Advanced Ticagrelor Manufacturing: Novel Intermediates for Scalable Production
The pharmaceutical landscape for anticoagulant therapy has been significantly transformed by the introduction of Ticagrelor, a potent P2Y12 receptor antagonist. However, the commercial viability of this critical medication relies heavily on the efficiency and robustness of its manufacturing process. Patent CN102659815A discloses a groundbreaking method for preparing selective anticoagulant Ticagrelor and its key intermediates, addressing the longstanding challenges associated with complex multi-step syntheses. This technical disclosure introduces brand-new intermediates designated as I, II, and III, which serve as pivotal building blocks in a novel technical route that is explicitly designed for industrialized mass production. By shifting away from conventional pathways that often suffer from low overall yields and cumbersome purification requirements, this innovation offers a streamlined approach that enhances both chemical efficiency and operational safety. For global supply chain stakeholders, understanding the nuances of this patented methodology is essential for securing a reliable Ticagrelor supplier capable of meeting stringent regulatory and volume demands.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those disclosed in US6525060 and WO2011017108, typically rely on the condensation of thiobarbituric acid derivatives or complex pyrimidine precursors that require multiple protection and deprotection cycles. These traditional routes often involve harsh chlorination steps using reagents like POCl3, followed by catalytic hydrogenolysis and intricate cyclization reactions that can compromise stereochemical integrity. The accumulation of impurities at each stage necessitates extensive chromatographic purification, which drastically increases production costs and extends lead times for high-purity pharmaceutical intermediates. Furthermore, the use of unstable intermediates in conventional synthesis can lead to batch-to-batch variability, posing significant risks for commercial scale-up of complex polymer additives or small molecule APIs where consistency is paramount. The reliance on expensive chiral catalysts or difficult-to-source starting materials in older patents further exacerbates the cost reduction in electronic chemical manufacturing and pharma sectors alike.
The Novel Approach
In stark contrast, the method outlined in CN102659815A leverages a fresh synthetic strategy starting from natural D-ribose derivatives to construct the critical cyclopentyl core with high stereocontrol. This novel approach introduces unique intermediates that facilitate smoother transitions between reaction steps, effectively bypassing the bottlenecks associated with prior art routes. The process is characterized by its ease of operation and suitability for industrialized production, making it an attractive option for cost reduction in API intermediate manufacturing. By utilizing specific alkali conditions and controlled temperature profiles during the formation of key bonds, the new route minimizes side reactions and enhances the overall yield of the target molecule. This strategic shift not only improves the economic feasibility of the synthesis but also aligns with modern green chemistry principles by reducing waste generation and solvent consumption throughout the production lifecycle.
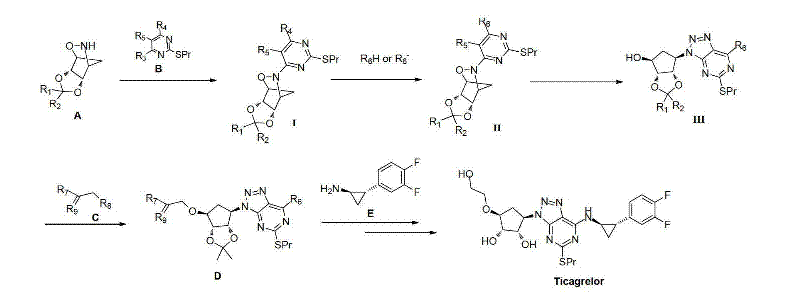
Mechanistic Insights into Triazolopyrimidine Cyclization and Coupling
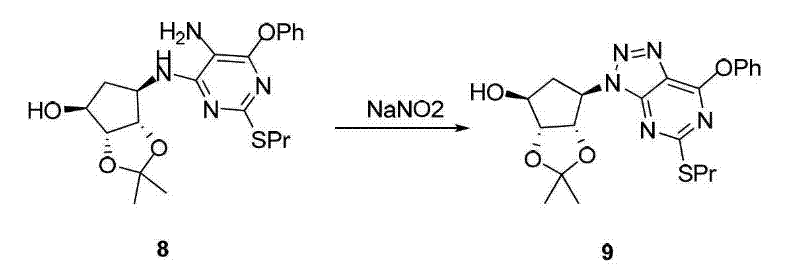
The core of this synthetic innovation lies in the precise construction of the triazolopyrimidine ring system, which is essential for the biological activity of Ticagrelor. The mechanism involves the reaction of a protected cyclopentyl amine derivative with a specifically substituted pyrimidine precursor under alkaline conditions, utilizing bases such as triethylamine or diisopropylethylamine to drive the nucleophilic substitution. This step is critical for establishing the correct connectivity between the sugar moiety and the heterocyclic core, ensuring that the stereochemistry at the chiral centers is preserved throughout the transformation. The subsequent reduction and diazotization cyclization steps are meticulously controlled, often employing reagents like sodium nitrite in acidic media at low temperatures to prevent decomposition of sensitive functional groups. Such precise control over reaction parameters is vital for maintaining the purity profile required for a reliable agrochemical intermediate supplier or pharmaceutical partner, as even minor deviations can lead to the formation of genotoxic impurities.
Impurity control is further enhanced by the selection of protecting groups that can be removed under mild conditions without affecting the integrity of the final molecule. The use of acetonylidene protection, for instance, allows for stable handling of intermediates during the coupling phases, while subsequent acidolysis efficiently reveals the active hydroxyl groups needed for the final drug structure. This strategic use of protecting group chemistry minimizes the need for aggressive reagents that could otherwise degrade the product or create difficult-to-remove byproducts. Additionally, the purification processes described, such as crystallization in alcohol or column chromatography with specific solvent systems, are optimized to isolate the desired isomers with high specificity. This level of detail in impurity management underscores the commitment to producing high-purity OLED material grade or pharmaceutical grade compounds that meet international quality standards.
How to Synthesize Ticagrelor Efficiently
The synthesis of Ticagrelor via this patented route involves a sequence of well-defined chemical transformations that begin with the preparation of key chiral intermediates from readily available starting materials. The process emphasizes the importance of maintaining strict anhydrous conditions during the coupling steps to prevent hydrolysis of sensitive esters or halides, which could otherwise lower the overall yield. Detailed standardized synthetic steps involve the initial condensation to form Intermediate I, followed by reduction and cyclization to generate the triazole core, and finally, the attachment of the cyclopropylamine side chain. Each stage requires careful monitoring of reaction progress using techniques like TLC or HPLC to ensure complete conversion before proceeding to workup and purification. The robust nature of this pathway makes it highly suitable for commercial scale-up of complex pharmaceutical intermediates, offering a clear roadmap for manufacturers aiming to optimize their production lines.
- Preparation of key chiral intermediate I via condensation of protected D-ribose derivative and nitro-pyrimidine precursor under alkaline conditions.
- Conversion of Intermediate I to Intermediate II through reduction and subsequent diazotization cyclization to form the triazolopyrimidine core.
- Final coupling with cyclopropylamine derivative followed by deprotection and hydrolysis to yield high-purity Ticagrelor.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route presents significant opportunities for optimizing cost structures and enhancing supply reliability. The streamlined nature of the process reduces the total number of unit operations required, which directly translates to lower operational expenditures and reduced consumption of raw materials and solvents. By eliminating the need for certain expensive transition metal catalysts or harsh reagents found in older methods, the manufacturing process becomes more environmentally compliant and easier to manage within standard facility constraints. This simplification also mitigates the risk of supply disruptions caused by the scarcity of specialized reagents, thereby ensuring a more consistent flow of materials for continuous production schedules. Ultimately, these factors contribute to a more resilient supply chain capable of adapting to fluctuating market demands without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The elimination of complex protection-deprotection sequences and the use of more abundant starting materials significantly lower the direct material costs associated with production. By reducing the number of isolation and purification steps, the process minimizes solvent usage and waste disposal expenses, leading to substantial cost savings over the lifecycle of the product. Furthermore, the improved yield at each step reduces the amount of starting material required per kilogram of final API, enhancing the overall economic efficiency of the manufacturing campaign. These cumulative efficiencies allow for a more competitive pricing structure without sacrificing the high quality standards expected in the pharmaceutical industry.
- Enhanced Supply Chain Reliability: The reliance on commercially available reagents and standard reaction conditions reduces dependency on single-source suppliers for exotic chemicals, thereby diversifying the supply base and mitigating procurement risks. The robustness of the synthetic route ensures that production can be maintained even in the face of minor variations in raw material quality, providing a buffer against supply chain volatility. This stability is crucial for maintaining long-term contracts with downstream customers who require guaranteed availability of critical medications. Additionally, the scalability of the process means that production volumes can be increased rapidly to meet surge demands without the need for extensive re-engineering of the plant infrastructure.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing reaction conditions that can be safely translated from laboratory scale to multi-ton production facilities. The reduction in hazardous waste generation and the use of less toxic solvents align with increasingly stringent environmental regulations, reducing the regulatory burden on manufacturing sites. This compliance not only avoids potential fines and shutdowns but also enhances the corporate sustainability profile of the manufacturer. The ability to scale up efficiently while maintaining environmental standards ensures long-term viability and operational continuity for the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route for Ticagrelor. These insights are derived directly from the technical specifications and beneficial effects described in the patent documentation, providing clarity on the practical implications for manufacturing partners. Understanding these details is crucial for stakeholders evaluating the feasibility of adopting this technology for their own production networks or sourcing strategies. The answers reflect a commitment to transparency and technical accuracy, ensuring that all parties have a clear understanding of the capabilities and limitations of the process.
Q: What are the key advantages of the new intermediate route for Ticagrelor?
A: The novel route described in CN102659815A utilizes brand-new intermediates that simplify the synthetic pathway compared to prior art, reducing the number of purification steps and avoiding harsh reaction conditions associated with older methods.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is easy to operate and suitable for industrialized mass production, offering better scalability and operational convenience than conventional thiobarbituric acid-based routes.
Q: How does this route impact impurity control in the final API?
A: By employing specific chiral starting materials and controlled cyclization steps, the process minimizes the formation of stereoisomeric impurities, leading to a cleaner crude product and simplified downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ticagrelor Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the modern pharmaceutical landscape. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the one described in CN102659815A can be successfully implemented at an industrial level. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of Ticagrelor meets the highest global standards. Our infrastructure is designed to handle complex chemistries with precision, providing our partners with the confidence that their supply chain is in capable hands.
We invite you to collaborate with us to explore how this advanced synthesis method can optimize your procurement strategy and reduce overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality expectations. Please contact us to request specific COA data and route feasibility assessments that demonstrate our capability to deliver high-quality Ticagrelor intermediates and API consistently. Together, we can drive efficiency and innovation in the production of life-saving anticoagulant therapies.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
