Advanced Catalytic Synthesis of Astaxanthin for Industrial Scale-Up and Commercial Viability
Introduction to Next-Generation Astaxanthin Manufacturing
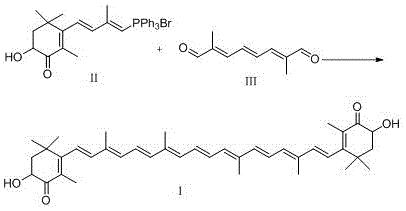
The global demand for high-purity astaxanthin, a potent fat-soluble carotenoid with exceptional antioxidant properties, continues to surge across the pharmaceutical, nutraceutical, and aquaculture sectors. As industries seek more sustainable and efficient production methodologies, the technical breakthroughs detailed in patent CN114369048A offer a compelling roadmap for modernizing synthesis capabilities. This patent introduces a refined catalytic approach that addresses long-standing challenges in the Wittig reaction pathway, specifically targeting the coupling of C15 phosphonium salts with C10 dialdehydes. By optimizing the base system and reaction conditions, this technology enables the production of astaxanthin with superior isomeric purity and reduced environmental impact. For R&D leaders and procurement strategists, understanding the nuances of this catalytic system is critical for securing a competitive edge in the supply of this high-value active ingredient.
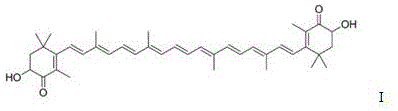
The structural complexity of astaxanthin, characterized by its conjugated polyene chain and terminal ionone rings, necessitates precise synthetic control to maintain stability and bioactivity. The innovation lies not merely in the coupling itself, but in the sophisticated management of the reaction environment to prevent degradation and isomerization issues that plague traditional routes. As we delve deeper into the technical specifics, it becomes evident that this method represents a significant leap forward in the reliable manufacturing of complex carotenoids, aligning perfectly with the rigorous quality standards demanded by top-tier global enterprises.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of astaxanthin via the 2C15 + C10 route has been fraught with significant technical and safety hurdles that compromise both yield and operational safety. Traditional protocols often rely heavily on strong bases such as sodium methylate or hydroxides without adequate buffering, leading to harsh reaction conditions that are difficult to control on a large scale. These aggressive environments frequently promote the formation of undesirable by-products like astacin and hemishrimps, which drastically reduce the overall yield of the target molecule and complicate downstream purification processes. Furthermore, alternative literature methods have attempted to utilize reagents like butylene oxide to moderate the reaction; however, this introduces severe safety liabilities due to the carcinogenic and explosive nature of such compounds, rendering them unsuitable for green, continuous chemical production. The inability to precisely manage the deprotonation kinetics in these legacy systems results in inconsistent batch quality and elevated production costs associated with waste disposal and extensive purification.
The Novel Approach
In stark contrast, the methodology disclosed in CN114369048A revolutionizes the process by introducing a dual-base system that harmonizes reaction kinetics with safety and efficiency. This novel approach strategically employs weak bases, such as sodium acetate, sodium oxalate, or potassium formate, acting effectively as phase transfer catalysts within the Wittig reaction matrix. By integrating these weak organic acid salts alongside a controlled addition of strong alkali liquor, the process creates a buffered environment that facilitates the smooth progression of the coupling reaction while suppressing side reactions. The result is a synthesis pathway that operates under milder, more controllable conditions, typically between -25°C and -10°C, which significantly mitigates the risk of thermal runaway or uncontrolled exotherms. This strategic modification not only enhances the selectivity for the desired all-trans isomer but also simplifies the operational workflow, making it an ideal candidate for robust commercial scale-up of complex carotenoid intermediates.
Mechanistic Insights into Weak Base-Catalyzed Wittig Coupling
The core mechanistic advantage of this patented process lies in the intricate interplay between the weak base catalyst and the strong alkali reagent during the ylide formation and subsequent nucleophilic attack. In the initial phase, the weak base, such as sodium acetate, interacts with the acidic protons generated during the reaction cycle, effectively capturing them to form the corresponding weak acid. This proton scavenging action drives the equilibrium of the Wittig reaction forward, ensuring high conversion rates of the C15 phosphonium salt and C10 dialdehyde precursors. Simultaneously, the presence of the weak base moderates the basicity of the medium, preventing the over-deprotonation or degradation of the sensitive polyene chain that often occurs in purely strong-base environments. The subsequent reaction of the generated weak acid with the added strong alkali (e.g., sodium hydroxide in ethanol) regenerates the weak base catalyst, establishing a sustainable catalytic cycle that maintains optimal pH levels throughout the reaction duration. This dynamic buffering capacity is crucial for minimizing the formation of cis-isomers and oxidative by-products, thereby preserving the integrity of the conjugated system essential for astaxanthin's biological function.
Furthermore, the impurity control mechanism is deeply rooted in the precise temperature regulation and the specific solvent system employed, which typically involves a mixture of dichloromethane and ethanol with a controlled water content of 8-10%. This specific solvent polarity and water content facilitate the solubility of the ionic intermediates while stabilizing the transition states involved in the oxaphosphetane formation and decomposition. By maintaining the reaction temperature strictly between -25°C and -10°C during the dropwise addition of the alkali liquor, the process kinetically favors the formation of the thermodynamically stable all-(E)-isomer. Post-reaction thermal isomerization in refluxing ethanol further ensures that any minor cis-isomers formed are converted to the desired trans-configuration, resulting in a final product profile where astacin content is reduced to less than 0.4% and semiastacin to less than 0.9%. This level of impurity suppression is a direct consequence of the mechanistic stability provided by the weak base catalyst system, offering a distinct advantage over unbuffered strong base methods.
How to Synthesize Astaxanthin Efficiently
Implementing this advanced catalytic synthesis requires strict adherence to the optimized parameters regarding reagent ratios, temperature gradients, and addition rates to maximize yield and purity. The process begins with the preparation of a homogeneous reaction mixture containing the C15 phosphonium salt and C10 dialdehyde in a dichloromethane and ethanol solvent system, supplemented with the selected weak base catalyst. It is imperative to cool this mixture to the specified sub-zero range before initiating the addition of the strong alkali liquor, as thermal control is the primary determinant of stereochemical outcome. The following guide outlines the critical operational phases derived from the patent data, serving as a foundational reference for process engineers aiming to replicate these high-efficiency results in a pilot or production setting.
- Dissolve Compound II (C15 phosphonium salt) and Compound III (C10 dialdehyde) in a solvent mixture of dichloromethane and ethanol containing 8-10% water, then add a weak base such as sodium acetate or sodium oxalate.
- Cool the mixture to between -25°C and -10°C, then continuously dropwise add a strong alkali liquor (e.g., sodium hydroxide in ethanol) while maintaining the low temperature to initiate the Wittig coupling.
- Upon completion, neutralize with acid, extract the organic phase, concentrate, and perform thermal isomerization in refluxing ethanol to convert the product into the all-(E)-isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic synthesis method translates into tangible strategic benefits that extend far beyond simple yield improvements. The elimination of hazardous and expensive reagents like butylene oxide removes a significant liability from the supply chain, reducing the regulatory burden and insurance costs associated with handling carcinogenic materials. Moreover, the use of commodity weak bases such as sodium acetate and sodium oxalate ensures a stable and cost-effective supply of catalysts, insulating the production process from the volatility of specialized reagent markets. The simplified workup procedure, characterized by straightforward neutralization and extraction, reduces the consumption of solvents and energy, contributing to a leaner manufacturing footprint. These factors collectively drive a substantial reduction in the cost of goods sold (COGS), making the final astaxanthin product more competitive in price-sensitive markets like animal feed and bulk nutraceuticals without compromising on quality specifications.
- Cost Reduction in Manufacturing: The replacement of expensive and dangerous reagents with inexpensive, widely available weak organic acid salts fundamentally alters the cost structure of astaxanthin production. By avoiding the need for specialized safety infrastructure required for explosive epoxides and minimizing the formation of hard-to-remove by-products, the process significantly lowers both raw material expenditures and downstream purification costs. The enhanced selectivity means less starting material is wasted on side reactions, directly improving the atom economy and overall process efficiency. This economic optimization allows manufacturers to offer high-purity astaxanthin at a more aggressive price point, enhancing market penetration potential.
- Enhanced Supply Chain Reliability: The reliance on common chemical feedstocks like sodium hydroxide, ethanol, and simple carboxylate salts ensures a robust and resilient supply chain that is less susceptible to geopolitical disruptions or single-source bottlenecks. The mild reaction conditions and the use of standard solvents like dichloromethane and ethanol facilitate easier sourcing and logistics management compared to processes requiring exotic or highly regulated chemicals. This reliability is crucial for maintaining consistent delivery schedules to major pharmaceutical and feed clients, thereby strengthening long-term contractual relationships and reducing the risk of production stoppages due to material shortages.
- Scalability and Environmental Compliance: The inherent safety and controllability of this weak-base catalyzed route make it exceptionally well-suited for scaling from pilot batches to multi-ton commercial production without the need for disproportionate increases in safety mitigation measures. The reduction in toxic by-products and the avoidance of carcinogenic reagents align perfectly with increasingly stringent global environmental regulations, simplifying the permitting process and reducing waste treatment costs. This green chemistry profile not only future-proofs the manufacturing asset against tightening regulatory frameworks but also enhances the brand value of the end-product for eco-conscious consumers and corporate buyers seeking sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic astaxanthin synthesis method, providing clarity for stakeholders evaluating its adoption. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is essential for assessing the feasibility of integrating this technology into existing production lines or new facility designs.
Q: How does the novel weak base system improve astaxanthin purity compared to conventional methods?
A: The use of weak bases like sodium acetate or sodium oxalate acts as a phase transfer catalyst and buffer, significantly reducing the formation of by-products such as astacin and semiastacin to levels below 0.9% and 0.4% respectively, compared to higher impurity profiles in strong-base-only methods.
Q: What are the safety advantages of this catalytic synthesis route?
A: This method eliminates the need for hazardous reagents like butylene oxide, which is a known carcinogen and flammable explosive, replacing it with safe, inexpensive, and green weak organic acid salts that are easier to handle in continuous chemical production.
Q: What is the expected isomeric composition of the final astaxanthin product?
A: Through a final thermal isomerization step involving heating in ethanol, the process ensures the conversion of the crude product into the highly desired all-(E)-isomer (all-trans astaxanthin), achieving contents typically exceeding 96%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Astaxanthin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalytic synthesis methods described in CN114369048A and are fully equipped to leverage this technology for our global partners. As a premier CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. Our state-of-the-art facilities are designed to handle the precise temperature controls and sensitive reaction conditions required for high-purity carotenoid synthesis, backed by rigorous QC labs that guarantee stringent purity specifications for every batch. We are committed to delivering astaxanthin intermediates and finished products that meet the highest international standards, supporting your R&D and commercial goals with unmatched reliability.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits this process offers for your specific application. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as your trusted partner in the sustainable and efficient manufacturing of high-value fine chemicals.
