Advanced Manufacturing of Chiral Alpha-Methyl Arylethylamine for Global Pharmaceutical Supply Chains
Advanced Manufacturing of Chiral Alpha-Methyl Arylethylamine for Global Pharmaceutical Supply Chains
The global demand for high-purity chiral intermediates continues to surge, driven by the expanding markets for respiratory and oncology therapeutics. A pivotal advancement in this sector is detailed in Chinese Patent CN112920053B, which discloses a robust preparation method for chiral alpha-methyl arylethylamine. This technology addresses critical bottlenecks in the synthesis of key pharmacophores, such as (R)-α-methyl-4-methoxyphenethylamine, a vital building block for asthma medications like Formoterol and Carmoterol. Furthermore, the methodology extends to the production of (R)-3-(2-aminopropyl)-1H-indole, an essential fragment for the breast cancer agent AZD9496. By leveraging a chiral pool strategy starting from Boc-amino acid methyl esters, this innovation bypasses the inefficiencies of traditional resolution techniques. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic advantages and scalability of this route is paramount for securing long-term supply chain resilience.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of chiral alpha-methyl arylethylamines has been plagued by significant operational and economic hurdles. Prior art, such as the route described in WO2017172957, relies heavily on palladium-catalyzed high-pressure hydrogenation. This dependency introduces severe safety hazards related to high-pressure reactors and necessitates expensive noble metal catalysts, which complicates impurity control and increases raw material costs. Alternative strategies involving diastereomeric resolution, as seen in older literature like Journal of Medicinal Chemistry (1988), suffer from poor atom economy. These methods typically generate a 1:1 mixture of isomers, requiring energy-intensive column chromatography or multiple recrystallizations to isolate the desired enantiomer. Consequently, nearly half of the synthesized material is discarded as waste, leading to substantial environmental burdens through silica gel disposal and drastically inflating the cost of goods sold (COGS). Such inefficiencies render these legacy processes unsuitable for modern, large-scale green manufacturing standards.
The Novel Approach
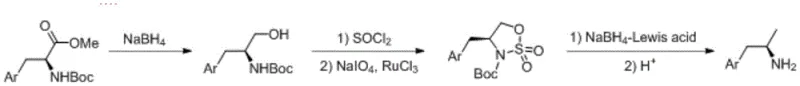
In stark contrast, the methodology outlined in CN112920053B presents a streamlined, three-step sequence that fundamentally redefines process efficiency. The route initiates with the reduction of commercially available Boc-amino acid methyl esters to Boc-amino alcohols, followed by a sophisticated oxidation step to generate a sulfonamide intermediate. The culmination of the synthesis involves a Lewis acid-promoted reduction that preserves stereochemical integrity without the need for external chiral catalysts during the bond-forming steps. This approach ensures that the chirality is inherited directly from the inexpensive starting material, theoretically allowing for 100% conversion to the desired optical isomer. By eliminating the need for high-pressure hydrogenation and chromatographic separation, the process not only enhances operator safety but also drastically simplifies the downstream processing workflow. This makes it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates, offering a clear pathway to cost reduction in API manufacturing.
Mechanistic Insights into Ruthenium-Catalyzed Oxidation and Lewis Acid Reduction
The core chemical innovation lies in the transformation of the amino alcohol into a sulfonamide compound via a ruthenium-catalyzed oxidation system. In this critical step, the Boc-amino alcohol reacts with thionyl chloride to form a chloro-intermediate, which is subsequently oxidized using sodium periodate in the presence of a catalytic amount of ruthenium trichloride trihydrate. This specific oxidative protocol is highly selective, ensuring that the sensitive indole or aromatic moieties remain intact while efficiently generating the sulfonamide functionality. The choice of ruthenium as a catalyst is strategic; it operates under mild conditions (0-5°C) and allows for precise control over the reaction kinetics, minimizing the formation of over-oxidized byproducts. For quality control teams, this translates to a cleaner reaction profile and a significant reduction in difficult-to-remove impurities, thereby facilitating higher final purity specifications without extensive purification loops.
Following the formation of the sulfonamide, the stereochemistry is locked in place during the reduction phase. The patent specifies the use of sodium borohydride in conjunction with a Lewis acid, such as anhydrous lithium chloride or zinc chloride. This combination activates the sulfonamide group towards hydride attack, enabling the cleavage of the sulfur-nitrogen bond under relatively mild thermal conditions (50-120°C). Unlike traditional hydride reductions that might require cryogenic temperatures or harsh reagents, this Lewis acid-promoted system provides a balanced reactivity that prevents racemization. The final deprotection step utilizes acidic conditions to remove the Boc group, yielding the free amine as a salt. This mechanistic pathway ensures that the optical purity established at the beginning of the synthesis is maintained throughout, resulting in products with enantiomeric excesses suitable for direct use in GMP pharmaceutical production.
How to Synthesize (R)-α-Methyl-4-Methoxyphenethylamine Efficiently
The practical implementation of this synthesis route is designed for straightforward execution in standard multipurpose chemical reactors. The process begins with the dissolution of the chiral starting material in an alcoholic solvent, followed by the controlled addition of sodium borohydride to generate the amino alcohol precursor. Subsequent steps involve careful temperature management during the sulfonylation and oxidation phases to maximize yield and minimize side reactions. The final reduction and deprotection are conducted in etheral solvents compatible with Lewis acids, ensuring complete conversion to the target amine hydrochloride salt. While the general principles are robust, precise adherence to molar ratios and temperature profiles is essential for reproducibility. The detailed standardized synthesis steps see the guide below for specific operational parameters optimized for industrial throughput.
- Reduce Boc-amino acid methyl ester with sodium borohydride to obtain Boc-amino alcohol.
- React the amino alcohol with thionyl chloride followed by sodium periodate oxidation catalyzed by ruthenium trichloride to form a sulfonamide compound.
- Perform Lewis acid-promoted reduction with sodium borohydride followed by acidic deprotection to yield the target chiral amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers transformative benefits that extend beyond mere technical feasibility. The primary advantage is the drastic simplification of the supply chain risk profile. By removing the requirement for high-pressure hydrogenation equipment, facilities can utilize standard glass-lined or stainless steel reactors, significantly lowering capital expenditure (CAPEX) barriers for production. Furthermore, the reliance on cheap, commodity-grade starting materials like Boc-amino acid esters insulates the manufacturing process from the volatility associated with specialized chiral catalysts or resolving agents. This stability ensures consistent lead times and reduces the likelihood of production stoppages due to raw material shortages. The elimination of silica gel chromatography also means a substantial reduction in hazardous solid waste disposal costs, aligning production with increasingly stringent environmental regulations and sustainability goals.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the avoidance of yield losses inherent in resolution techniques. Traditional methods often discard up to half of the produced material as the unwanted enantiomer, whereas this chiral pool approach theoretically utilizes the entire mass of the starting material for the desired product. Additionally, the replacement of expensive palladium catalysts with inexpensive sodium borohydride and ruthenium salts (used in catalytic amounts) results in a significantly lower bill of materials. The simplified workup procedures, which rely on crystallization and extraction rather than column chromatography, further reduce labor hours and solvent consumption, leading to substantial cost savings in the overall manufacturing budget.
- Enhanced Supply Chain Reliability: From a logistics perspective, the robustness of this synthetic route enhances supply continuity. The reagents employed, such as thionyl chloride, sodium periodate, and common organic solvents, are widely available from multiple global suppliers, reducing dependency on single-source vendors. The mild reaction conditions also decrease the wear and tear on production equipment, leading to higher asset availability and fewer unplanned maintenance downtimes. For buyers seeking a reliable pharmaceutical intermediate supplier, this translates to a more predictable delivery schedule and the ability to scale orders rapidly in response to market demand without compromising on quality or lead time.
- Scalability and Environmental Compliance: The process is inherently designed for large-scale industrial production, avoiding the pitfalls of laboratory-only techniques. The absence of high-pressure steps removes a major safety bottleneck that often limits batch sizes in traditional hydrogenation processes. Moreover, the high atom economy and the generation of minimal solid waste make this method environmentally superior. By avoiding the massive volumes of silica waste associated with chromatographic purification, manufacturers can significantly lower their environmental footprint. This compliance with green chemistry principles not only mitigates regulatory risks but also enhances the corporate social responsibility profile of the supply chain, a factor of increasing importance to end-user pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral amine synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a transparent view of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this new method improve safety compared to traditional hydrogenation?
A: The patented process replaces high-pressure palladium-catalyzed hydrogenation with a mild sodium borohydride reduction system promoted by Lewis acids, significantly eliminating explosion risks and high-pressure equipment requirements.
Q: Does this synthesis route require column chromatography for purification?
A: No, the method utilizes chiral starting materials to directly obtain single optical isomers, completely avoiding the need for column chromatography resolution and the associated generation of silica solid waste.
Q: What are the key pharmaceutical applications for these intermediates?
A: These chiral amines are critical fragments for major drugs such as Formoterol and Carmoterol for asthma treatment, as well as AZD9496 for breast cancer therapy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Alpha-Methyl Arylethylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation. Whether your project requires the specific (R)-α-methyl-4-methoxyphenethylamine derivative for respiratory drugs or custom analogues for oncology pipelines, our infrastructure is designed to support your most demanding requirements with precision and reliability.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and accelerate your time to market.
