Advanced Manufacturing of High-Purity Febuxostat Intermediates via Precision Reagent Purification
The pharmaceutical landscape for gout management has been significantly transformed by the introduction of non-purine xanthine oxidase inhibitors, with Febuxostat standing out as a critical therapeutic agent. As detailed in patent CN103012311A, the demand for ultra-high purity intermediates in this sector is driven by stringent regulatory requirements regarding genotoxic impurities and structural isomers. This technical disclosure presents a breakthrough methodology that addresses a persistent bottleneck in the supply chain: the control of the n-propoxy impurity during the etherification stage. By implementing a rigorous pre-reaction purification of the alkylating agent, manufacturers can achieve impurity profiles well below the 0.10% threshold, ensuring compliance with ICH guidelines for global markets.
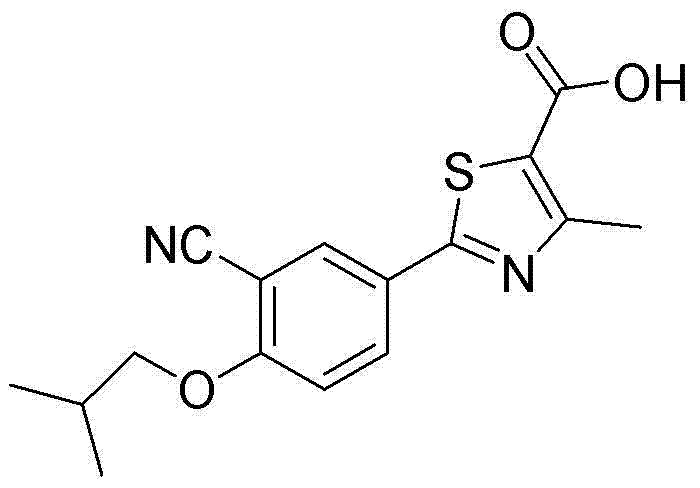
For procurement leaders and R&D directors, understanding the structural nuances of the target molecule is paramount. The presence of the isobutoxy group at the 4-position of the phenyl ring is essential for biological activity, yet its installation via nucleophilic substitution is fraught with risks if reagent quality is not meticulously managed. The patent highlights that traditional sourcing of isobutane bromide often introduces isomeric contaminants that lead to inseparable byproducts, complicating downstream purification and increasing overall production costs. This insight underscores the necessity of vertical integration or strict vendor qualification for key starting materials in the fine chemical supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Febuxostat has relied on several distinct pathways, each carrying inherent disadvantages that impact commercial viability and environmental sustainability. One prominent route involves the reduction of a nitro precursor using palladium on carbon, a process that necessitates the use of expensive noble metal catalysts and poses significant safety risks associated with hydrogenation on a large scale. Furthermore, the subsequent diazotization and Sandmeyer cyanidation steps generate substantial quantities of heavy metal waste, creating a burden for wastewater treatment facilities and increasing the environmental footprint of the manufacturing site.
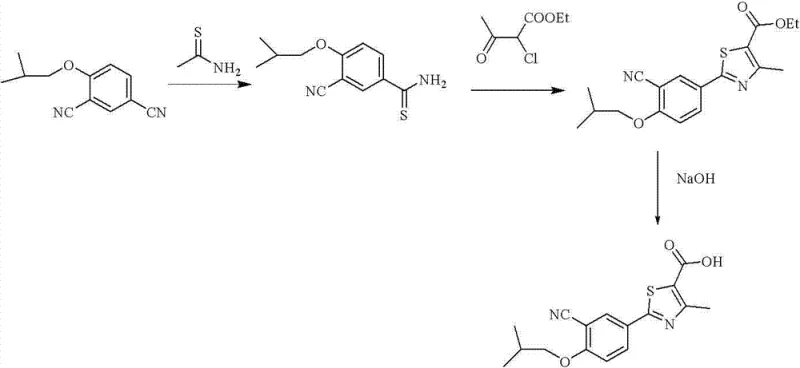
Another common approach utilizes thioacetamide condensation followed by cyclization with chloroacetoacetic ester. While this avoids hydrogenation, it often suffers from lower regioselectivity and requires harsh reaction conditions that can degrade sensitive functional groups. In both conventional scenarios, the final purification of the active pharmaceutical ingredient (API) is complicated by the presence of structurally similar impurities that co-elute during chromatography or co-crystallize, leading to yield losses. These inefficiencies translate directly into higher cost of goods sold (COGS) and extended lead times for batch release, which are critical pain points for supply chain managers aiming to maintain inventory stability.
The Novel Approach
The methodology disclosed in the patent represents a strategic pivot towards preventative quality control rather than corrective purification. By modifying the established aldehyde-based route, the inventors have identified that the root cause of the most stubborn impurity lies in the quality of the isobutane bromide reagent. The novel process integrates a fractional distillation step to refine the commercial bromide before it ever enters the reactor, effectively eliminating the 1-bromopropane contaminant at the source. This proactive measure simplifies the downstream workup, as the resulting etherification product contains negligible amounts of the n-propoxy analog.
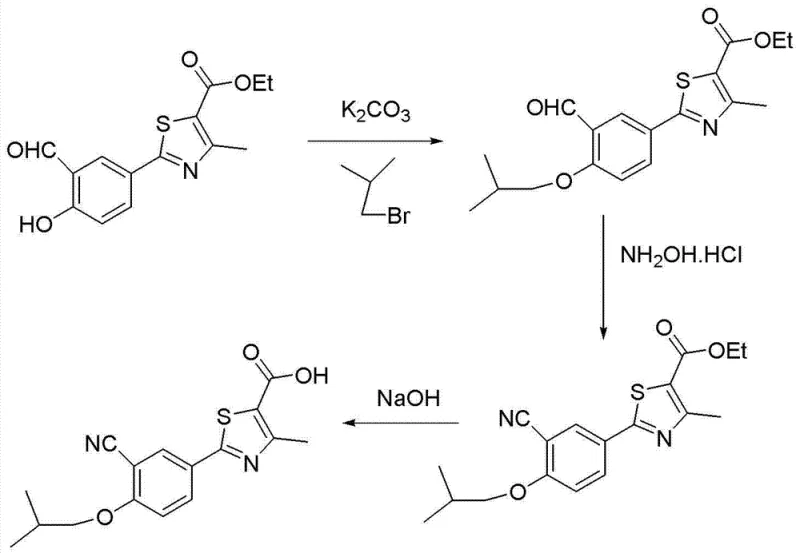
Following the etherification with the purified reagent, the process proceeds through a robust cyanidation using hydroxylamine hydrochloride and subsequent hydrolysis. This sequence avoids the use of toxic cyanide salts typically associated with Sandmeyer reactions, offering a safer operational profile for plant personnel. The final recrystallization from acetone serves as a polishing step to remove any trace mechanical impurities or residual solvents, yielding a product that meets the rigorous specifications required for generic drug filings. This streamlined approach not only enhances the chemical purity but also improves the overall mass balance of the process, making it highly attractive for cost-sensitive generic manufacturing.
Mechanistic Insights into Impurity Formation and Control
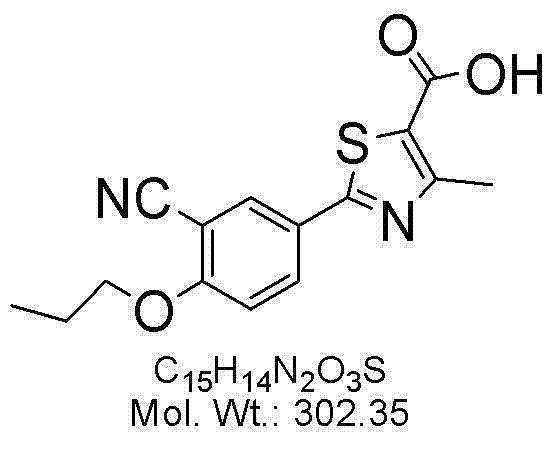
To fully appreciate the technical sophistication of this method, one must examine the mechanistic pathway of the side reaction that generates the critical impurity. During the Williamson ether synthesis, the phenoxide anion attacks the alkyl halide in an SN2 fashion. If the commercial isobutane bromide contains even trace amounts of 1-bromopropane, the phenoxide will indiscriminately attack this linear isomer due to its comparable reactivity and slightly lower steric hindrance. This results in the formation of 2-(3-cyano-4-n-propoxyphenyl)-4-methylthiazole-5-carboxylic acid, a structural isomer that is notoriously difficult to separate from the desired isobutoxy product due to their nearly identical polarity and solubility characteristics.
The patent elucidates that by controlling the boiling point cut of the isobutane bromide to a narrow range of 92.5-93°C at 100kPa, the concentration of 1-bromopropane can be reduced to less than 0.10%. This level of precision in reagent preparation is the linchpin of the entire process. By ensuring the alkylating agent is essentially free of the linear isomer, the competitive side reaction is kinetically suppressed. Consequently, the crude product emerging from the cyanidation and hydrolysis steps already possesses a purity profile that is significantly superior to that obtained via traditional routes, reducing the load on the final crystallization step and maximizing the recovery of the valuable API intermediate.
Furthermore, the choice of solvent and base in the etherification step plays a crucial role in optimizing the reaction kinetics. The use of dimethylformamide (DMF) as a polar aprotic solvent enhances the nucleophilicity of the phenoxide ion, while potassium carbonate serves as a mild yet effective base that minimizes hydrolysis of the ester moiety during the alkylation. This careful balancing of reaction parameters ensures that the conversion to the desired ether is high, while the integrity of the thiazole ring and the ester group is maintained throughout the multi-step sequence, demonstrating a deep understanding of physical organic chemistry principles applied to process development.
How to Synthesize High-Purity Febuxostat Efficiently
The implementation of this synthesis strategy requires a disciplined approach to unit operations, particularly regarding the initial purification of raw materials. Operators must be trained to monitor distillation columns closely to ensure the precise boiling point cut is maintained, as deviations can reintroduce the very impurities the process seeks to eliminate. The subsequent etherification is conducted under controlled thermal conditions to prevent thermal degradation, followed by a straightforward aqueous workup that isolates the intermediate aldehyde in high yield. For a comprehensive breakdown of the specific temperatures, stoichiometry, and monitoring techniques required to replicate this success, please refer to the standardized operating procedures outlined below.
- Purify commercial isobutane bromide via fractional distillation at 92.5-93°C (100kPa) to reduce 1-bromopropane content below 0.10%.
- Perform etherification of 2-(3-formyl-4-hydroxyphenyl)-4-methylthiazole-5-carboxylate with the refined bromide in DMF using potassium carbonate.
- Execute cyaniding with hydroxylamine hydrochloride followed by alkaline hydrolysis and acetone recrystallization to obtain the final acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this refined synthesis route offers compelling advantages that extend beyond mere chemical elegance. For procurement managers, the ability to utilize commercially available starting materials, provided they undergo a simple in-house distillation, reduces dependency on specialized custom synthesis vendors who often charge premiums for high-purity reagents. This decentralization of quality control allows manufacturing sites to buffer against supply shocks in the raw material market, ensuring continuous production schedules even when external supply chains face disruptions. The elimination of expensive palladium catalysts and toxic cyanide salts further contributes to a leaner cost structure.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the need for noble metal catalysts and complex heavy metal scavenging steps. By shifting the purification burden to a simple distillation of a low-cost alkyl halide, the overall consumption of high-value reagents is drastically reduced. Additionally, the improved yield in the final crystallization step, resulting from a cleaner crude profile, means that less raw material is wasted per kilogram of finished product, directly enhancing the gross margin of the manufacturing operation.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route translates into greater predictability for supply chain planners. Because the critical quality attribute (impurity level) is controlled at the very beginning of the synthesis via reagent refinement, the risk of batch failure at the final stage is minimized. This reliability reduces the need for safety stock and allows for more agile response to market demand fluctuations. Furthermore, the avoidance of hazardous reagents simplifies logistics and storage requirements, reducing the regulatory burden associated with transporting dangerous goods.
- Scalability and Environmental Compliance: The process is inherently scalable, relying on standard chemical engineering unit operations such as distillation, filtration, and crystallization that are easily transferred from pilot plant to commercial scale. The absence of heavy metal waste streams simplifies effluent treatment, aligning with increasingly stringent environmental regulations globally. This 'green chemistry' aspect not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the manufacturer, which is becoming a key differentiator in B2B procurement decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Febuxostat intermediates using this patented technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer or process validation.
Q: What is the primary impurity challenge in standard Febuxostat synthesis?
A: The formation of 2-(3-cyano-4-n-propoxyphenyl)-4-methylthiazole-5-carboxylic acid, caused by 1-bromopropane contamination in the alkylating agent.
Q: How does the patented method control impurity levels?
A: By strictly refining the isobutane bromide reagent through distillation to remove isomeric propyl bromides prior to the etherification reaction.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the method utilizes standard unit operations like distillation and crystallization, avoiding expensive catalysts and facilitating scalable manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Febuxostat Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and unwavering commitment to quality. Our facility is equipped to handle the precise distillation and controlled reaction conditions necessary to execute this high-purity synthesis route effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our rigorous QC labs employ state-of-the-art analytical methods to verify that every batch meets stringent purity specifications, particularly regarding the critical n-propoxy impurity levels.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing process can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic impact of switching to this impurity-controlled route. We are prepared to provide specific COA data and route feasibility assessments to support your regulatory filings and ensure a seamless integration of our high-quality intermediates into your final drug product manufacturing.
