Optimizing Febuxostat Production: A Novel One-Pot Synthesis Strategy for Commercial Scale
Optimizing Febuxostat Production: A Novel One-Pot Synthesis Strategy for Commercial Scale
The pharmaceutical industry is constantly seeking robust, scalable, and safe methodologies for the production of critical active pharmaceutical ingredients (APIs) such as Febuxostat, a potent non-purine xanthine oxidase inhibitor. Patent CN110229117B introduces a groundbreaking preparation method that fundamentally restructures the synthetic pathway for Febuxostat and its key intermediates. This technology shifts away from fragmented, hazardous multi-step processes towards a streamlined, one-pot synthesis strategy that integrates alkylation, oximation, and hydrolysis within a single reaction vessel. By leveraging this advanced protocol, manufacturers can achieve superior control over impurity profiles, specifically targeting the reduction of genotoxic contaminants that pose significant regulatory hurdles. The innovation lies not just in the chemical transformation but in the holistic process engineering that facilitates easier solid-liquid separation and solvent recovery, marking a significant leap forward in the industrial feasibility of producing high-quality gout therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Febuxostat has been plagued by significant safety and efficiency challenges inherent to traditional organic synthesis routes. The original patented pathways often relied on the use of highly toxic reagents such as cuprous cyanide and potassium cyanide to introduce the critical cyano group, presenting severe occupational health risks and complicating waste disposal protocols. Furthermore, these legacy methods frequently necessitated dangerous diazotization reactions and noble metal-catalyzed hydrogenation steps, which require specialized high-pressure equipment and rigorous safety monitoring. The reliance on multiple solvent systems, including formic acid which causes severe equipment corrosion at elevated temperatures, further exacerbated operational costs and maintenance requirements. Additionally, the fragmentation of the synthesis into isolated steps often resulted in cumulative yield losses and increased the likelihood of cross-contamination, making the consistent production of ultra-high purity API difficult to maintain on a commercial scale without extensive and costly purification measures.
The Novel Approach
In stark contrast to these cumbersome legacy routes, the novel methodology described in the patent data employs a cohesive, telescoped strategy that dramatically simplifies the manufacturing landscape. The process initiates with the efficient alkylation of the phenolic precursor using bromoisobutane in the presence of a mild organic base, yielding the crucial Intermediate I (Formula II) with high selectivity. 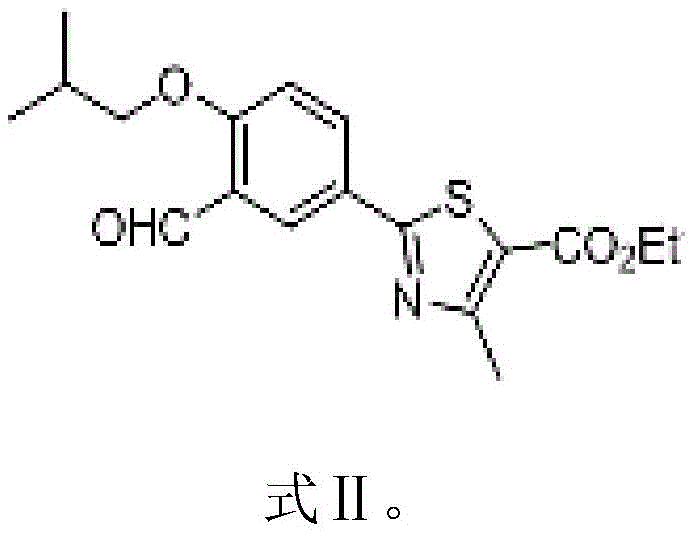 This intermediate is then seamlessly converted into Intermediate II (Formula III) through a direct oximation and dehydration sequence using hydroxylamine hydrochloride and acetyl chloride, effectively bypassing the need for toxic cyanide sources.
This intermediate is then seamlessly converted into Intermediate II (Formula III) through a direct oximation and dehydration sequence using hydroxylamine hydrochloride and acetyl chloride, effectively bypassing the need for toxic cyanide sources. 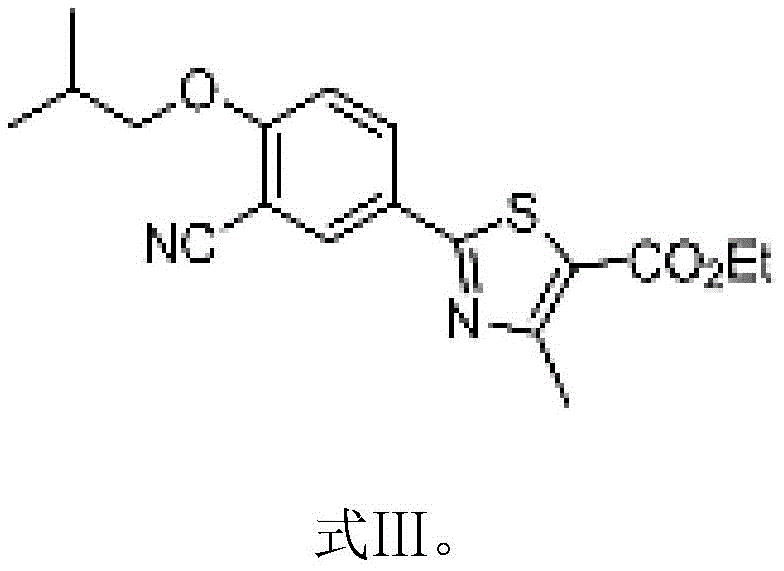 By maintaining the reaction within a single solvent system, typically dimethylformamide (DMF) or dimethylacetamide (DMAC), the process eliminates the need for intermediate isolation and drying, thereby preserving product integrity and minimizing thermal degradation. This integrated approach not only enhances the overall throughput but also aligns perfectly with green chemistry principles by reducing solvent consumption and simplifying the downstream processing train.
By maintaining the reaction within a single solvent system, typically dimethylformamide (DMF) or dimethylacetamide (DMAC), the process eliminates the need for intermediate isolation and drying, thereby preserving product integrity and minimizing thermal degradation. This integrated approach not only enhances the overall throughput but also aligns perfectly with green chemistry principles by reducing solvent consumption and simplifying the downstream processing train.
Mechanistic Insights into One-Pot Telescoped Synthesis
The core of this technological advancement lies in the precise orchestration of reaction conditions that allow for sequential transformations without compromising yield or purity. The initial alkylation step utilizes triethylamine as a catalyst and acid scavenger, operating optimally at temperatures between 70°C and 80°C to ensure complete conversion of the hydroxyl group while minimizing side reactions. Following this, the introduction of hydroxylamine hydrochloride facilitates the formation of the oxime, which is subsequently dehydrated by acetyl chloride to generate the nitrile functionality essential for the drug's pharmacological activity. The entire sequence is meticulously controlled to prevent the accumulation of deleterious by-products, with the reaction temperature carefully managed during the acetyl chloride addition phase to avoid exothermic runaways. 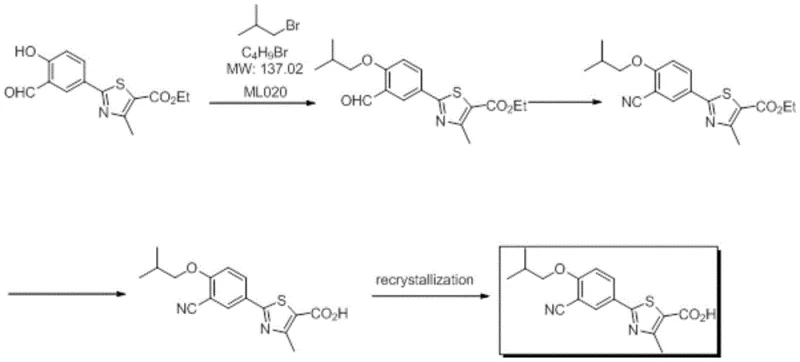 The final hydrolysis step is equally critical, where the addition of water and an organic base like sodium hydroxide cleaves the ester moiety to reveal the free carboxylic acid. The patent highlights a unique 'wet cake' processing technique where the crude product is not dried prior to recrystallization; instead, it is directly dissolved in a methanol-water mixture. This specific maneuver prevents the potential polymorphic changes or degradation that can occur during the drying of the intermediate, ensuring that the final API possesses the desired crystal habit and purity profile required for bioavailability.
The final hydrolysis step is equally critical, where the addition of water and an organic base like sodium hydroxide cleaves the ester moiety to reveal the free carboxylic acid. The patent highlights a unique 'wet cake' processing technique where the crude product is not dried prior to recrystallization; instead, it is directly dissolved in a methanol-water mixture. This specific maneuver prevents the potential polymorphic changes or degradation that can occur during the drying of the intermediate, ensuring that the final API possesses the desired crystal habit and purity profile required for bioavailability.
Impurity control is a paramount concern in the synthesis of complex heterocyclic APIs, and this process demonstrates exceptional capability in suppressing genotoxic impurities. The avoidance of cyanide salts inherently removes a major class of genotoxic risks, while the use of high-purity solvents and controlled reaction stoichiometry minimizes the formation of alkyl halides and other reactive species. The patent data indicates that the final product consistently exhibits total impurities of less than 0.1% and single impurities below 0.05%, with thirteen specific genotoxic impurities remaining undetected. This level of purity is achieved through the synergistic effect of the one-pot design, which limits exposure to external contaminants, and the optimized crystallization protocol that effectively purges trace solvents and minor by-products. For R&D directors, this implies a robust platform where the critical quality attributes (CQAs) of the API are built into the process design rather than relying solely on end-of-pipe purification, significantly de-risking the regulatory filing and validation phases.
How to Synthesize Febuxostat Efficiently
The implementation of this synthesis route requires a disciplined approach to reaction parameter control and equipment selection to fully realize its commercial potential. The process is designed to be executed in a standard glass-lined or stainless steel reactor capable of handling mild exotherms and corrosive by-products like hydrochloric acid generated during the dehydration step. Operators must adhere to strict temperature profiles, particularly during the addition of acetyl chloride and the subsequent hydrolysis, to maintain the delicate balance between reaction rate and selectivity. The detailed standardized synthesis steps outlined below provide a roadmap for scaling this chemistry from laboratory benchtop to pilot plant and eventually to full commercial production, ensuring reproducibility and safety at every stage.
- Perform alkylation of ethyl 2-(3-formyl-4-hydroxyphenyl)-4-methylthiazole-5-carboxylate with bromoisobutane using triethylamine in DMF/DMAC at 70-80°C.
- Directly add hydroxylamine hydrochloride and acetyl chloride to the reaction mixture for oximation and dehydration to form the nitrile intermediate.
- Hydrolyze the intermediate with aqueous base, adjust pH to 6.0-7.0, and crystallize the crude product without drying before final recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this novel synthesis method offers transformative benefits that directly impact the bottom line and operational resilience. The consolidation of multiple reaction steps into a single vessel drastically reduces the requirement for intermediate storage tanks, transfer lines, and associated cleaning validation procedures, leading to a significantly simplified facility footprint. By utilizing a single solvent system throughout the majority of the process, the complexity of solvent recovery and recycling is minimized, allowing for more efficient resource utilization and a reduction in the volume of hazardous waste requiring disposal. The elimination of the drying step for the crude intermediate not only saves energy but also shortens the overall cycle time, enabling faster turnover of production batches and improved responsiveness to market demand fluctuations. These operational efficiencies translate into a more competitive cost structure, making the supply of high-quality Febuxostat more sustainable and reliable in the long term.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the reduction in unit operations and the optimization of raw material usage. By removing the need for expensive and hazardous reagents like cuprous cyanide and noble metal catalysts, the direct material costs are substantially lowered. Furthermore, the ability to recover and reuse the primary solvent (DMF or DMAC) through simple gravity bed techniques reduces the recurring expenditure on fresh solvents. The streamlined workflow also decreases labor hours and utility consumption per kilogram of API produced, resulting in a leaner manufacturing model that is less susceptible to cost volatility in the supply chain.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on specialized reagents or complex multi-vendor supply chains for intermediates. This one-pot method mitigates such risks by utilizing readily available commodity chemicals such as bromoisobutane and hydroxylamine hydrochloride, which are widely sourced and less prone to supply disruptions. The robustness of the reaction conditions, which tolerate slight variations without significant yield loss, ensures consistent output even under variable operating environments. This reliability allows supply chain managers to forecast production schedules with greater confidence and maintain lower safety stock levels, freeing up working capital and reducing inventory holding costs.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but the温和 (mild) nature of this reaction sequence facilitates smooth scale-up from pilot to commercial volumes. The process generates fewer hazardous by-products and avoids the use of heavy metals, simplifying the environmental permitting process and reducing the burden on wastewater treatment facilities. Compliance with increasingly stringent environmental regulations is easier to achieve, as the process aligns with green chemistry metrics by maximizing atom economy and minimizing waste generation. This environmental stewardship not only protects the corporate reputation but also future-proofs the manufacturing site against tightening regulatory frameworks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced Febuxostat synthesis technology. These insights are derived directly from the patent specifications and experimental data, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for stakeholders evaluating the feasibility of adopting this route for their own manufacturing portfolios or sourcing strategies.
Q: How does this novel process improve safety compared to traditional Febuxostat synthesis?
A: Traditional routes often involve hazardous reagents like cuprous cyanide and potassium cyanide for nitrile introduction, alongside dangerous diazotization steps. This novel method eliminates the need for toxic cyanide salts and noble metal hydrogenation, significantly reducing operational risks and environmental hazards while simplifying waste treatment protocols.
Q: What are the purity specifications achievable with this one-pot method?
A: The process is designed to achieve exceptional purity levels, consistently exceeding 99.9% by HPLC. Crucially, it controls genotoxic impurities to below detection limits (standard ≤ 15 ppm) and ensures single impurities remain under 0.05%, meeting stringent regulatory requirements for API manufacturing without complex purification steps.
Q: Does this method offer advantages in solvent recovery and cost efficiency?
A: Yes, the process utilizes a single solvent system (DMF or DMAC) throughout the reaction sequence, which greatly simplifies solvent recovery via gravity bed techniques. By avoiding multiple solvent swaps and eliminating the drying step of the crude intermediate, the method reduces energy consumption and processing time, leading to substantial overall cost reductions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Febuxostat Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our team of expert chemists and process engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering Febuxostat and its intermediates with stringent purity specifications, leveraging our rigorous QC labs to verify that every batch meets the highest international standards for safety and efficacy. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required by this novel one-pot process, guaranteeing a consistent and high-quality supply for our partners.
We invite potential collaborators to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific project requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener, more efficient manufacturing method. We encourage you to request specific COA data and route feasibility assessments to validate the superior impurity profile and scalability of our Febuxostat offerings. Let us work together to drive down costs and enhance the reliability of your supply chain for this vital therapeutic agent.
