Advanced Asymmetric Synthesis of Chiral Spiroindane Skeletons for Ligand Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient routes to access complex chiral scaffolds, particularly those serving as privileged structures for asymmetric catalysis. Patent CN106365949B introduces a groundbreaking methodology for the preparation of chiral spiro-dihydroindene skeleton compounds, addressing critical bottlenecks in ligand synthesis. Unlike conventional approaches that depend on costly chiral pool starting materials or inefficient resolution processes, this invention leverages a sophisticated sequence of asymmetric hydrogenation followed by Lewis acid-catalyzed intramolecular Friedel-Crafts cyclization. The result is a robust pathway capable of delivering products with exceptional optical purity, often exceeding 99% ee, and high chemical yields. This technological leap represents a significant advancement for manufacturers aiming to secure a reliable pharmaceutical intermediate supplier status while minimizing environmental impact and production costs associated with chiral separation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically pure spiroalkane molecules has been fraught with inefficiencies that hinder large-scale adoption. Traditional strategies predominantly rely on the resolution of racemates or diastereoisomers, a process that is inherently wasteful as it discards at least half of the synthesized material. Furthermore, these resolution techniques necessitate the use of expensive chiral resolving agents and involve complicated operational procedures such as repeated crystallizations or chromatographic separations. The reliance on optically pure starting materials like (R)- or (S)-1,1'-spirodihydroindene-7,7'-biphenol derivatives further escalates raw material costs and limits substrate diversity. From a supply chain perspective, these constraints create vulnerabilities, as the availability of specific chiral precursors can be inconsistent, leading to extended lead times and unpredictable pricing structures for downstream ligand manufacturers.
The Novel Approach
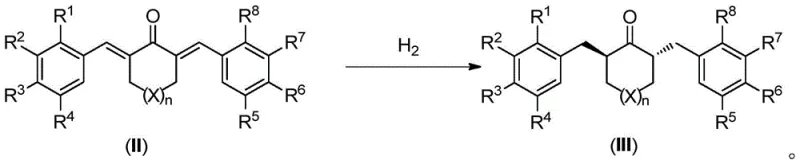
The methodology disclosed in the patent circumvents these historical limitations by employing a direct asymmetric synthesis strategy. By utilizing achiral starting materials—specifically alpha,alpha'-di(arylene) ketone compounds derived from simple aldol condensations—the process establishes chirality through catalytic asymmetric hydrogenation rather than separation. This is followed by a stereospecific ring-closing step that preserves the established stereochemistry. The elimination of resolution steps not only theoretically doubles the maximum yield compared to racemic resolution but also drastically simplifies the purification workflow. This approach enables the production of diverse spiroindane derivatives with various substituents (such as bromo, methoxy, or alkyl groups) without the need for custom-synthesized chiral building blocks, thereby offering a versatile platform for cost reduction in chiral ligand manufacturing.
Mechanistic Insights into TiCl4-Catalyzed Cyclization
The core innovation of this synthesis lies in the final ring-closing transformation, which converts the acyclic chiral ketone intermediates into the rigid spirocyclic framework. This intramolecular Friedel-Crafts reaction is mediated by strong Lewis acids, with Titanium Tetrachloride (TiCl4) identified as the optimal catalyst. The mechanism involves the activation of the carbonyl group or the generation of a carbocationic species that facilitates electrophilic aromatic substitution on the pendant aryl rings. Crucially, the reaction conditions are meticulously controlled, typically initiating at 0°C and slowly warming to room temperature, to prevent racemization of the sensitive chiral centers formed during the preceding hydrogenation step. Experimental data indicates that while Bronsted acids like polyphosphoric acid can effect cyclization, they often lead to racemic products due to harsh acidic conditions. In contrast, the Lewis acid protocol maintains the integrity of the chiral information, delivering products with ee values consistently above 99%.
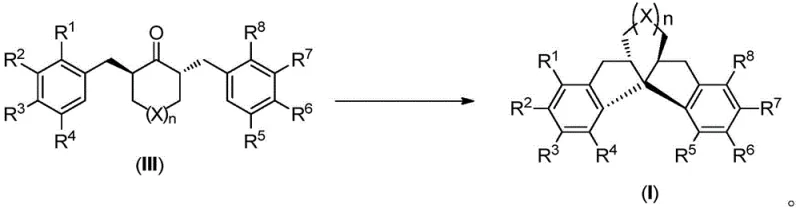
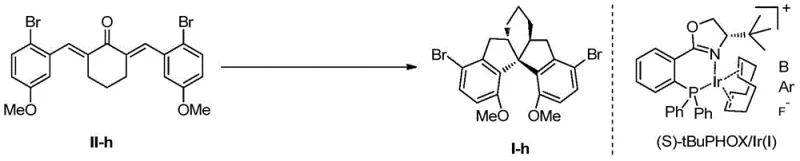
Furthermore, the precursor to this cyclization is generated via highly enantioselective hydrogenation using chiral iridium complexes, such as Ir/(S)-tBu-PHOX. This two-stage catalytic cascade ensures that the stereochemical outcome is dictated by the catalyst rather than thermodynamic equilibrium. The compatibility of this system with a wide range of electronic and steric environments on the aromatic rings demonstrates its robustness. Whether the substrate contains electron-donating methoxy groups or electron-withdrawing bromine atoms, the catalytic system adapts to provide high diastereoselectivity (anti/syn ratios >19:1) and enantioselectivity. This mechanistic reliability is essential for R&D teams focused on impurity control, as it minimizes the formation of diastereomeric by-products that are difficult to separate.
How to Synthesize Chiral Spiroindane Efficiently
The synthesis of these high-value chiral skeletons follows a logical progression designed for scalability and reproducibility. It begins with the construction of the carbon backbone through aldol condensation, followed by the critical installation of chirality via hydrogenation, and concludes with the cyclization event. The patent details specific molar ratios, solvent choices, and temperature profiles that are critical for success. For instance, maintaining anhydrous and oxygen-free conditions during the hydrogenation and cyclization steps is paramount to prevent catalyst deactivation and side reactions. The detailed standardized synthesis steps provided below outline the precise operational parameters required to replicate the high yields and optical purities reported in the patent examples, serving as a foundational guide for process chemists.
- Perform aldol condensation between substituted benzaldehydes and cyclic ketones to form alpha,alpha'-di(arylene) ketone precursors.
- Conduct asymmetric hydrogenation using chiral iridium/phosphine-oxazoline complexes to establish stereocenters with high enantiomeric excess.
- Execute intramolecular Friedel-Crafts cyclization using Titanium Tetrachloride (TiCl4) to close the spiro-ring system without racemization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from resolution-based synthesis to direct asymmetric catalysis offers profound strategic benefits beyond mere technical elegance. The primary advantage is the drastic simplification of the supply chain for raw materials. By relying on commodity chemicals like substituted benzaldehydes and cyclohexanone derivatives, manufacturers can mitigate the risks associated with sourcing specialized chiral intermediates. This transition significantly enhances supply chain reliability, as the availability of achiral precursors is generally stable and less susceptible to market fluctuations compared to niche chiral pools. Moreover, the streamlined process flow reduces the number of unit operations, which directly correlates to lower capital expenditure and reduced operational complexity in a manufacturing setting.
- Cost Reduction in Manufacturing: The elimination of chiral resolution steps removes the need for expensive resolving agents and the associated loss of 50% of the material mass. Additionally, the high yields reported in the hydrogenation (>90%) and cyclization (>80%) steps maximize the conversion of raw materials into finished product. The ability to run these reactions in common solvents like dichloromethane without requiring exotic reagents further drives down the cost of goods sold. Qualitatively, this represents a substantial cost saving opportunity by transforming a multi-step, low-efficiency process into a high-yielding catalytic sequence.
- Enhanced Supply Chain Reliability: Dependence on a single source for chiral starting materials is a significant vulnerability in pharmaceutical supply chains. This novel method diversifies the supply base by allowing the use of widely available achiral building blocks. The robustness of the TiCl4 catalyzed cyclization ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed resolution attempts or inconsistent optical purity. This reliability is crucial for maintaining continuous production schedules and meeting the stringent delivery timelines demanded by global pharmaceutical clients.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reaction conditions that translate well from laboratory to pilot and commercial scales. The one-pot variation, where hydrogenation and cyclization are telescoped, further reduces solvent consumption and waste generation. By avoiding the heavy use of chiral auxiliaries and resolution reagents, the environmental footprint of the manufacturing process is significantly reduced. This aligns with modern green chemistry principles and simplifies regulatory compliance regarding waste disposal, making it an attractive option for environmentally conscious manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation. They are intended to clarify the operational feasibility and strategic value of adopting this method for the production of chiral spiroindane intermediates. Understanding these nuances is vital for stakeholders evaluating the integration of this technology into their existing manufacturing portfolios.
Q: Why is this synthesis method superior to traditional resolution?
A: Traditional methods rely on resolving racemic mixtures, which theoretically limits yield to 50% and requires expensive chiral reagents. This patented route uses asymmetric catalysis to achieve >99% ee directly from achiral starting materials, significantly improving atom economy and reducing waste.
Q: What catalysts are used for the cyclization step?
A: The process utilizes Lewis acids, specifically Titanium Tetrachloride (TiCl4), which has been optimized to promote ring closure at low temperatures (0°C to 25°C) while maintaining the optical purity established in the previous hydrogenation step.
Q: Is this process scalable for industrial production?
A: Yes, the methodology employs robust reaction conditions and commercially available solvents like dichloromethane. The one-pot variation further simplifies processing by eliminating intermediate isolation, making it highly suitable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Spiroindane Supplier
The technical potential of this asymmetric synthesis route is immense, offering a pathway to high-purity chiral ligands that are essential for modern drug discovery and development. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative chemistry to the market. Our facility is equipped with rigorous QC labs and stringent purity specifications to ensure that every batch of chiral spiroindane skeleton compound meets the exacting standards of the global pharmaceutical industry. We understand the critical nature of chiral intermediates and are committed to delivering products with consistent optical purity and chemical quality.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain that prioritizes efficiency, quality, and reliability, ensuring that your development timelines are met without compromise.
