Advanced Synthesis of Biotin-Labeled Matrine Probes for Target Identification
The pharmaceutical industry's relentless pursuit of understanding drug mechanisms has elevated the importance of chemical proteomics, where small molecule probes play a pivotal role. Patent CN113754686A introduces a sophisticated and robust methodology for the synthesis of a biotin-labeled matrine probe, a critical tool for identifying the direct protein targets of matrine, a well-known alkaloid with significant therapeutic potential. This patent addresses the urgent technical challenge of constructing high-affinity probes that can withstand the rigorous conditions of affinity purification and mass spectrometry analysis. By detailing a multi-step synthetic route that integrates ring-opening chemistry, protective group strategies, and advanced purification techniques, the invention provides a reliable blueprint for producing research-grade intermediates. For R&D directors and procurement specialists alike, this technology represents a significant advancement in the availability of high-purity chemical biology tools, ensuring that downstream biological assays are conducted with reagents of defined structure and exceptional quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of alkaloid-based probes has been fraught with challenges related to regioselectivity and purification. Conventional methods often struggle with the inherent stability of the matrine skeleton, particularly the lactam ring, which is resistant to mild functionalization attempts. Without a controlled ring-opening strategy, introducing a linker for biotinylation often leads to complex mixtures of isomers or degradation products that are difficult to separate. Furthermore, standard purification techniques such as silica gel chromatography are frequently inadequate for highly polar biotin conjugates, resulting in final products with insufficient purity for sensitive proteomic applications. The lack of specific protective group strategies in older routes often leads to side reactions, such as the double coupling of diamine linkers, which drastically reduces yield and complicates the isolation of the desired mono-substituted probe.
The Novel Approach
The approach disclosed in patent CN113754686A overcomes these hurdles through a convergent synthesis strategy that meticulously controls reactivity at each stage. By initially hydrolyzing the lactam ring of matrine under alkaline conditions and immediately protecting the newly formed amine with a Boc group, the process creates a stable carboxylic acid handle for subsequent coupling. This strategic use of Boc protection not only stabilizes the intermediate but also prevents unwanted side reactions during the linker attachment phase. The synthesis further employs a pre-functionalized biotin linker, prepared separately to ensure high purity before the final coupling step. This modular approach allows for the optimization of each fragment independently, significantly enhancing the overall efficiency and reproducibility of the probe synthesis compared to linear, one-pot methods.
Mechanistic Insights into Alkaloid Ring-Opening and Amidation
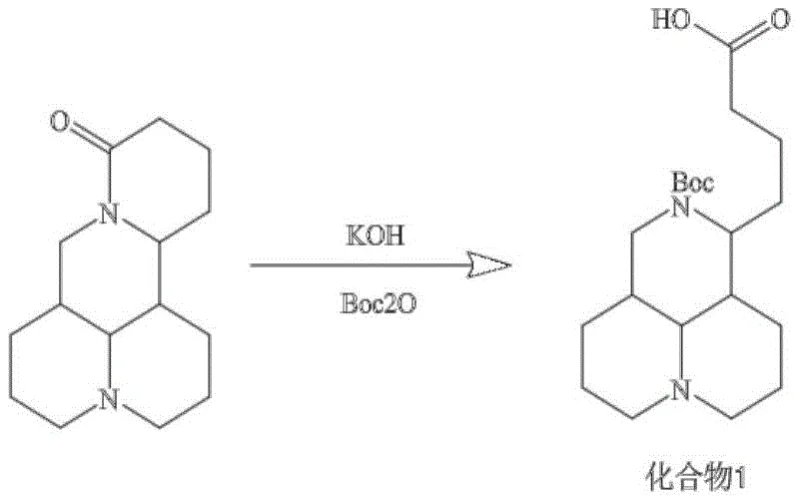
The core chemical transformation in this synthesis is the base-mediated hydrolysis of the matrine lactam ring, followed by in-situ protection. Under reflux conditions with potassium hydroxide at 105°C, the cyclic amide bond is cleaved to generate a carboxylate and a secondary amine. The immediate addition of di-tert-butyl dicarbonate (Boc2O) ensures that the nucleophilic amine is capped as a carbamate, preventing it from interfering with the carboxylic acid activation in later steps. This sequence is critical because free amines could otherwise compete with the intended coupling partners or lead to intramolecular cyclization. The resulting Compound 1 possesses a free carboxylic acid group that serves as the anchor point for the biotin linker, effectively converting the rigid alkaloid scaffold into a flexible probe precursor capable of interacting with target proteins.
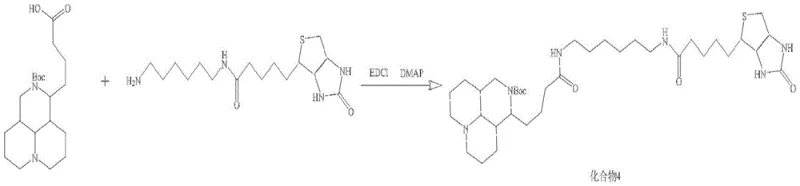
Impurity control is rigorously managed through the selection of coupling reagents and purification media. The use of EDCI (1-ethyl-3-(3-dimethylaminopropyl)carbodiimide) and DMAP (4-dimethylaminopyridine) facilitates efficient amide bond formation between the matrine derivative and the biotin linker while minimizing racemization, although stereochemistry is less of a concern for the linker itself. A major mechanistic advantage is the prevention of linker polymerization; by using 1-amino-6-tert-butoxyaminohexane, the synthesis ensures that only one end of the diamine reacts with biotin initially. The final deprotection step using trifluoroacetic acid (TFA) not only removes the Boc groups to reveal the active amine but also forms a stable TFA salt with the probe, which enhances its stability and solubility. The patent emphasizes the necessity of reverse-phase HPLC purification to remove urea byproducts from the EDCI coupling and any unreacted starting materials, ensuring the final probe meets the stringent purity requirements for mass spectrometry.
How to Synthesize Biotin-Labeled Matrine Probe Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step guide for producing the target probe with high fidelity. The process begins with the preparation of the ring-opened matrine derivative, followed by the independent synthesis of the biotin-linker fragment. These two key intermediates are then coupled under mild conditions to form the protected probe, which is finally deprotected and purified. This modular workflow allows for quality control checks at the intermediate stage, reducing the risk of losing valuable materials in the final steps. For detailed operational parameters, including specific solvent ratios, reaction times, and purification gradients, please refer to the standardized synthesis guide below.
- Hydrolyze matrine with KOH and protect the resulting amine with Boc2O to form Compound 1.
- Couple biotin with Boc-protected hexamethylenediamine using EDCI/DMAP to form Compound 2, then deprotect to get Compound 3.
- Condense Compound 1 and Compound 3 using EDCI/DMAP to form Compound 4, followed by final TFA deprotection and HPLC purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the methodology described in CN113754686A offers substantial advantages in terms of raw material availability and process robustness. The reagents employed, such as potassium hydroxide, Boc anhydride, EDCI, and TFA, are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. This broad availability ensures that production schedules can be maintained without interruption due to reagent shortages. Furthermore, the reaction conditions utilize standard laboratory equipment, such as reflux setups and rotary evaporators, meaning that the process can be transferred to pilot plants or manufacturing facilities without requiring specialized high-pressure or cryogenic infrastructure. This ease of technology transfer significantly reduces the capital expenditure required for scaling up production.
- Cost Reduction in Manufacturing: The synthetic route is designed to maximize atom economy and minimize waste through high-yielding steps, particularly the formation of the biotin-linker intermediate which proceeds with nearly quantitative yield. By avoiding the use of expensive transition metal catalysts or exotic chiral ligands, the process eliminates the need for costly metal scavenging steps and complex waste treatment protocols associated with heavy metals. The strategic use of Boc protection prevents the formation of difficult-to-separate side products, thereby reducing the loss of material during purification and lowering the overall cost per gram of the final probe. Additionally, the ability to purify intermediates before the final coupling step prevents the compounding of impurities, which often leads to low yields in the final stage of traditional syntheses.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents and solvents like DMF and dichloromethane ensures that the supply chain is resilient to fluctuations in the availability of sensitive or hazardous materials. The process does not require inert atmosphere conditions for all steps, simplifying the operational requirements and allowing for more flexible manufacturing scheduling. The high purity achieved through the specified HPLC purification method means that the final product has a longer shelf life and reduced risk of degradation during storage and transport. This reliability is crucial for pharmaceutical companies that depend on consistent reagent quality for long-term research projects and regulatory filings.
- Scalability and Environmental Compliance: The synthesis avoids the generation of highly toxic byproducts, and the solvents used are amenable to standard recovery and recycling processes, aligning with modern green chemistry principles. The use of aqueous workups and standard organic extractions simplifies the waste management profile compared to processes requiring complex chromatographic separations on a large scale. The final purification step, while utilizing HPLC, is performed on the final product only, minimizing the volume of solvent waste generated compared to purifying every intermediate by preparative HPLC. This streamlined approach facilitates easier regulatory compliance regarding solvent residues and environmental discharge, making the process suitable for cGMP manufacturing environments.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the synthesis and application of the biotin-labeled matrine probe, based on the specific embodiments and data provided in the patent documentation. These answers are derived directly from the experimental examples and beneficial effects described in the text, ensuring accuracy for technical decision-makers.
Q: Why is HPLC reverse-phase purification critical for this probe?
A: The final biotin-labeled matrine probe exhibits high polarity, making conventional purification methods like recrystallization or normal phase chromatography ineffective. The patent specifies using reverse-phase HPLC with an acetonitrile/water/TFA system to achieve high purity (over 96% by LCMS), which is essential for accurate chemical proteomics results.
Q: What is the role of Boc protection in the synthesis?
A: Boc (tert-butyloxycarbonyl) protection serves two critical functions: it protects the amine generated from the ring-opening of matrine during subsequent steps, and it prevents the double coupling of the hexamethylenediamine linker with biotin, ensuring the formation of the correct mono-substituted intermediate.
Q: Can this synthesis be scaled for commercial supply?
A: Yes, the synthesis utilizes standard reagents such as EDCI, DMAP, and TFA, and employs common solvents like DMF and dichloromethane. The process avoids exotic catalysts, making it highly amenable to scale-up from gram to kilogram quantities for industrial supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biotin-Labeled Matrine Probe Supplier
The synthesis of complex chemical biology tools like the biotin-labeled matrine probe requires a partner with deep expertise in organic synthesis and process optimization. NINGBO INNO PHARMCHEM stands ready to support your research needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art purification capabilities, including preparative HPLC systems identical to those required for this high-polarity probe, ensuring that we can meet your stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques such as LC-MS and NMR to verify the structural integrity and purity of every batch, guaranteeing that the probes you receive are fit for purpose in sensitive target identification assays.
We invite you to contact our technical procurement team to discuss your specific requirements for this probe or related alkaloid derivatives. By partnering with us, you gain access to a Customized Cost-Saving Analysis that can identify opportunities to optimize your supply chain further. We encourage you to request specific COA data and route feasibility assessments to see how our manufacturing capabilities can accelerate your drug discovery timeline. Let us be your trusted partner in transforming complex patent methodologies into reliable, commercial-grade reality.
