Scalable Synthesis of Functionalized BODIPY Dyes for Advanced Bio-Analytical Applications
Introduction to Next-Generation Fluorescent Probes
The landscape of analytical chemistry and life sciences is being rapidly transformed by the advent of advanced fluorophores capable of high-resolution imaging and precise quantification. Patent CN101205416A introduces a groundbreaking methodology for the synthesis of pyrromethine fluorescent dyes, specifically focusing on the BODIPY (boron-dipyrromethene) class, which are renowned for their exceptional photophysical properties. This technology addresses a critical gap in the market by integrating active phenolic hydroxyl groups directly into the dye scaffold during the synthesis process, rather than relying on cumbersome post-synthetic modifications. For R&D directors and procurement specialists seeking a reliable electronic chemical supplier or bio-reagent partner, this patent outlines a robust pathway to producing high-performance materials that exhibit narrow spectral half-widths and superior photostability. The ability to synthesize these complex molecules under mild conditions represents a significant leap forward in cost reduction in electronic chemical manufacturing and bio-probe production, offering a streamlined route that minimizes waste and maximizes yield consistency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for functionalized BODIPY dyes often suffer from significant inefficiencies that hinder large-scale commercial adoption. Conventional methods typically involve the construction of the BODIPY core first, followed by electrophilic aromatic substitution or cross-coupling reactions to introduce functional handles. These post-synthetic modifications frequently require harsh reaction conditions, expensive transition metal catalysts, and rigorous purification steps to remove trace metal impurities that can quench fluorescence or interfere with biological assays. Furthermore, the introduction of sensitive groups like phenols at a later stage often leads to low overall yields due to side reactions and the instability of intermediates under aggressive conditions. This multi-step complexity not only inflates the cost of goods sold but also extends the reducing lead time for high-purity fluorescent dyes, creating bottlenecks in the supply chain for downstream applications in diagnostics and imaging.
The Novel Approach
In stark contrast, the methodology disclosed in CN101205416A employs a convergent strategy that incorporates the active functionality at the precursor stage. By utilizing p-hydroxybenzaldehyde as a direct building block in the condensation step, the phenolic hydroxyl group is seamlessly integrated into the final molecular architecture. This approach leverages a mild oxidative condensation using chloranil at room temperature, which preserves the integrity of the sensitive hydroxyl moiety while efficiently forming the conjugated pyrromethene system. The subsequent complexation with boron trifluoride is conducted under inert atmosphere to ensure high purity without degrading the active site. This streamlined process eliminates the need for late-stage functionalization, thereby simplifying the workflow and enhancing the overall atom economy. 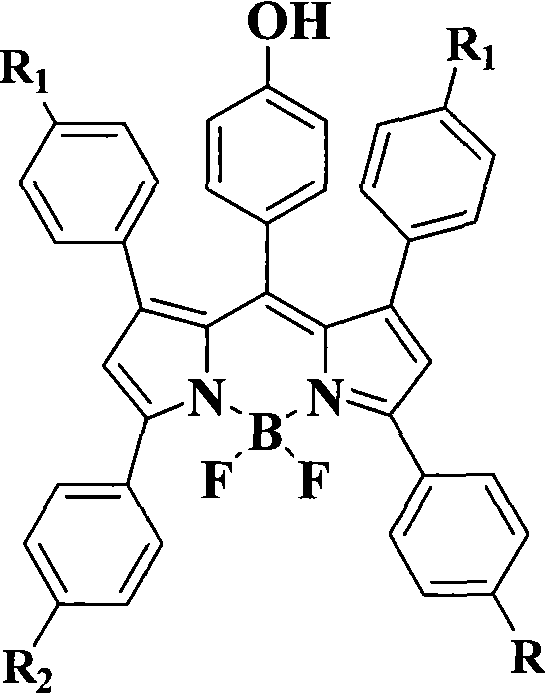 This structural versatility allows for the creation of a diverse library of dyes tailored for specific spectral requirements, making it an ideal solution for commercial scale-up of complex optoelectronic materials.
This structural versatility allows for the creation of a diverse library of dyes tailored for specific spectral requirements, making it an ideal solution for commercial scale-up of complex optoelectronic materials.
Mechanistic Insights into Chloranil-Mediated Oxidative Condensation
The core of this synthetic innovation lies in the oxidative condensation step, where 2,4-diphenylpyrrole reacts with p-hydroxybenzaldehyde in the presence of chloranil (tetrachloro-1,4-benzoquinone). Mechanistically, chloranil acts as a mild yet effective oxidant that facilitates the removal of hydrogen atoms to establish the extended conjugated system required for fluorescence. The patent specifies a weight ratio of pyrrole to chloranil between 1:3 and 1:7, indicating that a significant excess of oxidant is employed to drive the reaction to completion and suppress the formation of reduced byproducts. Trifluoroacetic acid (TFA) serves as a catalyst, protonating the aldehyde to increase its electrophilicity, thereby accelerating the nucleophilic attack by the pyrrole ring. This acid-catalyzed condensation proceeds rapidly at room temperature, generating a deep blue pyrromethene intermediate. The mildness of this protocol is crucial for maintaining the stability of the phenolic group, which might otherwise undergo oxidation or polymerization under more vigorous conditions typical of traditional BODIPY synthesis.
Following the formation of the pyrromethene backbone, the final step involves the coordination of the nitrogen atoms with a boron center using boron trifluoride etherate. This step rigidifies the molecular structure, locking the conformation and preventing non-radiative decay pathways, which is essential for achieving the high quantum yields characteristic of BODIPY dyes. The reaction is performed under inert gas protection (nitrogen or argon) to prevent moisture interference, which could hydrolyze the boron complex. Triethylamine is added as a base to neutralize the HF generated during complexation, ensuring the reaction mixture remains stable. The resulting product is a robust, highly fluorescent dye with excellent chemical stability. The inclusion of halogen substituents (Cl or Br) on the peripheral phenyl rings, as shown in the specific embodiments, further modulates the electronic properties, allowing for fine-tuning of the absorption and emission maxima to match specific laser lines or detector filters used in analytical instrumentation.
How to Synthesize Functionalized BODIPY Dyes Efficiently
The synthesis protocol detailed in the patent provides a clear, reproducible roadmap for manufacturing these high-value fluorophores. The process begins with the preparation of 2-phenylaziridine from styrene, followed by ring-opening and cyclization with acetophenone to form the pyrrole core. The critical oxidative condensation and boron complexation steps follow, yielding the final dye after standard workup procedures including extraction and column chromatography. This sequence is designed to be operationally simple, avoiding the need for specialized high-pressure equipment or cryogenic conditions. For technical teams looking to implement this chemistry, the detailed standardized synthesis steps are outlined below to ensure consistent batch-to-batch quality and optimal yield.
- Synthesize 2-phenylaziridine from styrene via bromination and azide substitution followed by thermal cyclization.
- React 2-phenylaziridine with acetophenone derivatives in the presence of sodium hydride to form 2,4-diphenylpyrrole intermediates.
- Condense the pyrrole intermediate with p-hydroxybenzaldehyde using chloranil as an oxidant and trifluoroacetic acid as a catalyst at room temperature.
- Complex the resulting pyrromethene with boron trifluoride etherate under inert gas protection to finalize the BODIPY core structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the synthetic route described in CN101205416A offers compelling advantages for supply chain optimization and cost management. The reliance on commodity chemicals such as styrene, acetophenone, and p-hydroxybenzaldehyde ensures a stable and cost-effective raw material supply base, mitigating the risks associated with sourcing exotic or proprietary intermediates. The ability to perform key reaction steps at room temperature significantly reduces energy consumption compared to processes requiring prolonged heating or cooling, directly contributing to lower utility costs and a smaller carbon footprint. Furthermore, the use of chloranil as an oxidant, while requiring careful handling, avoids the generation of heavy metal waste streams associated with transition-metal catalyzed couplings, simplifying wastewater treatment and environmental compliance protocols. 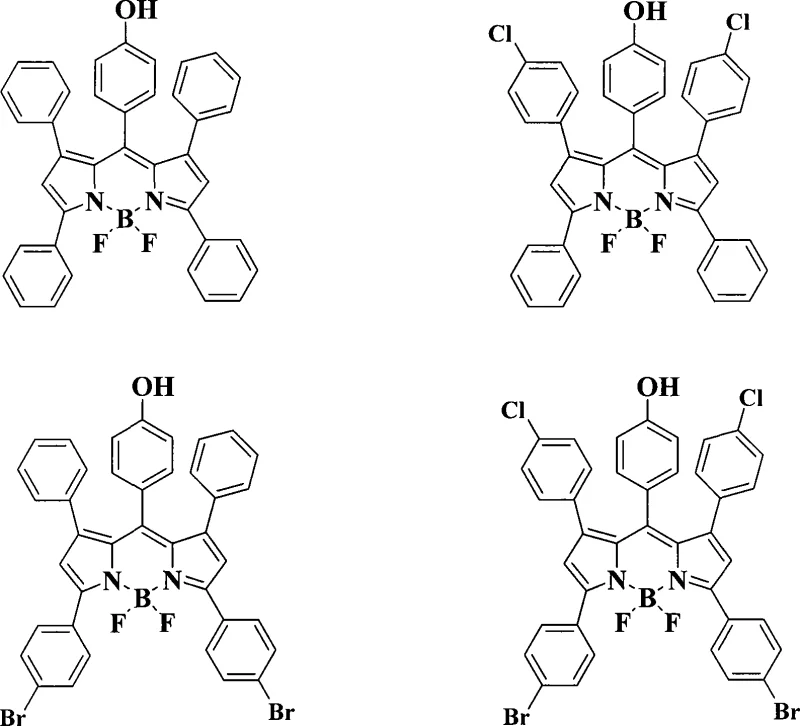 These factors collectively enhance the scalability and environmental compliance of the manufacturing process, making it highly attractive for large-volume production.
These factors collectively enhance the scalability and environmental compliance of the manufacturing process, making it highly attractive for large-volume production.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction of energy-intensive heating steps lead to substantial operational savings. The direct incorporation of the functional group avoids the yield losses typically associated with multi-step post-modification strategies, effectively increasing the overall process efficiency. By streamlining the synthesis into fewer, high-yielding steps, manufacturers can achieve a lower cost per gram of the final active pharmaceutical ingredient or electronic material, providing a competitive edge in pricing negotiations.
- Enhanced Supply Chain Reliability: The use of widely available starting materials ensures that production is not vulnerable to the supply disruptions often seen with specialized reagents. The robustness of the room-temperature reactions means that the process is less sensitive to minor fluctuations in utility supply, ensuring consistent output even in varying manufacturing environments. This reliability is critical for maintaining continuous supply to downstream customers in the pharmaceutical and diagnostic sectors, where batch consistency is paramount.
- Scalability and Environmental Compliance: The process is inherently scalable, as the exothermic nature of the reactions can be easily managed with standard cooling systems, and the workup involves conventional liquid-liquid extraction and crystallization techniques familiar to plant operators. The absence of heavy metals simplifies the purification process and reduces the burden on waste treatment facilities, aligning with increasingly stringent global environmental regulations. This ease of scale-up facilitates the rapid transition from laboratory benchtop quantities to multi-ton annual production capacities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel fluorescent dyes. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners and clients. Understanding these details is essential for evaluating the feasibility of integrating these materials into your existing product lines or research workflows.
Q: What is the primary advantage of the phenolic hydroxyl group in these BODIPY dyes?
A: The phenolic hydroxyl group serves as a critical active functional handle, allowing for facile bioconjugation with biomolecules such as proteins and antibodies, thereby transforming the dye into a specific fluorescent probe for life science applications.
Q: How does the chloranil oxidation method improve process safety compared to traditional methods?
A: The use of chloranil allows the oxidative condensation step to proceed efficiently at room temperature, eliminating the need for high-temperature reflux or hazardous strong oxidants, which significantly reduces energy consumption and operational risks during commercial scale-up.
Q: Can the optical properties of these dyes be tuned for specific wavelengths?
A: Yes, by substituting the starting styrene or acetophenone with halogenated derivatives such as p-chlorostyrene or p-bromoacetophenone, the conjugation system is altered, enabling precise tuning of excitation and emission wavelengths across the visible to near-infrared spectrum.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable BODIPY Dyes Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced fluorescent materials in driving innovation across the life sciences and electronics sectors. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to market is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of BODIPY dye meets the highest standards of performance and consistency required for sensitive analytical applications. We are committed to delivering high-purity OLED material and bio-probes that empower your research and product development.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your specific needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized processes can reduce your total cost of ownership. Please contact us today to request specific COA data and route feasibility assessments, and let us help you accelerate your time-to-market with superior fluorescent solutions.
