Advanced Manufacturing of High All-Trans Beta-Carotene via IBX Oxidation
The global demand for high-purity carotenoids, particularly beta-carotene, continues to surge across the nutraceutical, food coloring, and feed additive sectors. However, traditional synthetic routes have long been plagued by issues regarding product stability, low yields, and the use of hazardous oxidants. A groundbreaking technical solution detailed in patent CN113321604A introduces a novel preparation method that fundamentally shifts the paradigm of beta-carotene manufacturing. This innovative approach utilizes Vitamin A derivatives as starting materials, employing a sophisticated sequence involving C20 phosphonium salt formation, followed by a highly selective oxidative condensation mediated by IBX (o-iodoxybenzoic acid) compounds, and concluding with a transition metal-catalyzed isomerization. By replacing aggressive oxidants with environmentally benign high-iodine compounds and integrating a recyclable supported catalyst system, this technology not only achieves exceptional all-trans content but also drastically mitigates the peroxidation risks inherent in prior art. For industry leaders seeking a reliable beta-carotene supplier, understanding this mechanistic leap is crucial for securing a competitive edge in quality and cost efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of beta-carotene has relied heavily on oxidative coupling strategies that utilize harsh chemical environments, leading to significant operational and economic drawbacks. Traditional methods, such as those disclosed in earlier patents like CN101041631A and CN108047112A, frequently employ strong oxidizing agents like hypochlorite, nitrate, or peroxyacids under alkaline or acidic conditions. While these reagents are potent, their lack of selectivity is a critical failure point; they aggressively attack the delicate polyene chain of the carotenoid backbone, resulting in extensive product peroxidation and degradation. Consequently, reaction yields in these legacy processes often stagnate at unacceptably low levels, typically ranging between 26% and 38%, which translates directly into inflated production costs and substantial waste generation. Furthermore, many conventional routes depend on Vitamin A aldehyde as a key precursor, a material known for its thermal instability and difficulty in large-scale preparation, creating a fragile supply chain bottleneck that jeopardizes production continuity and complicates inventory management for procurement teams.
The Novel Approach
In stark contrast, the methodology outlined in CN113321604A presents a robust alternative that addresses these systemic inefficiencies through precise chemical engineering. The core innovation lies in the substitution of non-selective strong oxidants with IBX (o-iodoxybenzoic acid) derivatives, which exhibit remarkable chemoselectivity by targeting specific hydroxyl groups while leaving the conjugated double bond system intact. This specificity effectively eliminates the pervasive issue of product peroxidation, allowing for significantly higher reaction yields and purity profiles without the need for complex downstream purification to remove oxidized byproducts. Additionally, the process incorporates a transition metal supported catalyst in the final stage to facilitate the isomerization of cis-isomers into the thermodynamically stable all-trans form. This catalytic step operates under mild thermal conditions and allows for catalyst recovery and reuse, transforming what was once a linear, wasteful process into a circular, cost-effective operation that aligns perfectly with modern green chemistry principles and cost reduction in vitamin manufacturing initiatives.
Mechanistic Insights into IBX-Mediated Oxidative Condensation
The heart of this advanced synthesis lies in the formation and subsequent reaction of the C20 phosphonium salt intermediate. The process initiates by reacting a Vitamin A derivative, such as Vitamin A acetate, with triphenylphosphine in the presence of a strong inorganic acid. This step generates the critical C20 phosphonium salt, a stable cationic species that serves as the nucleophilic partner in the subsequent coupling reaction. The structural integrity of this salt is paramount, as depicted in the chemical representation below, where the phosphonium center is stabilized by the surrounding organic framework and the counter-anion derived from the acid source.
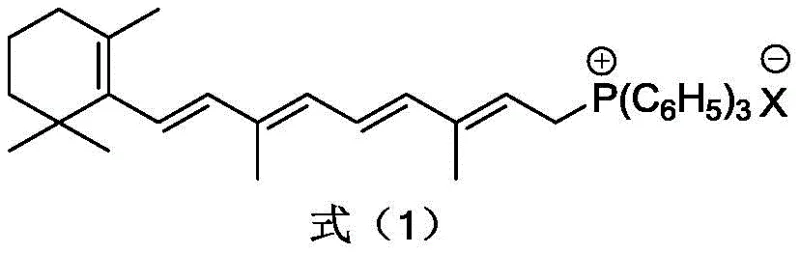
Following the formation of the phosphonium salt, the process advances to the oxidative condensation stage, which is the defining feature of this patent's novelty. Instead of traditional oxidants, the reaction utilizes an IBX high-iodine compound, specifically an o-iodoxybenzoic acid derivative, in conjunction with a base. The structure of this oxidant, shown below, features a hypervalent iodine center that acts as a mild yet effective oxygen transfer agent. The mechanism involves the selective oxidation of the alcoholic hydroxyl group (generated in situ or present in the substrate) to a carbonyl group, facilitating the Wittig-like coupling without inducing radical attacks on the polyene chain. This high degree of selectivity is the primary driver for the observed increase in yield and the reduction of impurities, as the IBX reagent simply does not possess the oxidative potential to degrade the sensitive carotenoid structure under the controlled low-temperature conditions employed.
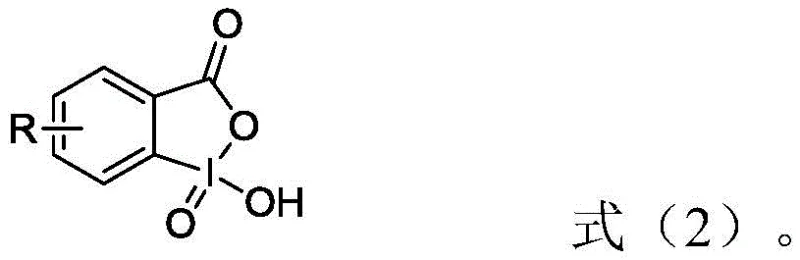
The final mechanistic component involves the stereochemical control of the product through transition metal catalysis. The initial condensation yields a mixture of cis and trans isomers, which is suboptimal for most commercial applications requiring the all-trans configuration for maximum biological activity and color intensity. By introducing a transition metal supported catalyst, such as rhodium or nickel dispersed on coconut shell activated carbon, the system lowers the activation energy required for cis-to-trans isomerization. The metal centers coordinate with the double bonds, facilitating rotation and relaxation into the more stable all-trans conformation. This step is crucial for achieving the high purity specifications demanded by the pharmaceutical and food industries, ensuring that the final high-purity beta-carotene meets rigorous regulatory standards without the need for extensive chromatographic separation.
How to Synthesize Beta-Carotene Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters, particularly temperature control and stoichiometry, to maximize the benefits of the IBX oxidation and catalytic isomerization. The process is designed to be scalable, moving from laboratory benchtop conditions to industrial reactors with minimal modification to the core chemical logic. Operators must ensure strict adherence to the specified molar ratios of the IBX oxidant relative to the phosphonium salt, as well as the precise loading of the transition metal catalyst, to maintain the delicate balance between reaction rate and selectivity. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for technical teams aiming to replicate these results in a production environment.
- React Vitamin A derivatives with triphenylphosphine and strong inorganic acid to form C20 phosphonium salt.
- Perform oxidative condensation using IBX (o-iodoxybenzoic acid) and base at low temperature to form cis-trans beta-carotene.
- Add transition metal supported catalyst (e.g., Rh/Carbon) to isomerize the mixture into high all-trans beta-carotene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this IBX-based synthesis route offers transformative advantages that extend far beyond simple yield improvements. The shift away from unstable raw materials like Vitamin A aldehyde to robust derivatives like Vitamin A acetate fundamentally de-risks the supply chain, ensuring consistent availability of starting materials even during market fluctuations. Furthermore, the elimination of harsh oxidants reduces the burden on waste treatment facilities and lowers the costs associated with hazardous material handling and disposal. The ability to recycle the transition metal catalyst represents a direct reduction in variable operating costs, as the expensive metal components do not need to be replenished after every batch. These factors combine to create a manufacturing process that is not only chemically superior but also economically resilient, offering commercial scale-up of complex carotenoids with predictable margins and reduced exposure to raw material volatility.
- Cost Reduction in Manufacturing: The implementation of the IBX oxidation system eliminates the need for expensive downstream purification steps typically required to remove peroxidized byproducts generated by traditional strong oxidants. By preventing product degradation at the source, the overall material throughput is maximized, meaning less raw material is wasted to produce the same amount of final product. Additionally, the recyclability of the transition metal supported catalyst significantly lowers the consumption of precious metals like rhodium, which are major cost drivers in catalytic processes. This closed-loop catalyst usage model ensures that the cost per kilogram of produced beta-carotene is substantially optimized, providing a clear financial advantage over legacy methods that rely on single-use reagents and generate high volumes of chemical waste.
- Enhanced Supply Chain Reliability: One of the most critical supply chain vulnerabilities in traditional beta-carotene synthesis is the reliance on Vitamin A aldehyde, a compound notorious for its instability and difficulty in long-term storage and transport. By utilizing Vitamin A acetate or alcohol as the primary feedstock, this new method leverages widely available, stable, and commercially mature raw materials that are easier to source globally. This switch mitigates the risk of production stoppages due to raw material degradation or supply shortages. Moreover, the mild reaction conditions, which avoid extreme temperatures and pressures, reduce the likelihood of equipment failure or safety incidents that could disrupt production schedules, thereby ensuring a steady and reliable flow of finished goods to meet customer demand without unexpected delays.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing a one-pot methodology that simplifies reactor operations and reduces the need for intermediate isolation steps. This streamlined workflow minimizes solvent usage and energy consumption, aligning with increasingly stringent environmental regulations regarding industrial emissions and effluent discharge. The use of environmentally friendly IBX oxidants, which degrade into benign o-iodobenzoic acid, further simplifies waste management compared to processes generating heavy metal sludge or chlorinated organic waste. For facilities aiming to expand capacity, this technology offers a clear path to reducing lead time for high-purity beta-carotene production while maintaining a smaller environmental footprint, making it an attractive option for companies committed to sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The technical nuances of this synthesis method often raise specific questions regarding operational feasibility, catalyst performance, and product quality assurance. The following answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing clarity on how this technology performs under real-world conditions. Understanding these details is essential for technical teams evaluating the integration of this process into their existing manufacturing portfolios.
Q: Why is IBX preferred over hypochlorite for beta-carotene synthesis?
A: IBX offers superior chemical selectivity, oxidizing only the hydroxyl group without attacking the sensitive polyene chain, thereby preventing peroxidation and significantly improving yield compared to strong oxidants like hypochlorite.
Q: Can the transition metal catalyst be recycled in this process?
A: Yes, the transition metal supported catalyst, such as rhodium on coconut shell activated carbon, can be filtered, washed, and reused for multiple batches without significant loss of catalytic activity, reducing overall production costs.
Q: What represents the main advantage of using Vitamin A acetate over Vitamin A aldehyde?
A: Vitamin A acetate is significantly more stable and easier to handle industrially than Vitamin A aldehyde, which is prone to instability and difficult preparation, thus ensuring a more reliable supply chain and consistent reaction outcomes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Carotene Supplier
The transition to advanced synthesis methodologies like the IBX-mediated route described in CN113321604A requires a partner with deep technical expertise and proven industrial capability. NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the precise temperature controls and catalyst handling requirements of this sophisticated chemistry, ensuring that every batch meets stringent purity specifications. With rigorous QC labs dedicated to monitoring impurity profiles and isomer ratios, we guarantee that our beta-carotene products deliver the consistent quality and performance that global brands demand for their nutritional and food applications.
We invite procurement leaders and R&D directors to engage with us to explore how this technology can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this greener, more efficient synthesis route. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments tailored to your volume requirements. Let us collaborate to secure a sustainable and cost-effective future for your beta-carotene supply needs.
