Advanced Astaxanthin Manufacturing: A Safe, High-Yield C15+C10+C15 Synthetic Strategy
Introduction to Next-Generation Astaxanthin Synthesis
The global demand for high-purity astaxanthin, a potent keto-carotenoid with significant applications in aquaculture feed and nutraceuticals, necessitates manufacturing processes that balance efficiency with rigorous safety standards. Patent CN113461583B introduces a transformative synthesis method that addresses the critical limitations of prior art by utilizing a C15+C10+C15 convergent strategy. This innovative approach replaces hazardous alkynol-based chemistries with a safer, acetal-protected pathway, ensuring a reliable astaxanthin supplier can meet increasing market volume without compromising on operational safety. The core of this technology lies in the strategic manipulation of C15 intermediates, specifically converting 4-halo-2-methyl-2-butene-1-aldehyde into stable acetals before coupling, thereby mitigating side reactions and enhancing overall process robustness.
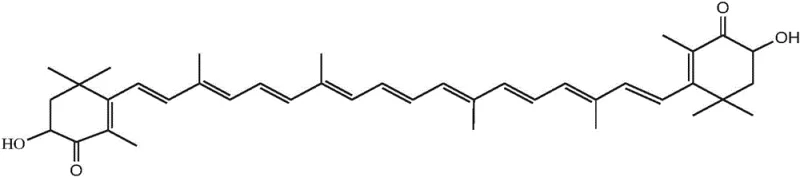
By focusing on the synthesis of key intermediates such as Compound 5 (a phosphonium salt) and Compound 6 (a C10 dialdehyde), this method achieves superior selectivity. The elimination of dangerous reagents like liquid ammonia and metallic lithium not only lowers the barrier for entry for manufacturing facilities but also significantly reduces the environmental footprint associated with waste disposal. For procurement managers and supply chain heads, this represents a pivotal shift towards a more sustainable and predictable sourcing model for high-purity carotenoid intermediates, ensuring continuity of supply in a volatile chemical market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, industrial astaxanthin synthesis has been plagued by reliance on perilous chemical transformations that complicate scale-up and increase liability. For instance, earlier routes disclosed in patents such as CN200410073427.0 utilized alkynol precursors prepared from acetylene and liquid ammonia, creating severe safety hazards due to the explosive nature of these feedstocks. Furthermore, the Roche C15+C10+C15 route, while effective, often necessitates the use of lithium reagents and high-pressure hydrogenation, which require specialized equipment and stringent safety protocols. Another significant drawback in traditional methodologies is the issue of selectivity during halogenation; specifically, the bromination of hydroxyl groups often leads to the formation of unwanted dibromo by-products, complicating purification and reducing overall yield.
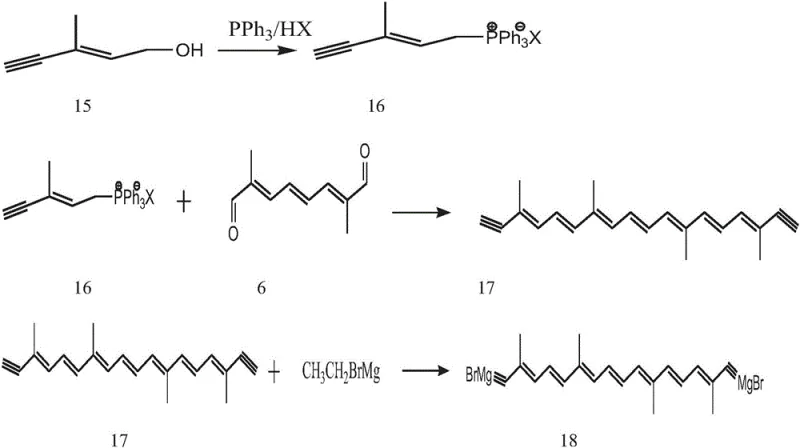
These conventional pathways also suffer from operational complexity, often requiring cryogenic conditions that are energy-intensive and difficult to maintain on a multi-ton scale. The presence of transition metals or reactive organometallic species necessitates extensive downstream processing to remove trace impurities, which is a critical concern for R&D directors focused on API intermediate quality. Consequently, the industry has long sought a method that retains the stereochemical precision of the polyene chain while simplifying the reaction engineering to allow for cost reduction in carotenoid manufacturing.
The Novel Approach
The methodology outlined in Patent CN113461583B offers a decisive break from these constraints by employing a modular assembly of stable intermediates. The process begins with the protection of the aldehyde functionality in Compound 20 to form the dimethyl acetal Compound 21, which serves as a robust building block resistant to premature oxidation or polymerization. This is coupled with a methylenated beta-ionone derivative (Compound 22), generated via a Nysted reagent and titanium tetrachloride mediated reaction, which avoids the use of hazardous lithium metal. The convergence of these fragments creates a C15 unit that is subsequently converted into a phosphonium salt, ready for the final Wittig condensation.
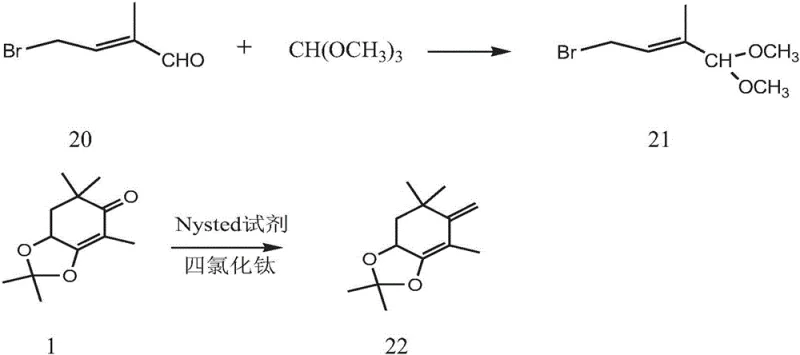
This novel approach streamlines the commercial scale-up of complex carotenoids by utilizing readily available starting materials and standard laboratory equipment. The reaction conditions are notably milder, with key coupling steps occurring at manageable temperatures between -40°C and 55°C, eliminating the need for extreme cryogenics. By avoiding the formation of dibromo impurities and utilizing a high-yield Wittig reaction for the final chain extension, this route ensures a cleaner crude product profile. For stakeholders, this translates to a drastic simplification of the purification workflow, directly impacting the bottom line through reduced solvent consumption and shorter cycle times.
Mechanistic Insights into Lewis Acid Catalyzed Coupling and Wittig Olefination
The heart of this synthetic innovation lies in the precise control of carbon-carbon bond formation through Lewis acid catalysis and phosphonium ylide chemistry. In the coupling of Compound 21 and Compound 22, catalysts such as anhydrous ferric chloride (FeCl3), zinc chloride, or boron trifluoride act as potent Lewis acids to activate the acetal functionality. This activation generates a transient carbocation species that undergoes electrophilic attack on the electron-rich exocyclic double bond of Compound 22. The choice of catalyst is critical; experimental data suggests that FeCl3 provides an optimal balance of reactivity and selectivity, minimizing oligomerization side reactions that often plague cationic polymerizations of conjugated systems. This mechanistic pathway ensures the preservation of the delicate stereochemistry required for the biological activity of the final astaxanthin molecule.
Following the coupling and subsequent hydrolysis to reveal the aldehyde, the formation of the phosphonium salt (Compound 5) sets the stage for the final chain elongation. The reaction with triphenylphosphine proceeds via a nucleophilic substitution mechanism, displacing the halide to form the stable salt. In the final step, the treatment of this salt with a sodium alkoxide base generates the reactive ylide in situ, which then attacks the C10 dialdehyde (Compound 6). This double Wittig reaction is highly stereoselective, favoring the formation of the all-trans polyene chain characteristic of natural astaxanthin. The use of sodium methoxide or ethoxide in alcoholic solvents facilitates this transformation under homogeneous conditions, ensuring rapid kinetics and high conversion rates without the need for heterogeneous catalysts that could introduce metal contaminants.
How to Synthesize Astaxanthin Efficiently
The execution of this synthesis requires careful attention to stoichiometry and temperature control across six distinct operational stages. The process is designed to be telescoped where possible, minimizing the isolation of unstable intermediates. Initially, the protection of the C5 aldehyde is performed at moderate temperatures (30-35°C) to ensure complete conversion to the acetal. Subsequently, the methylenation step requires strict low-temperature control (-40°C) during the addition of titanium tetrachloride to manage the exotherm and prevent decomposition of the Nysted reagent complex. The coupling reaction similarly demands cooling to -20°C to maintain selectivity, while the final Wittig condensation is optimized at -15 to -20°C to prevent isomerization of the sensitive polyene chain. Detailed standardized operating procedures for these critical control points are essential for reproducibility.
- Protect 4-halo-2-methyl-2-butene-1-aldehyde (Compound 20) with trimethyl orthoformate to form the stable acetal Compound 21.
- Perform methylenation on the protected beta-ionone derivative (Compound 1) using Nysted reagent and titanium tetrachloride to generate the exocyclic alkene Compound 22.
- Couple Compound 21 and Compound 22 using a Lewis acid catalyst (e.g., FeCl3) to form Compound 23, followed by hydrolysis and phosphonium salt formation to yield Compound 5.
- Execute a double Wittig reaction between two equivalents of Compound 5 and one equivalent of C10 dialdehyde (Compound 6) under basic conditions to finalize the astaxanthin polyene chain.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the radical simplification of the raw material portfolio. By eliminating the need for specialized, high-hazard reagents like liquid ammonia and acetylene, facilities can source inputs from a broader, more competitive supplier base, thereby reducing dependency on single-source vendors. This diversification inherently strengthens supply chain resilience, mitigating the risk of production stoppages due to raw material shortages. Furthermore, the stability of the acetal intermediates allows for potential inventory buffering, enabling manufacturers to decouple production steps and optimize facility utilization rates without the fear of rapid degradation.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive and dangerous reagents, as well as the reduction in energy consumption associated with cryogenic operations. By operating at higher average temperatures compared to lithium-mediated routes, the process significantly lowers utility costs for refrigeration and heating. Additionally, the high selectivity of the Lewis acid coupling minimizes the formation of hard-to-remove impurities, which reduces the load on downstream purification units such as chromatography columns or crystallization tanks. This efficiency gain translates directly into lower variable costs per kilogram of finished product, enhancing margin potential in a competitive market.
- Enhanced Supply Chain Reliability: The use of commodity chemicals such as trimethyl orthoformate, triphenylphosphine, and common solvents like dichloromethane and ethanol ensures a stable and predictable supply chain. Unlike routes dependent on custom-synthesized alkynols which may have long lead times, the precursors for this method are widely available from multiple global chemical distributors. This accessibility reduces lead time for high-purity carotenoid intermediates, allowing for more agile response to market demand fluctuations. The robustness of the chemistry also implies fewer batch failures, ensuring a consistent output of material that meets stringent quality specifications.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this route is markedly superior. The absence of heavy metals like chromium or toxic gases like ammonia simplifies waste treatment protocols and reduces the regulatory burden on the manufacturing site. The solvents used are standard and easily recoverable via distillation, supporting green chemistry initiatives and reducing the volume of hazardous waste requiring disposal. This environmental compatibility facilitates easier permitting for capacity expansion, making it an ideal candidate for commercial scale-up of complex fine chemical intermediates in regions with strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this astaxanthin synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in Patent CN113461583B, offering clarity on how this method outperforms legacy technologies in terms of safety, yield, and operational simplicity. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer.
Q: How does this synthesis method improve safety compared to traditional alkynol routes?
A: Traditional routes often rely on hazardous raw materials such as acetylene, liquid ammonia, and metallic lithium, which pose significant explosion and handling risks. This patented method utilizes stable acetal intermediates and mild Lewis acid catalysis, completely eliminating the need for high-risk cryogenic reagents and high-pressure hydrogenation steps.
Q: What are the purity and yield advantages of this C15+C10+C15 approach?
A: The process demonstrates exceptional control over impurity profiles, specifically avoiding the formation of dibromo by-products common in older bromination-selective pathways. Experimental data indicates final product purity exceeding 96% with high overall yields, attributed to the efficient protection-deprotection strategy of the C15 intermediates.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the route is designed for scalability. It operates under moderate temperature conditions (ranging from -40°C to 55°C depending on the step) and utilizes common industrial solvents like dichloromethane, toluene, and ethanol. The avoidance of exotic reagents ensures a robust supply chain suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Astaxanthin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a superior synthetic route requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of astaxanthin intermediate meets the exacting standards required for feed and pharmaceutical applications, providing our clients with peace of mind regarding product quality and regulatory compliance.
We invite forward-thinking organizations to collaborate with us to leverage this advanced synthesis technology for their supply chains. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing processes can drive value and efficiency for your astaxanthin sourcing needs.
