoxide extends the carbon chain by two units, creating the essential ethylphenol backbone required for metoprolol. The use of nitrogen protection throughout this stage is critical to prevent moisture ingress, which could quench the reactive Grignard species and lead to significant yield losses. 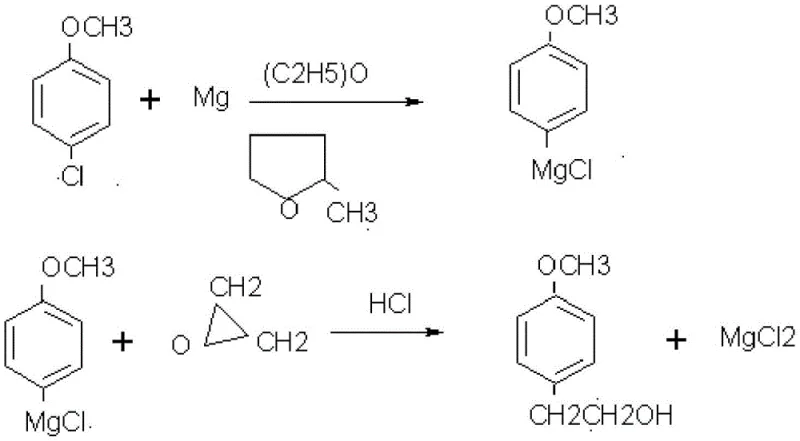
Impurity control is further enhanced in the final step through a transesterification and hydrolysis sequence that avoids the formation of difficult-to-remove byproducts. The reaction of p-methoxyphenethyl alcohol with dimethyl carbonate in toluene, followed by acid hydrolysis with dilute sulfuric acid, ensures that the methoxy group is positioned correctly without generating halogenated impurities common in other routes. The hydrolysis step is conducted at 40°C for 2.5 to 3 hours, allowing for complete conversion while minimizing thermal degradation of the sensitive phenolic structure. By washing the organic layer with sodium hydroxide solution to neutrality, acidic residues are effectively removed, resulting in a crude product that requires minimal purification. This mechanistic precision ensures that the impurity profile remains within stringent specifications, reducing the burden on downstream crystallization processes. The overall pathway demonstrates a deep understanding of reaction kinetics and thermodynamics to optimize the production of high-purity pharmaceutical intermediates.
How to Synthesize p-(2-methoxy) ethylphenol Efficiently
The synthesis of this critical intermediate is structured into three distinct operational phases that can be seamlessly integrated into existing fine chemical manufacturing infrastructure. The process begins with the etherification of p-chlorophenol, followed by the Grignard extension, and concludes with the carbonate-mediated transformation to the final phenol. Each step has been optimized for industrial throughput, with specific attention paid to solvent volumes and reaction times to maximize batch efficiency. Detailed standardized synthetic steps, including precise reagent quantities and safety protocols, are essential for replicating the high yields reported in the patent data. 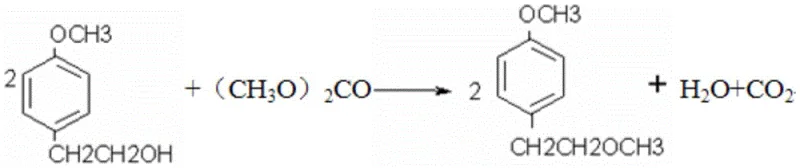
- Etherify p-chlorophenol with dimethyl sulfate and sodium hydroxide in toluene to form p-chloroanisole, ensuring strict temperature control at 40-45°C.
- React p-chloroanisole with magnesium chips in diethyl ether to form a Grignard reagent, followed by the addition of ethylene oxide to yield p-methoxyphenethyl alcohol.
- Treat p-methoxyphenethyl alcohol with dimethyl carbonate in toluene, followed by acid hydrolysis using dilute sulfuric acid to obtain the final p-(2-methoxy) ethylphenol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain leaders, the adoption of this patented route offers tangible benefits that extend beyond simple chemical yield. The elimination of palladium catalysts and the use of commodity chemicals like p-chlorophenol and dimethyl sulfate significantly lower the raw material cost base, providing a buffer against market volatility. This cost structure allows for more competitive pricing strategies while maintaining healthy margins, a critical factor in the highly price-sensitive generic pharmaceutical market. Furthermore, the ability to recycle solvents such as toluene and diethyl ether reduces the volume of hazardous waste requiring disposal, leading to substantial cost savings in environmental compliance and waste management. The robustness of the process ensures consistent quality, reducing the risk of batch failures that can disrupt supply continuity and damage customer relationships.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with magnesium and the use of readily available starting materials directly translates to a lower cost of goods sold. By avoiding the procurement of high-price reagents like p-bromophenol and palladium ligands, manufacturers can achieve significant operational expenditure reductions. The process design also minimizes the need for specialized metal scavenging resins, which are often a hidden cost in traditional catalytic routes. This economic efficiency makes the route highly attractive for large-scale production where margin compression is a constant challenge.
- Enhanced Supply Chain Reliability: Relying on bulk commodity chemicals ensures that raw material availability is not a bottleneck for production scheduling. Unlike specialized catalysts that may have long lead times or single-source dependencies, the reagents for this process are widely available from multiple global suppliers. This diversification of the supply base mitigates the risk of shortages and allows for more flexible inventory management strategies. Additionally, the stability of the intermediates allows for safer storage and transportation, further enhancing the reliability of the supply chain for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, with reaction conditions that are easily managed in large-volume reactors without requiring exotic high-pressure or high-temperature equipment. The ability to recover and reuse solvents aligns with green chemistry principles, reducing the environmental footprint of the manufacturing facility. This compliance with environmental standards facilitates smoother regulatory approvals and reduces the risk of production shutdowns due to waste disposal limits. The simplified work-up procedures also reduce the time required for batch turnover, increasing overall plant capacity and throughput.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential manufacturing partners. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production lines. The answers reflect a commitment to transparency and technical excellence in the delivery of chemical solutions.
Q: How does this patent method improve upon conventional palladium-catalyzed routes?
A: Conventional methods often rely on expensive palladium catalysts and phosphine ligands, which significantly increase raw material costs and require complex metal removal steps. The patented method utilizes magnesium and cheap reagents like dimethyl sulfate, eliminating the need for precious metals and simplifying the purification process for better commercial viability.
Q: What are the solvent recovery capabilities in this manufacturing process?
A: The process is designed for high efficiency, utilizing toluene and diethyl ether as primary solvents which can be distilled and recycled for subsequent batches. This closed-loop solvent management drastically reduces waste disposal costs and enhances the overall environmental compliance of the production facility.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the patent explicitly details operations suitable for industrial reactors, including specific temperature controls (40-50°C) and large-volume solvent handling (500L+). The use of readily available raw materials like p-chlorophenol ensures supply chain stability for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-(2-methoxy) ethylphenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final drug product. Our CDMO expertise allows us to adapt complex synthetic pathways like the one described in Patent CN111116328A to meet your specific volume and purity requirements. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our rigorous QC labs and stringent purity specifications guarantee that every batch of p-(2-methoxy) ethylphenol meets the highest industry standards, minimizing the risk of downstream processing issues.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through advanced chemical engineering. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our capabilities can support your long-term business goals. Together, we can achieve greater efficiency and reliability in the production of essential pharmaceutical materials.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
