Optimizing Ezetimibe Production: A Novel Lewis Acid-Catalyzed Route for High-Purity Intermediates
The pharmaceutical landscape for cardiovascular therapeutics continues to evolve, driven by the demand for safer, more efficient manufacturing processes for critical drugs like Ezetimibe. Patent CN112441959A introduces a transformative synthesis pathway that addresses long-standing inefficiencies in producing this cholesterol absorption inhibitor. Unlike legacy methods relying on hazardous reagents and complex multi-step sequences, this innovation employs a sophisticated Lewis acid-catalyzed condensation strategy. By utilizing a dual titanium catalyst system comprising titanium tetrachloride and tetraisopropyl titanate, the process achieves exceptional stereocontrol under remarkably mild conditions ranging from -20°C to 0°C. This technical breakthrough not only streamlines the production of key intermediates but also establishes a robust foundation for commercial scale-up of complex pharmaceutical intermediates, ensuring that supply chains can meet the rigorous quality standards required for global regulatory approval.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Ezetimibe has been plagued by significant operational hurdles that inflate costs and complicate supply chain logistics. Early methodologies, such as those disclosed in EP0720599 and CN1131416, relied heavily on Grignard reagents and Palladium-catalyzed cross-coupling reactions. These processes necessitated ultra-low temperature environments and high-pressure hydrogenation equipment, creating substantial safety risks and capital expenditure barriers for manufacturers. Furthermore, the reliance on transition metal catalysts introduced severe downstream purification challenges, requiring expensive metal scavenging steps to meet strict residual metal limits in Active Pharmaceutical Ingredients (APIs). Other approaches, like those in WO0034240, attempted to shorten the route but suffered from low yields during chiral reduction steps and poor intermediate purity, often mandating resource-intensive chromatographic separations that are impractical for multi-ton production.
The Novel Approach
The methodology outlined in CN112441959A represents a paradigm shift by replacing hazardous metal catalysis with a controlled Lewis acid mediation system. This novel route initiates with the simultaneous trimethylsilyl (TMS) protection of both the ketone substrate (SM1) and the imine partner (SM2), effectively masking reactive hydroxyl groups to prevent premature side reactions. The core transformation involves a highly selective condensation facilitated by a specific molar ratio of titanium tetrachloride and tetraisopropyl titanate. This dual-catalyst approach ensures precise activation of the electrophile while maintaining the integrity of the chiral centers. Following condensation, a mild deprotection sequence using aqueous tartaric acid yields the critical intermediate INT2 with purity exceeding 99%. This strategic redesign eliminates the need for cryogenic conditions and precious metal catalysts, directly contributing to cost reduction in cardiovascular drug manufacturing by simplifying reactor requirements and waste treatment protocols.
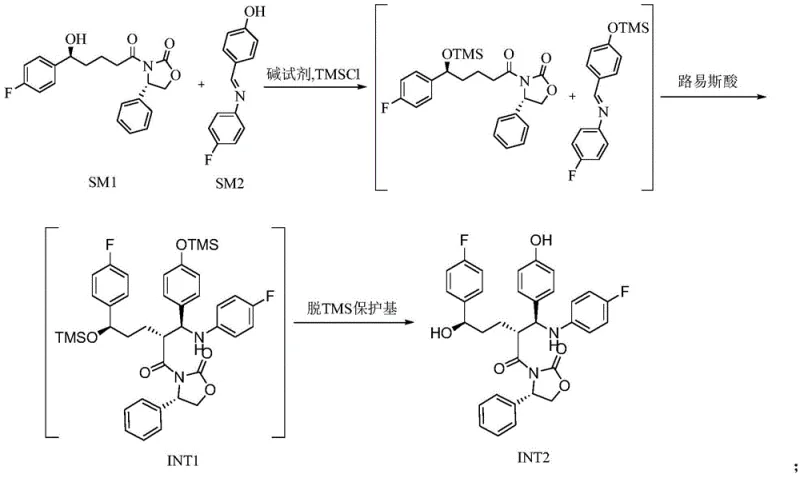
Mechanistically, the success of this synthesis hinges on the precise orchestration of steric and electronic factors during the carbon-carbon bond-forming event. The use of the (4S)-3-[(5S)-5-(4-fluorophenyl)-5-hydroxy-1-oxopentyl]-4-phenyl-2-oxazolidinone (SM1) serves a dual purpose: it acts as a chiral pool precursor and provides a rigid structural framework that directs the incoming nucleophile. When the TMS-protected species interact in the presence of the titanium complex, the Lewis acid coordinates with the carbonyl oxygen, increasing its electrophilicity without inducing racemization. The subsequent addition of the protected phenol derivative occurs with high diastereoselectivity, governed by the existing chiral environment of the oxazolidinone ring. This intrinsic stereocontrol is superior to external chiral catalysts, as it is built into the molecular architecture of the starting material. Furthermore, the isolation of intermediate INT2 acts as a crucial purification checkpoint, effectively filtering out any minor diastereomers or unreacted starting materials before the final cyclization step, thereby guaranteeing the high optical purity observed in the final API.
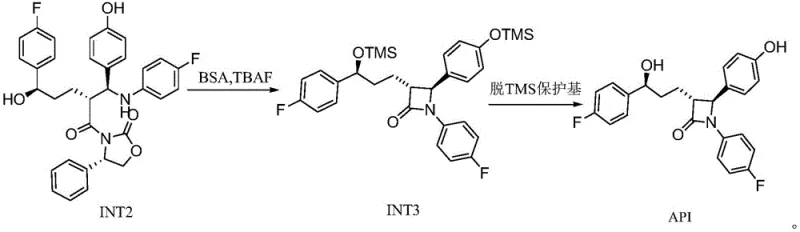
Impurity control is further enhanced by the specific choice of deprotection reagents. The use of dilute tartaric acid for the removal of TMS groups in the first stage is particularly ingenious; it is mild enough to prevent the hydrolysis of the sensitive beta-lactam precursor moieties while being effective at cleaving the silyl ethers. In the final cyclization stage, the deployment of tetrabutylammonium fluoride (TBAF) facilitates an intramolecular attack to close the four-membered azetidinone ring. The mechanism involves the fluoride ion activating the silyl-protected hydroxyl group, generating a transient alkoxide that attacks the adjacent amide carbonyl. This fluoride-mediated cyclization is kinetically favorable and proceeds with minimal epimerization, a common pitfall in beta-lactam formation. The final acid treatment with sulfuric acid cleanly removes the remaining protecting groups, yielding the target molecule with a chemical purity of not less than 99.5% and optical purity surpassing 99.9%, demonstrating the robustness of this mechanistic design against impurity generation.
How to Synthesize Ezetimibe Efficiently
The practical execution of this synthesis protocol requires careful attention to stoichiometry and temperature control to maximize the benefits of the Lewis acid system. The process is divided into two distinct phases: the preparation of the linear intermediate INT2 and the subsequent cyclization to the final beta-lactam structure. Operators must ensure that the TMS protection is complete before introducing the titanium catalysts to avoid oligomerization side products. The reaction mixture should be maintained strictly within the -20°C to 0°C window during the condensation phase to preserve stereochemical integrity. Following the reaction, the quenching procedure with isopropanol and subsequent washing with sodium bisulfite is critical for removing excess oxidants and metal residues. For the final cyclization, the use of N,O-bis(trimethylsilyl)acetamide (BSA) ensures full silylation of the hydroxyl groups prior to TBAF addition, which is essential for driving the ring-closure equilibrium towards the product. Detailed standardized operating procedures for these steps are essential for maintaining batch-to-batch consistency in a GMP environment.
- Prepare Intermediate INT2 by reacting SM1 and SM2 with TMSCl under alkaline conditions, followed by Lewis acid condensation using TiCl4 and tetraisopropyl titanate, and subsequent deprotection.
- Convert INT2 to the final API by protecting hydroxyl groups with BSA, inducing cyclization with TBAF to form the beta-lactam ring, and final acid-mediated deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthesis route offers tangible strategic benefits that extend beyond simple yield improvements. The elimination of Palladium catalysts and Grignard reagents removes a significant layer of complexity from the raw material sourcing matrix. Precious metals are subject to volatile market pricing and geopolitical supply risks; by substituting these with abundant titanium salts, manufacturers can stabilize their Bill of Materials (BOM) costs and reduce exposure to supply shocks. Additionally, the avoidance of high-pressure hydrogenation equipment lowers the barrier to entry for contract manufacturing organizations (CMOs), expanding the pool of qualified reliable ezetimibe intermediate supplier partners capable of executing the campaign. This diversification of the supply base enhances overall supply chain resilience and reduces the risk of production bottlenecks.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the simplification of the purification train. Traditional routes often require multiple chromatographic columns to remove metal residues and separate diastereomers, which consumes vast quantities of silica gel and solvents. In contrast, this novel method relies on crystallization and aqueous washes for purification, significantly reducing solvent consumption and waste disposal costs. The higher overall yield means less starting material is required per kilogram of finished API, directly lowering the variable cost of goods sold. Furthermore, the mild reaction conditions reduce energy consumption associated with cryogenic cooling and high-pressure systems, contributing to a leaner and more cost-effective manufacturing footprint.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions translates directly into improved delivery reliability. Processes that operate at ambient or near-ambient pressures and moderate temperatures are less prone to equipment failures and safety shutdowns compared to those requiring extreme conditions. The use of stable, commercially available reagents like TMSCl and DIPEA ensures that production schedules are not disrupted by the long lead times often associated with specialized chiral catalysts or organometallic reagents. This stability allows for more accurate forecasting and inventory planning, enabling pharmaceutical companies to maintain optimal stock levels of this critical lipid-lowering agent without the need for excessive safety stock.
- Scalability and Environmental Compliance: As regulatory pressure mounts regarding environmental sustainability, the green chemistry attributes of this process become a major competitive advantage. The reduction in heavy metal usage simplifies wastewater treatment and aligns with increasingly stringent environmental regulations in key manufacturing hubs. The ability to telescope certain steps or perform continuous processing, as suggested by the smooth reaction profile, facilitates seamless scale-up from pilot plant to commercial tonnage. This scalability ensures that reducing lead time for high-purity statin combinations is achievable, allowing suppliers to respond rapidly to surges in market demand for Ezetimibe combination therapies without compromising on quality or compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term viability of the supply source. The answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering a clear picture of the operational realities.
Q: Why is the new Lewis acid method preferred over traditional Palladium-catalyzed routes?
A: Traditional routes often require expensive Palladium catalysts and harsh conditions like ultra-low temperatures or high-pressure hydrogenation. The novel Lewis acid method utilizes Titanium tetrachloride and tetraisopropyl titanate under mild conditions (-20 to 0°C), eliminating the need for costly metal scavenging and improving safety profiles for industrial scale-up.
Q: How does this process ensure high optical purity for Ezetimibe?
A: The process leverages chiral starting materials (SM1) containing an oxazolidinone auxiliary, which controls stereochemistry from the outset. By avoiding racemization-prone steps like chiral reduction found in older patents, and isolating the high-purity intermediate INT2, the final product achieves optical purity exceeding 99.9% without complex chromatographic purification.
Q: What are the key advantages of the TMS protection strategy in this synthesis?
A: The use of Trimethylsilyl (TMS) protecting groups on both reactants prevents side reactions during the condensation phase. This strategy enhances reaction selectivity and yield. Furthermore, the reversible nature of TMS protection allows for efficient deprotection using mild acids like tartaric acid or sulfuric acid, simplifying the workup and reducing waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ezetimibe Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthesis platforms requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the Lewis acid-catalyzed route are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped to verify the optical purity and chemical identity of every batch, guaranteeing that our intermediates meet the exacting standards required for downstream API synthesis. Our commitment to quality assurance ensures that the high purity profiles described in the patent are consistently delivered to our global clientele.
We invite pharmaceutical developers and procurement leaders to engage with our technical procurement team to discuss how this optimized route can enhance your supply chain efficiency. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how switching to this intermediate can lower your total landed costs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring a seamless integration of high-quality Ezetimibe intermediates into your manufacturing pipeline.
