Advanced Synthesis of Biotin-Labeled Matrine Probes for Commercial Scale-Up and High-Purity Research Applications
Advanced Synthesis of Biotin-Labeled Matrine Probes for Commercial Scale-Up and High-Purity Research Applications
The rapid advancement of chemical proteomics has necessitated the development of highly specific molecular tools capable of identifying drug targets with precision. Patent CN113754686B introduces a groundbreaking synthetic methodology for producing a biotin-labeled matrine probe, a critical reagent for mapping the interactome of matrine-derived alkaloids. This technology addresses the longstanding challenge of synthesizing polar, complex alkaloid derivatives that maintain biological activity while incorporating affinity tags. For R&D directors and procurement specialists, this patent represents a significant leap forward in reliable chemical proteomics probe supplier capabilities, offering a route that balances structural complexity with manufacturability. The method leverages strategic protective group chemistry and advanced purification techniques to ensure that the final product meets the rigorous standards required for mass spectrometry-based target identification.
Furthermore, the integration of this synthesis into a commercial supply chain offers substantial benefits for organizations seeking cost reduction in pharmaceutical intermediate manufacturing. By optimizing the reaction sequence to minimize side products and utilizing scalable coupling reagents, the process reduces the burden on downstream purification units. This efficiency translates directly into improved supply chain reliability and reduced lead times for high-purity research probes. As the demand for targeted therapy discovery tools grows, the ability to source such specialized intermediates from a partner with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production becomes a strategic asset. This report analyzes the technical merits and commercial implications of this patented technology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing biotinylated alkaloid probes often suffer from significant drawbacks related to regioselectivity and purification efficiency. Conventional methods frequently struggle with the direct conjugation of biotin to complex alkaloid scaffolds like matrine due to the presence of multiple nucleophilic sites, leading to heterogeneous mixtures that are difficult to separate. Without precise control over the reaction environment, researchers often encounter low yields and the formation of polymeric byproducts, which severely compromise the utility of the probe in affinity purification experiments. Additionally, the high polarity of the final conjugated products typically renders standard silica gel chromatography ineffective, forcing reliance on labor-intensive preparative TLC or low-resolution column chromatography. These limitations not only increase the cost of goods but also extend the timeline for project milestones, creating bottlenecks in the drug discovery pipeline.
The Novel Approach
The methodology disclosed in patent CN113754686B overcomes these hurdles through a meticulously designed multi-step sequence that prioritizes chemoselectivity and ease of isolation. By employing a ring-opening strategy on the matrine lactam moiety followed by Boc-protection, the synthesis creates a unique carboxylic acid handle that is orthogonal to the existing amine functionalities. This allows for the subsequent attachment of a pre-functionalized biotin linker without risking non-specific labeling. The use of HPLC reversed-phase purification at critical intermediate stages ensures that impurities are removed before they can propagate through the synthesis, resulting in a final product of exceptional purity. This novel approach not only enhances the quality of the chemical probe but also streamlines the manufacturing process, making it a viable candidate for commercial scale-up of complex alkaloid derivatives.
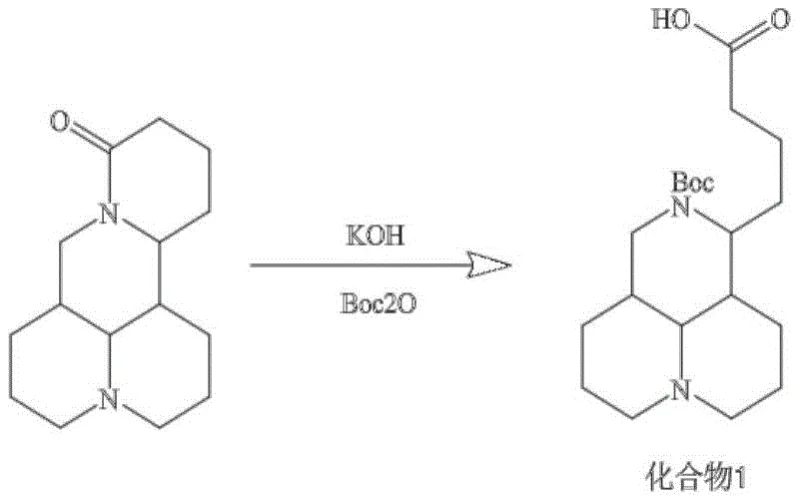
Mechanistic Insights into Amide Coupling and Protective Group Strategy
The core of this synthesis lies in the strategic manipulation of functional groups to achieve the desired connectivity while maintaining the integrity of the sensitive alkaloid core. The initial step involves the hydrolysis of the matrine lactam ring under alkaline conditions using potassium hydroxide, which generates a free carboxylic acid and a secondary amine. Immediate protection of this amine with di-tert-butyl dicarbonate (Boc2O) is crucial, as it prevents self-condensation and ensures that the subsequent coupling reaction occurs exclusively at the carboxyl terminus. This level of control is essential for producing a homogeneous probe, as any variation in the labeling site could alter the binding affinity of the matrine moiety to its protein targets. The reaction conditions are optimized to balance reaction rate with selectivity, ensuring high conversion without degrading the sensitive polycyclic framework.
Subsequent coupling steps utilize standard peptide synthesis reagents such as EDCI and DMAP to forge the amide bonds between the matrine derivative and the biotin linker. The choice of a Boc-protected hexamethylenediamine linker is particularly ingenious, as it serves as a temporary mask for one of the amine groups, preventing the formation of bis-biotinylated species. After the initial coupling, the Boc group is removed using trifluoroacetic acid (TFA), revealing the primary amine necessary for the final conjugation. This stepwise assembly allows for the purification of intermediates, which is a key advantage over one-pot strategies. The final deprotection and salt formation with TFA further enhance the stability and solubility of the probe, facilitating its handling in aqueous biological buffers.
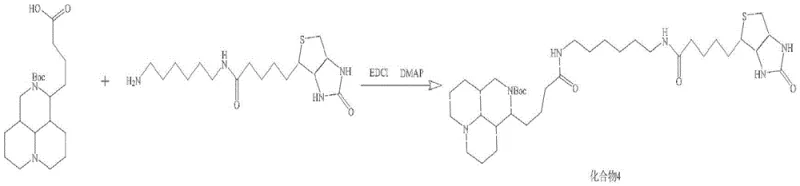
How to Synthesize Biotin-Labeled Matrine Probe Efficiently
The synthesis of this high-value probe requires strict adherence to the patented protocol to ensure reproducibility and quality. The process begins with the preparation of the key matrine intermediate, followed by the independent synthesis of the biotin-linker construct. These two fragments are then converged through a carbodiimide-mediated coupling reaction, which demands precise control over stoichiometry and temperature to minimize racemization or side reactions. The final steps involve global deprotection and rigorous purification using preparative HPLC to remove any remaining reagents or byproducts. Detailed standardized synthesis steps see the guide below.
- Hydrolyze matrine with potassium hydroxide and protect the resulting amine with Boc2O to form Compound 1.
- Couple biotin with Boc-protected hexamethylenediamine using EDCI and DMAP to generate Compound 2, followed by TFA deprotection to yield Compound 3.
- Condense Compound 1 and Compound 3 using peptide coupling reagents, followed by final deprotection and HPLC purification to isolate the target probe.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers compelling advantages for procurement managers and supply chain heads looking to optimize their sourcing strategies for research chemicals. The process eliminates the need for expensive transition metal catalysts often used in C-H activation or cross-coupling reactions, relying instead on readily available organic reagents that are cost-effective and easy to source globally. This simplification of the bill of materials reduces the risk of supply disruptions caused by shortages of specialized catalysts. Furthermore, the high purity achieved through the described HPLC purification steps means that end-users spend less time and resources on quality control testing, accelerating the time-to-market for downstream drug discovery programs.
- Cost Reduction in Manufacturing: The synthetic pathway is designed to maximize atom economy and minimize waste generation, which directly correlates to lower production costs. By avoiding complex catalytic cycles and utilizing robust coupling chemistry, the process reduces the consumption of high-value reagents. The ability to purify intermediates effectively prevents the loss of valuable materials in later stages, ensuring that the overall yield remains commercially viable. This efficiency allows suppliers to offer competitive pricing without compromising on the quality or purity of the final biotin-labeled matrine probe.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as DMF, dichloromethane, and common coupling agents ensures that the supply chain is resilient against market volatility. Unlike processes dependent on rare earth metals or proprietary ligands, this route can be executed in multiple manufacturing facilities worldwide, providing redundancy and security of supply. For procurement teams, this means reduced lead times and greater flexibility in ordering schedules, ensuring that critical research projects are never delayed due to material shortages.
- Scalability and Environmental Compliance: The reaction conditions are mild and operate at temperatures and pressures that are easily manageable in standard stainless steel reactors, facilitating seamless scale-up from gram to kilogram quantities. The waste streams generated are primarily organic solvents and salts, which can be treated using standard industrial waste management protocols. This alignment with environmental compliance standards reduces the regulatory burden on manufacturers and supports sustainable chemistry initiatives, making the product attractive to environmentally conscious organizations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this biotin-labeled matrine probe. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation, providing clarity on the process capabilities and product performance. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this probe into their research workflows.
Q: Why is HPLC reversed-phase purification critical for this probe?
A: The final biotin-labeled matrine probe exhibits high polarity, making conventional purification methods like recrystallization or normal-phase chromatography ineffective. The patent specifies using an HPLC reversed-phase preparative column to ensure the removal of polar impurities and unreacted starting materials, thereby achieving the stringent purity specifications required for sensitive chemical proteomics assays.
Q: How does the linker strategy prevent side reactions?
A: The synthesis utilizes 1-amino-6-tert-butoxyaminohexane where one amino group is protected by a Boc group. This selective protection prevents the diamine from reacting with biotin at both ends, which would create polymeric byproducts. By ensuring mono-substitution, the process significantly simplifies downstream purification and improves the overall yield of the functional probe.
Q: What are the scalability advantages of this synthetic route?
A: The route relies on robust, scalable reactions such as amide coupling and acid-base hydrolysis which are well-suited for kilogram-scale production. The use of common solvents like DMF and dichloromethane, combined with standard workup procedures like liquid-liquid extraction, ensures that the process can be transferred from laboratory benchtop to commercial manufacturing without requiring exotic equipment or hazardous conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biotin-Labeled Matrine Probe Supplier
At NINGBO INNO PHARMCHEM, we understand the critical role that high-quality chemical probes play in accelerating drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to replicate complex synthetic routes like the one described in CN113754686B demonstrates our technical prowess and dedication to supporting the pharmaceutical and biotechnology sectors.
We invite you to collaborate with us to leverage this advanced technology for your research needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project requirements. We are ready to provide specific COA data and route feasibility assessments to help you make informed decisions. By partnering with us, you gain access to a reliable supply chain and a wealth of technical expertise dedicated to your success.
